Korean Circ J.
2019 Mar;49(3):252-263. 10.4070/kcj.2018.0220.
Cost-Effectiveness of Rivaroxaban Compared to Warfarin for Stroke Prevention in Atrial Fibrillation
- Affiliations
-
- 1Department of Preventive Medicine, Institute of Human Complexity and Systems Science, Yonsei University College of Medicine, Seoul, Korea. preman@yuhs.ac
- 2Division of Cardiology, Department of Internal Medicine, Severance Cardiovascular Hospital, Yonsei University College of Medicine, Seoul, Korea. cby6908@yuhs.ac
- 3Division of Cardiology, Department of Internal Medicine, Kyung Hee University Hospital, Kyung Hee University, Seoul, Korea.
- KMID: 2438536
- DOI: http://doi.org/10.4070/kcj.2018.0220
Abstract
- BACKGROUND AND OBJECTIVES
Rivaroxaban is noninferior to warfarin for preventing stroke or systemic embolism in patients with high-risk atrial fibrillation (AF) and is associated with a lower rate of intracranial hemorrhage (ICH). We assessed the cost-effectiveness of rivaroxaban compared to adjusted-dose warfarin for the prevention of stroke in patients with nonvalvular AF.
METHODS
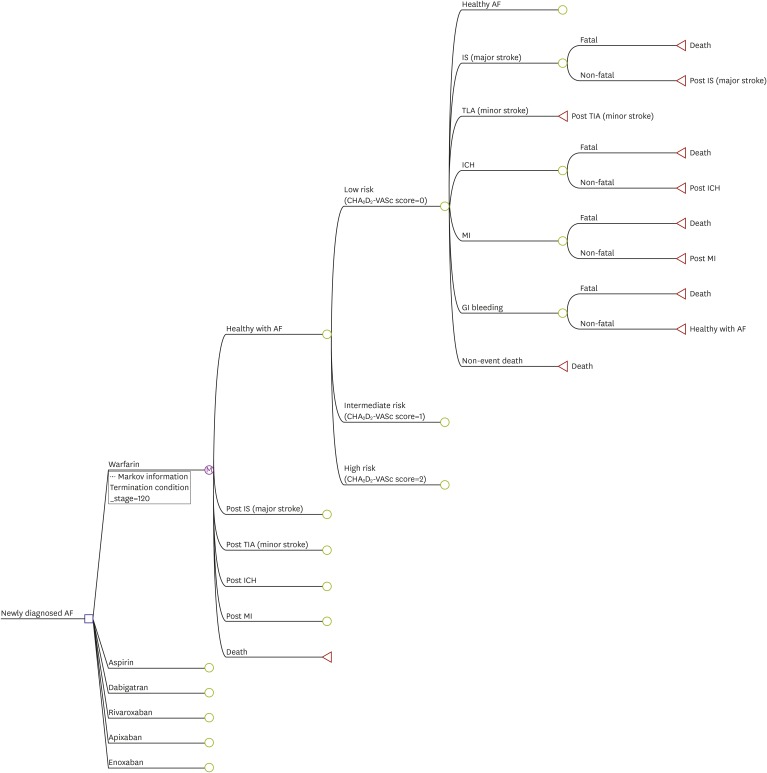
We built a Markov model using the Korean Health Insurance Review & Assessment Service database. The base-case analysis assumed a cohort of patients with prevalent AF who were aged 18 years or older without contraindications to anticoagulation.
RESULTS
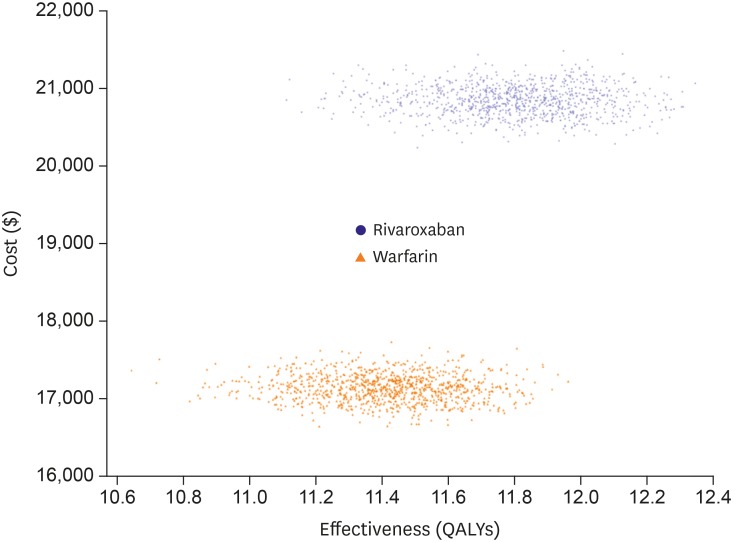
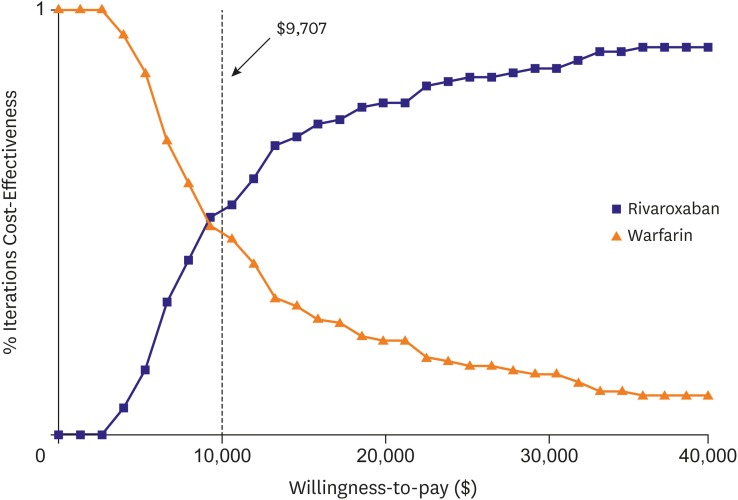
Number of patients with CHA2DS2-VASc scores 0, 1 and ≥2 were 56 (0.2%), 1,944 (6.3%) and 28,650 (93.5%), respectively. In patients with CHA2DS2-VASc scores ≥2, the incidence rate of ischemic stroke was 3.11% and 3.76% in warfarin and rivaroxaban groups, respectively. The incidence rates of ICH were 0.42% and 0.15%, and those of gastrointestinal bleeding were 0.32% and 0.15% in warfarin and rivaroxaban, respectively. Patients with AF treated with rivaroxaban lived an average of 11.8 quality-adjusted life years (QALYs) at a lifetime treatment cost of $20,886. Those receiving warfarin lived an average of 11.4 QALYs and incurred costs of $17,151. Patients with rivaroxaban gained an additional 0.4 QALYs over a lifetime with an additional cost of $3,735, resulting in an incremental cost-effectiveness ratio of $9,707 per QALY.
CONCLUSIONS
Patients who had been treated with rivaroxaban may be a cost-effective alternative to warfarin for stroke prevention in Korean patients with AF.
Keyword
MeSH Terms
Figure
Cited by 2 articles
-
Benefit, Risk and Cost in Oral Anticoagulation for Stroke Prevention: Is It the Third Factor?
Jong-Il Choi
Korean Circ J. 2019;49(3):264-266. doi: 10.4070/kcj.2019.0007.Cost-Effectiveness of Rate- and Rhythm-Control Drugs for Treating Atrial Fibrillation in Korea
Min Kim, Woojin Kim, Changsoo Kim, Boyoung Joung
Yonsei Med J. 2019;60(12):1157-1163. doi: 10.3349/ymj.2019.60.12.1157.
Reference
-
1. Lee H, Kim TH, Baek YS, et al. The trends of atrial fibrillation-related hospital visit and cost, treatment pattern and mortality in Korea: 10-year nationwide sample cohort data. Korean Circ J. 2017; 47:56–64. PMID: 28154592.2. Kim TH, Yang PS, Uhm JS, et al. CHA2DS2-VASc score (congestive heart failure, hypertension, age ≥75 [doubled], diabetes mellitus, prior stroke or transient ischemic attack [doubled], vascular disease, age 65–74, female) for stroke in Asian patients with atrial fibrillation: a Korean nationwide sample cohort study. Stroke. 2017; 48:1524–1530. PMID: 28455320.3. Hart RG, Pearce LA, Aguilar MI. Meta-analysis: antithrombotic therapy to prevent stroke in patients who have nonvalvular atrial fibrillation. Ann Intern Med. 2007; 146:857–867. PMID: 17577005.4. Decker C, Garavalia L, Garavalia B, et al. Exploring barriers to optimal anticoagulation for atrial fibrillation: interviews with clinicians. J Multidiscip Healthc. 2012; 5:129–135. PMID: 22936848.5. Connolly SJ, Ezekowitz MD, Yusuf S, et al. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med. 2009; 361:1139–1151. PMID: 19717844.6. Patel MR, Mahaffey KW, Garg J, et al. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med. 2011; 365:883–891. PMID: 21830957.7. Granger CB, Alexander JH, McMurray JJ, et al. Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med. 2011; 365:981–992. PMID: 21870978.8. Giugliano RP, Ruff CT, Braunwald E, et al. Edoxaban versus warfarin in patients with atrial fibrillation. N Engl J Med. 2013; 369:2093–2104. PMID: 24251359.9. Nelson WW, Song X, Coleman CI, et al. Medication persistence and discontinuation of rivaroxaban versus warfarin among patients with non-valvular atrial fibrillation. Curr Med Res Opin. 2014; 30:2461–2469. PMID: 24926732.10. Camm AJ, Lip GY, De Caterina R, et al. 2012 focused update of the ESC Guidelines for the management of atrial fibrillation: an update of the 2010 ESC Guidelines for the management of atrial fibrillation. Developed with the special contribution of the European Heart Rhythm Association. Eur Heart J. 2012; 33:2719–2747. PMID: 22922413.11. January CT, Wann LS, Alpert JS, et al. 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2014; 64:e1–76. PMID: 24685669.12. Kim D, Yang PS, Jang E, et al. 10-year nationwide trends of the incidence, prevalence, and adverse outcomes of non-valvular atrial fibrillation nationwide health insurance data covering the entire Korean population. Am Heart J. 2018; 202:20–26. PMID: 29802976.13. Kim D, Yang PS, Jang E, et al. Increasing trends in hospital care burden of atrial fibrillation in Korea, 2006 through 2015. Heart. 2018; heartjnl-2017-312930.14. Baek YS, Yang PS, Kim TH, et al. Associations of abdominal obesity and new-onset atrial fibrillation in the general population. J Am Heart Assoc. 2017; 6:e004705. PMID: 28588091.15. Lee HY, Yang PS, Kim TH, et al. Atrial fibrillation and the risk of myocardial infarction: a nation-wide propensity-matched study. Sci Rep. 2017; 7:12716. PMID: 28983076.16. Ades AE, Cliffe S. Markov chain Monte Carlo estimation of a multiparameter decision model: consistency of evidence and the accurate assessment of uncertainty. Med Decis Making. 2002; 22:359–371. PMID: 12150601.17. Kirchhof P, Benussi S, Kotecha D, et al. 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Eur Heart J. 2016; 37:2893–2962. PMID: 27567408.18. Korean Statistical Information Service (KOSIS). Korean life expectation. 2016.19. Kim TH, Yang PS, Yu HT, et al. Age threshold for ischemic stroke risk in atrial fibrillation: cohort data covering the entire Korean population. Stroke. 2018; 39:ehy565.P2904.20. O'Brien CL, Gage BF. Costs and effectiveness of ximelagatran for stroke prophylaxis in chronic atrial fibrillation. JAMA. 2005; 293:699–706. PMID: 15701911.21. Hylek EM, Singer DE. Risk factors for intracranial hemorrhage in outpatients taking warfarin. Ann Intern Med. 1994; 120:897–902. PMID: 8172435.22. Neumann PJ, Sandberg EA, Bell CM, Stone PW, Chapman RH. Are pharmaceuticals cost-effective? A review of the evidence. Health Aff (Millwood). 2000; 19:92–109. PMID: 10718025.23. Sullivan PW, Ghushchyan V. Preference-Based EQ-5D index scores for chronic conditions in the United States. Med Decis Making. 2006; 26:410–420. PMID: 16855129.24. Lanitis T, Kongnakorn T, Jacobson L, De Geer A. Cost-effectiveness of apixaban versus warfarin and aspirin in Sweden for stroke prevention in patients with atrial fibrillation. Thromb Res. 2014; 134:278–287. PMID: 24935675.25. Magnuson EA, Vilain K, Wang K, et al. Cost-effectiveness of edoxaban vs warfarin in patients with atrial fibrillation based on results of the ENGAGE AF-TIMI 48 trial. Am Heart J. 2015; 170:1140–1150. PMID: 26678636.26. Freeman JV, Zhu RP, Owens DK, et al. Cost-effectiveness of dabigatran compared with warfarin for stroke prevention in atrial fibrillation. Ann Intern Med. 2011; 154:1–11. PMID: 21041570.27. Sorensen SV, Kansal AR, Connolly S, et al. Cost-effectiveness of dabigatran etexilate for the prevention of stroke and systemic embolism in atrial fibrillation: a Canadian payer perspective. Thromb Haemost. 2011; 105:908–919. PMID: 21431243.28. Pink J, Lane S, Pirmohamed M, Hughes DA. Dabigatran etexilate versus warfarin in management of non-valvular atrial fibrillation in UK context: quantitative benefit-harm and economic analyses. BMJ. 2011; 343:d6333. PMID: 22042753.29. Lee S, Anglade MW, Pham D, Pisacane R, Kluger J, Coleman CI. Cost-effectiveness of rivaroxaban compared to warfarin for stroke prevention in atrial fibrillation. Am J Cardiol. 2012; 110:845–851. PMID: 22651881.30. López-López JA, Sterne JA, Thom HH, et al. Oral anticoagulants for prevention of stroke in atrial fibrillation: systematic review, network meta-analysis, and cost effectiveness analysis. BMJ. 2017; 359:j5058. PMID: 29183961.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Application of New Oral Anticoagulants: Prevention of Stroke in Patients with Nonvalvular Atrial Fibrillation
- New oral anticoagulants
- Use of NOAC in Cardioversion
- Real World Comparison of Rivaroxaban and Warfarin in Korean Patients with Atrial Fibrillation: Propensity Matching Cohort Analysis
- Practical Issues to Prevent Stroke Associated with Non-valvular Atrial Fibrillation