Ann Rehabil Med.
2019 Feb;43(1):45-53. 10.5535/arm.2019.43.1.45.
Optimal Ultrasonographic Measurements for Diagnosing Carpal Tunnel Syndrome in Patients With Diabetic Sensorimotor Polyneuropathy: A Case-Control Study
- Affiliations
-
- 1Department of Physical Medicine and Rehabilitation, Soonchunhyang University Seoul Hospital, Soonchunhyang University College of Medicine, Seoul, Korea. 115881@schmc.ac.kr
- KMID: 2440951
- DOI: http://doi.org/10.5535/arm.2019.43.1.45
Abstract
OBJECTIVE
To investigate the optimal sonographic method for diagnosing carpal tunnel syndrome (CTS) in patients with diabetic sensorimotor polyneuropathy (DSP).
METHODS
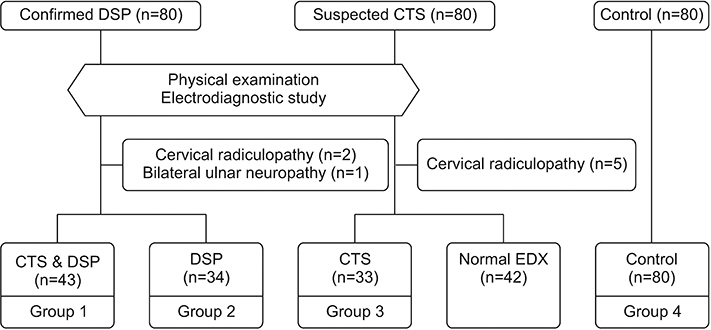
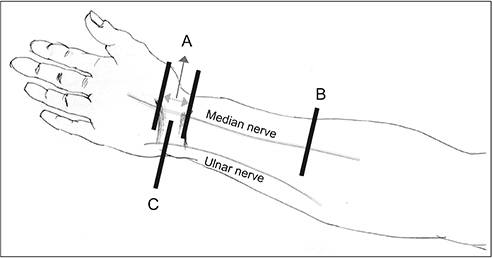
A total of 190 participants were divided into four groups based on DSP history and electrodiagnostic results of CTS. The absolute parameters were measured at baseline and the relative values were calculated: maximal cross-sectional area (CSA) of the median nerve throughout the carpal tunnel (Mmax), median nerve CSA at the forearm level (Mf), ulnar nerve CSA at the pisiform level (Upi), difference between Mmax and Mf (∆MM), and difference between Mmax and Upi (∆MU). Then, the optimal ultrasonographic parameters for diagnosing CTS, according to the presence of DSP, using absolute and relative cutoff values were analyzed.
RESULTS
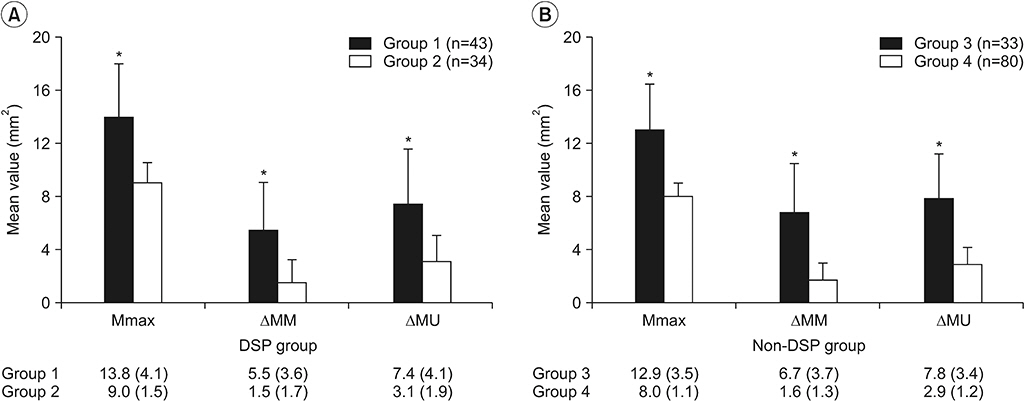
Median and ulnar nerve CSAs were significantly larger in the DSP group than in the control group. In the DSP participants, the mean Mmax, ∆MM, and ∆MU values were significantly larger in patients with both DSP and CTS than in patients with DSP only. The Mmax thresholds of 9.5 mm² in the control group and 11.5 mm² in the DSP group showed the greatest sensitivity and specificity for diagnosing CTS. The ∆MM thresholds of 2.5 mm² and ∆MU thresholds of 4.5 mm² had the greatest sensitivity and specificity in both the DSP and control groups.
CONCLUSION
Measurement of Mmax, ∆MM and ∆MU is an optimal ultrasonographic evaluation method for diagnosing CTS in patients with DSP.
Keyword
MeSH Terms
Figure
Reference
-
1. Gelberman RH, Eaton RG, Urbaniak JR. Peripheral nerve compression. Instr Course Lect. 1994; 43:31–53.
Article2. Dyck PJ, Kratz KM, Karnes JL, Litchy WJ, Klein R, Pach JM, et al. The prevalence by staged severity of various types of diabetic neuropathy, retinopathy, and nephropathy in a population-based cohort: the Rochester Diabetic Neuropathy Study. Neurology. 1993; 43:817–24.
Article3. Dyck PJ, Albers JW, Andersen H, Arezzo JC, Biessels GJ, Bril V, et al. Diabetic polyneuropathies: update on research definition, diagnostic criteria and estimation of severity. Diabetes Metab Res Rev. 2011; 27:620–8.
Article4. Atroshi I, Gummesson C, Johnsson R, Ornstein E, Ranstam J, Rosen I. Prevalence of carpal tunnel syndrome in a general population. JAMA. 1999; 282:153–8.
Article5. Wong SM, Griffith JF, Hui AC, Lo SK, Fu M, Wong KS. Carpal tunnel syndrome: diagnostic usefulness of sonography. Radiology. 2004; 232:93–9.
Article6. Abrishamchi F, Zaki B, Basiri K, Ghasemi M, Mohaghegh M. A comparison of the ultrasonographic median nerve cross-sectional area at the wrist and the wrist-to-forearm ratio in carpal tunnel syndrome. J Res Med Sci. 2014; 19:1113–7.7. Koyuncuoglu HR, Kutluhan S, Yesildag A, Oyar O, Guler K, Ozden A. The value of ultrasonographic measurement in carpal tunnel syndrome in patients with negative electrodiagnostic tests. Eur J Radiol. 2005; 56:365–9.
Article8. Lee D, van Holsbeeck MT, Janevski PK, Ganos DL, Ditmars DM, Darian VB. Diagnosis of carpal tunnel syndrome: ultrasound versus electromyography. Radiol Clin North Am. 1999; 37:859–72.9. Wiesler ER, Chloros GD, Cartwright MS, Smith BP, Rushing J, Walker FO. The use of diagnostic ultrasound in carpal tunnel syndrome. J Hand Surg Am. 2006; 31:726–32.
Article10. Yasuda H, Sanada M, Kitada K, Terashima T, Kim H, Sakaue Y, et al. Rationale and usefulness of newly devised abbreviated diagnostic criteria and staging for diabetic polyneuropathy. Diabetes Res Clin Pract. 2007; 77 Suppl 1:S178–83.
Article11. Watanabe T, Ito H, Morita A, Uno Y, Nishimura T, Kawase H, et al. Sonographic evaluation of the median nerve in diabetic patients: comparison with nerve conduction studies. J Ultrasound Med. 2009; 28:727–34.12. Hsu WC, Chiu YH, Chen WH, Chiu HC, Liou HH, Chen TH. Simplified electrodiagnostic criteria of diabetic polyneuropathy in field study (KCIS No. 14). Neuroepidemiology. 2007; 28:50–5.
Article13. Yagci I, Gunduz OH, Sancak S, Agirman M, Mesci E, Akyuz G. Comparative electrophysiological techniques in the diagnosis of carpal tunnel syndrome in patients with diabetic polyneuropathy. Diabetes Res Clin Pract. 2010; 88:157–63.
Article14. Werner RA, Andary M. Electrodiagnostic evaluation of carpal tunnel syndrome. Muscle Nerve. 2011; 44:597–607.
Article15. Giannini F, Cioni R, Mondelli M, Padua R, Gregori B, D’Amico P, et al. A new clinical scale of carpal tunnel syndrome: validation of the measurement and clinical-neurophysiological assessment. Clin Neurophysiol. 2002; 113:71–7.
Article16. Kim LN, Kwon HK, Moon HI, Pyun SB, Lee HJ. Sonography of the median nerve in carpal tunnel syndrome with diabetic neuropathy. Am J Phys Med Rehabil. 2014; 93:897–907.
Article17. Jakobsen J. Peripheral nerves in early experimental diabetes: expansion of the endoneurial space as a cause of increased water content. Diabetologia. 1978; 14:113–9.18. Lee D, Dauphinee DM. Morphological and functional changes in the diabetic peripheral nerve: using diagnostic ultrasound and neurosensory testing to select candidates for nerve decompression. J Am Podiatr Med Assoc. 2005; 95:433–7.19. Gabbay KH. Role of sorbitol pathway in neuropathy. In : Camerini-Dgvalos RA, Cole HS, editors. Vascular and neurological changes in early diabetes. New York: Academic Press;1973. p. 417–24.20. Lefevre PG, Davies RI. Active transport into the human erythrocyte; evidence from comparative kinetics and competition among monosaccharides. J Gen Physiol. 1951; 34:515–24.
Article21. Kotb MA, Bedewi MA, Aldossary NM, Mahmoud G, Naguib MF. Sonographic assessment of carpal tunnel syndrome in diabetic patients with and without polyneuropathy. Medicine (Baltimore). 2018; 97:e11104.
Article22. Steinkohl F, Loizides A, Gruber L, Karpf M, Morsdorf G, Gruber I, et al. Ultrasonography for the diagnosis of carpal tunnel syndrome in diabetic patients: missing the mark? Rofo. 2018.
Article23. Hassan A, Leep Hunderfund AN, Watson J, Boon AJ, Sorenson EJ. Median nerve ultrasound in diabetic peripheral neuropathy with and without carpal tunnel syndrome. Muscle Nerve. 2013; 47:437–9.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Electrodiagnosis of Carpal Tunnel Syndrome in Patients with Diabetic Neuropathy
- Prevalence and Risk Factors of Carpal Tunnel Syndrome in Diabetic Patients
- Ultrasonographic Findings of Median Nerve Change according to the Wrist Position
- The Correlation Between Electrodiagnostic Results and Ultrasonographic Findings in the Severity of Carpal Tunnel Syndrome in Females
- Outcomes of Carpal Tunnel Release in Diabetic and Non-Diabetic Patients