Korean J Radiol.
2017 Oct;18(5):799-808. 10.3348/kjr.2017.18.5.799.
Percutaneous Dual-Switching Monopolar Radiofrequency Ablation Using a Separable Clustered Electrode: A Preliminary Study
- Affiliations
-
- 1Department of Radiology, Seoul National University Hospital, Seoul 03080, Korea. jmsh@snu.ac.kr
- 2Institute of Radiation Medicine, Seoul National University Hospital, Seoul 03080, Korea.
- 3Department of Internal Medicine, Seoul National University Hospital, Seoul 03080, Korea.
- KMID: 2427216
- DOI: http://doi.org/10.3348/kjr.2017.18.5.799
Abstract
OBJECTIVE
To prospectively evaluate the safety and therapeutic effectiveness of dual-switching monopolar (DSM) radiofrequency ablation (RFA) for the treatment of hepatocellular carcinoma (HCC), and to retrospectively compare the results with those of single-switching monopolar (SSM) RFA in a historical control group.
MATERIALS AND METHODS
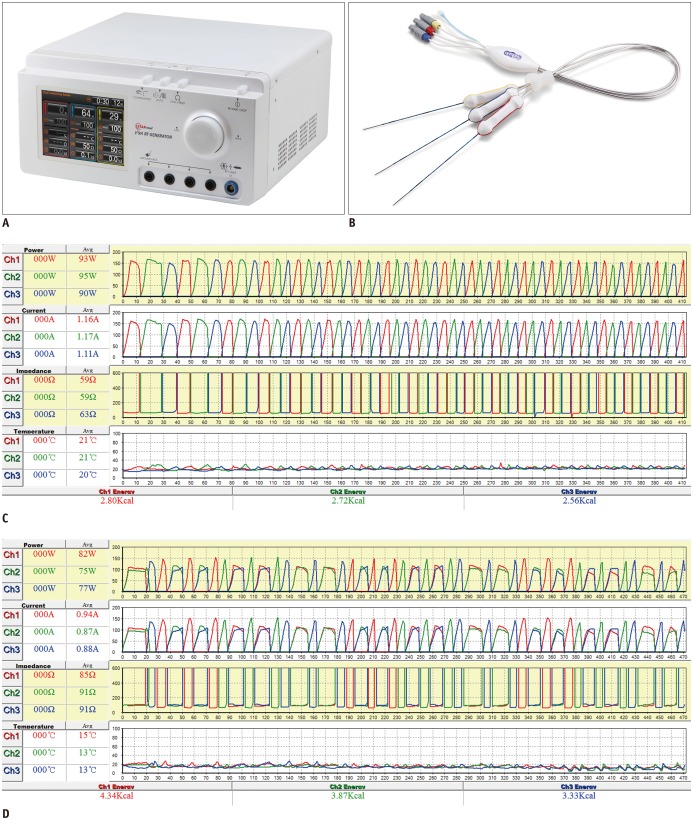
This study was approved by the Institutional Review Board, with informed consent obtained from all patients. Fifty-two HCC patients who underwent DSM-RFA using a separable clustered electrode and dual-generators were prospectively enrolled. Technical parameters, complications, technical success, technical effectiveness, and local tumor progression (LTP) rates were evaluated by means of post-procedural and follow-up imaging. Thereafter, the outcome of DSM-RFA was compared with those of 249 retrospectively included HCC patients treated with SSM-RFA.
RESULTS
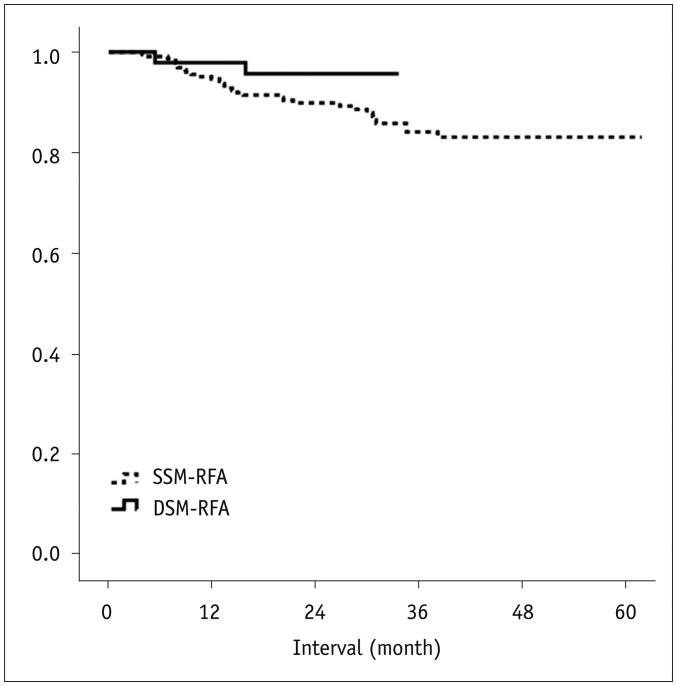
There were two major complications (3.8%, 2/52) including pleural and pericardial effusion in the DSM-RFA group. The DSM-RFA yielded a 100% technical success rate, a 98.1% technical effectiveness rate, and a 4.3% 2-year LTP rate. In a retrospective comparison between the two groups, DSM-RFA created significantly larger ablation volume (4.20 ± 2.07 cm³/min vs. 3.03 ± 1.99 cm³/min, p < 0.01), and delivered higher energy (1.43 ± 0.37 kcal/min vs. 1.25 ± 0.50 kcal/min, p < 0.01) per given time, than SSM-RFA. There was no significant difference in major procedure-related complications (3.8% vs. 4.4%) and technical effectiveness rate (98.1% vs. 96.4%) between the two groups (p = 1.00). In addition, the 2-year LTP rate of DSM-RFA and SSM-RFA were 4.3% and 10.1%, respectively (p = 0.15).
CONCLUSION
DSM-RFA using a separable clustered electrode is safe and provides high local tumor control and good preliminary clinical outcome for small HCCs, which are at least comparable to those of SSM-RFA.
Keyword
MeSH Terms
-
Aged
Carcinoma, Hepatocellular/mortality/*surgery
Catheter Ablation/adverse effects/instrumentation/*methods
Electrodes
Female
Humans
Liver Neoplasms/mortality/*surgery
Male
Middle Aged
Pericardial Effusion/etiology
Pleural Effusion/etiology
Retrospective Studies
Serum Albumin/analysis
Survival Rate
Serum Albumin
Figure
Cited by 1 articles
-
Recent Advances in the Image-Guided Tumor Ablation of Liver Malignancies: Radiofrequency Ablation with Multiple Electrodes, Real-Time Multimodality Fusion Imaging, and New Energy Sources
Dong Ho Lee, Jeong Min Lee
Korean J Radiol. 2018;19(4):545-559. doi: 10.3348/kjr.2018.19.4.545.
Reference
-
1. Kim JW, Shin SS, Heo SH, Hong JH, Lim HS, Seon HJ, et al. Ultrasound-guided percutaneous radiofrequency ablation of liver tumors: how we do it safely and completely. Korean J Radiol. 2015; 16:1226–1239. PMID: 26576111.
Article2. Pompili M, Saviano A, de Matthaeis N, Cucchetti A, Ardito F, Federico B, et al. Long-term effectiveness of resection and radiofrequency ablation for single hepatocellular carcinoma ≤3 cm. Results of a multicenter Italian survey. J Hepatol. 2013; 59:89–97. PMID: 23523578.3. Bruix J, Sherman M. American Association for the Study of Liver Diseases. Management of hepatocellular carcinoma: an update. Hepatology. 2011; 53:1020–1022. PMID: 21374666.
Article4. Yu SJ. A concise review of updated guidelines regarding the management of hepatocellular carcinoma around the world: 2010-2016. Clin Mol Hepatol. 2016; 22:7–17. PMID: 27044761.
Article5. Ruzzenente A, Guglielmi A, Sandri M, Campagnaro T, Valdegamberi A, Conci S, et al. Surgical resection versus local ablation for HCC on cirrhosis: results from a propensity case-matched study. J Gastrointest Surg. 2012; 16:301–311. discussion 311. PMID: 22095524.
Article6. Montorsi M, Santambrogio R, Bianchi P, Donadon M, Moroni E, Spinelli A, et al. Survival and recurrences after hepatic resection or radiofrequency for hepatocellular carcinoma in cirrhotic patients: a multivariate analysis. J Gastrointest Surg. 2005; 9:62–67. discussion 67-68. PMID: 15623446.
Article7. Nakazawa T, Kokubu S, Shibuya A, Ono K, Watanabe M, Hidaka H, et al. Radiofrequency ablation of hepatocellular carcinoma: correlation between local tumor progression after ablation and ablative margin. AJR Am J Roentgenol. 2007; 188:480–488. PMID: 17242258.
Article8. Kim YS, Lee WJ, Rhim H, Lim HK, Choi D, Lee JY. The minimal ablative margin of radiofrequency ablation of hepatocellular carcinoma (> 2 and < 5 cm) needed to prevent local tumor progression: 3D quantitative assessment using CT image fusion. AJR Am J Roentgenol. 2010; 195:758–765. PMID: 20729457.9. Kim KW, Lee JM, Klotz E, Kim SJ, Kim SH, Kim JY, et al. Safety margin assessment after radiofrequency ablation of the liver using registration of preprocedure and postprocedure CT images. AJR Am J Roentgenol. 2011; 196:W565–W572. PMID: 21512046.
Article10. Pereira PL. Actual role of radiofrequency ablation of liver metastases. Eur Radiol. 2007; 17:2062–2070. PMID: 17429644.
Article11. Ni Y, Mulier S, Miao Y, Michel L, Marchal G. A review of the general aspects of radiofrequency ablation. Abdom Imaging. 2005; 30:381–400. PMID: 15776302.
Article12. Kim YS, Lim HK, Rhim H, Lee MW, Choi D, Lee WJ, et al. Ten-year outcomes of percutaneous radiofrequency ablation as first-line therapy of early hepatocellular carcinoma: analysis of prognostic factors. J Hepatol. 2013; 58:89–97. PMID: 23023009.
Article13. Lee J, Lee JM, Yoon JH, Lee JY, Kim SH, Lee JE, et al. Percutaneous radiofrequency ablation with multiple electrodes for medium-sized hepatocellular carcinomas. Korean J Radiol. 2012; 13:34–43. PMID: 22247634.
Article14. Lee JM, Han JK, Kim HC, Choi YH, Kim SH, Choi JY, et al. Switching monopolar radiofrequency ablation technique using multiple, internally cooled electrodes and a multichannel generator: ex vivo and in vivo pilot study. Invest Radiol. 2007; 42:163–171. PMID: 17287646.15. Woo S, Lee JM, Yoon JH, Joo I, Kim SH, Lee JY, et al. Small- and medium-sized hepatocellular carcinomas: monopolar radiofrequency ablation with a multiple-electrode switching system-mid-term results. Radiology. 2013; 268:589–600. PMID: 23513241.
Article16. Lee ES, Lee JM, Kim WS, Choi SH, Joo I, Kim M, et al. Multiple-electrode radiofrequency ablations using Octopus® electrodes in an in vivo porcine liver model. Br J Radiol. 2012; 85:e609–e615. PMID: 22422385.17. Park MJ, Kim YS, Rhim H, Lim HK, Lee MW, Choi D. A comparison of US-guided percutaneous radiofrequency ablation of medium-sized hepatocellular carcinoma with a cluster electrode or a single electrode with a multiple overlapping ablation technique. J Vasc Interv Radiol. 2011; 22:771–779. PMID: 21616430.
Article18. Weisbrod AJ, Atwell TD, Callstrom MR, Farrell MA, Mandrekar JN, Charboneau JW. Percutaneous radiofrequency ablation with a multiple-electrode switching-generator system. J Vasc Interv Radiol. 2007; 18:1528–1532. PMID: 18057287.
Article19. Yoon JH, Lee JM, Hwang EJ, Hwang IP, Baek J, Han JK, et al. Monopolar radiofrequency ablation using a dual-switching system and a separable clustered electrode: evaluation of the in vivo efficiency. Korean J Radiol. 2014; 15:235–244. PMID: 24643383.20. Yoon JH, Lee JM, Han JK, Choi BI. Dual switching monopolar radiofrequency ablation using a separable clustered electrode: comparison with consecutive and switching monopolar modes in ex vivo bovine livers. Korean J Radiol. 2013; 14:403–411. PMID: 23690705.21. Mitchell DG, Bruix J, Sherman M, Sirlin CB. LI-RADS (Liver Imaging Reporting and Data System): summary, discussion, and consensus of the LI-RADS Management Working Group and future directions. Hepatology. 2015; 61:1056–1065. PMID: 25041904.
Article22. Hope TA, Fowler KJ, Sirlin CB, Costa EA, Yee J, Yeh BM, et al. Hepatobiliary agents and their role in LI-RADS. Abdom Imaging. 2015; 40:613–625. PMID: 25287679.
Article23. Lee MW. Fusion imaging of real-time ultrasonography with CT or MRI for hepatic intervention. Ultrasonography. 2014; 33:227–239. PMID: 25036756.
Article24. Min JH, Lim HK, Lim S, Kang TW, Song KD, Choi SY, et al. Radiofrequency ablation of very-early-stage hepatocellular carcinoma inconspicuous on fusion imaging with B-mode US: value of fusion imaging with contrast-enhanced US. Clin Mol Hepatol. 2014; 20:61–70. PMID: 24757660.
Article25. Rhim H, Lim HK, Kim YS, Choi D. Percutaneous radiofrequency ablation with artificial ascites for hepatocellular carcinoma in the hepatic dome: initial experience. AJR Am J Roentgenol. 2008; 190:91–98. PMID: 18094298.
Article26. Kudo M, Matsui O, Izumi N, Iijima H, Kadoya M, Imai Y, et al. JSH consensus-based clinical practice guidelines for the management of hepatocellular carcinoma: 2014 update by the Liver Cancer Study Group of Japan. Liver Cancer. 2014; 3:458–468. PMID: 26280007.
Article27. Ahmed M, Solbiati L, Brace CL, Breen DJ, Callstrom MR, Charboneau JW, et al. Image-guided tumor ablation: standardization of terminology and reporting criteria--a 10-year update. Radiology. 2014; 273:241–260. PMID: 24927329.
Article28. Sacks D, McClenny TE, Cardella JF, Lewis CA. Society of Interventional Radiology clinical practice guidelines. J Vasc Interv Radiol. 2003; 14(9 Pt 2):S199–S202. PMID: 14514818.
Article29. Iezzi R, Pompili M, Posa A, Coppola G, Gasbarrini A, Bonomo L. Combined locoregional treatment of patients with hepatocellular carcinoma: state of the art. World J Gastroenterol. 2016; 22:1935–1942. PMID: 26877601.
Article30. Hyun D, Cho SK, Shin SW, Park KB, Park HS, Choo SW, et al. Early stage hepatocellular carcinomas not feasible for ultrasound-guided radiofrequency ablation: comparison of transarterial chemoembolization alone and combined therapy with transarterial chemoembolization and radiofrequency ablation. Cardiovasc Intervent Radiol. 2016; 39:417–425. PMID: 26246215.
Article31. Ahmed M, Solbiati L, Brace CL, Breen DJ, Callstrom MR, Charboneau JW, et al. Image-guided tumor ablation: standardization of terminology and reporting criteria--a 10-year update. J Vasc Interv Radiol. 2014; 25:1691–1705.e4. PMID: 25442132.
Article32. Wang X, Hu Y, Ren M, Lu X, Lu G, He S. Efficacy and safety of radiofrequency ablation combined with transcatheter arterial chemoembolization for hepatocellular carcinomas compared with radiofrequency ablation alone: a time-to-event meta-analysis. Korean J Radiol. 2016; 17:93–102. PMID: 26798221.
Article33. Jun HY, Ryu JH, Byun SJ, Jeong CW, Kim TH, Lee YH, et al. Combined radiofrequency ablation and double anti-angiogenic protein therapy to increase coagulation efficacy: an experimental study in a murine renal carcinoma model. Korean J Radiol. 2015; 16:776–782. PMID: 26175576.
Article34. Livraghi T, Solbiati L, Meloni MF, Gazelle GS, Halpern EF, Goldberg SN. Treatment of focal liver tumors with percutaneous radio-frequency ablation: complications encountered in a multicenter study. Radiology. 2003; 226:441–451. PMID: 12563138.
Article35. Rhim H. Complications of radiofrequency ablation in hepatocellular carcinoma. Abdom Imaging. 2005; 30:409–418. PMID: 15688113.
Article36. Rhim H, Yoon KH, Lee JM, Cho Y, Cho JS, Kim SH, et al. Major complications after radio-frequency thermal ablation of hepatic tumors: spectrum of imaging findings. Radiographics. 2003; 23:123–134. discussion 134-136. PMID: 12533647.
Article37. Seror O, N'Kontchou G, Van Nhieu JT, Rabahi Y, Nahon P, Laurent A, et al. Histopathologic comparison of monopolar versus no-touch multipolar radiofrequency ablation to treat hepatocellular carcinoma within Milan criteria. J Vasc Interv Radiol. 2014; 25:599–607. PMID: 24529547.
Article38. Seror O, N'Kontchou G, Ibraheem M, Ajavon Y, Barrucand C, Ganne N, et al. Large (>or=5.0-cm) HCCs: multipolar RF ablation with three internally cooled bipolar electrodes--initial experience in 26 patients. Radiology. 2008; 248:288–296. PMID: 18483229.39. Bruners P, Schmitz-Rode T, Günther RW, Mahnken A. Multipolar hepatic radiofrequency ablation using up to six applicators: preliminary results. Rofo. 2008; 180:216–222. PMID: 18278730.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Monopolar Radiofrequency Ablation Using a Dual-Switching System and a Separable Clustered Electrode: Evaluation of the In Vivo Efficiency
- Dual Switching Monopolar Radiofrequency Ablation Using a Separable Clustered Electrode: Comparison with Consecutive and Switching Monopolar Modes in Ex Vivo Bovine Livers
- Radiofrequency Ablation Using a Separable Clustered Electrode for the Treatment of Hepatocellular Carcinomas: A Randomized Controlled Trial of a DualSwitching Monopolar Mode Versus a Single-Switching Monopolar Mode
- Ablative Outcomes of Various Energy Modes for No-Touch and Peripheral Tumor-Puncturing Radiofrequency Ablation: An Ex Vivo Simulation Study
- Radio Frequency Ablation in the Rabbit Lung Using Wet Electrodes: Comparison of Monopolar and Dual Bipolar Electrode Mode