Korean J Radiol.
2018 Dec;19(6):1099-1109. 10.3348/kjr.2018.19.6.1099.
No-Touch Radiofrequency Ablation of VX2 Hepatic Tumors In Vivo in Rabbits: A Proof of Concept Study
- Affiliations
-
- 1Department of Radiology, Seoul National University Hospital, Seoul 03080, Korea. jmlshy2000@gmail.com
- 2Department of Radiology, Seoul National University College of Medicine, Seoul 03080, Korea.
- 3Institute of Radiation Medicine, Seoul National University Medical Research Center, Seoul 03080, Korea.
- KMID: 2424849
- DOI: http://doi.org/10.3348/kjr.2018.19.6.1099
Abstract
OBJECTIVE
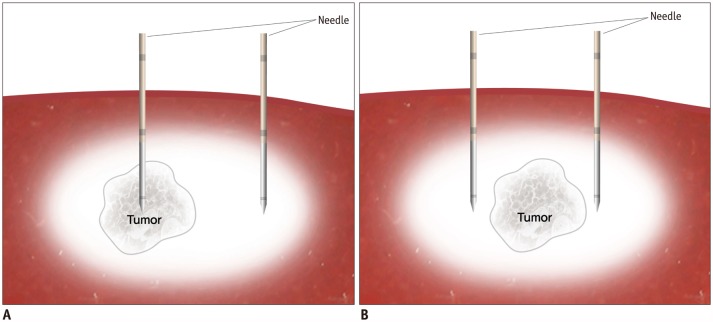
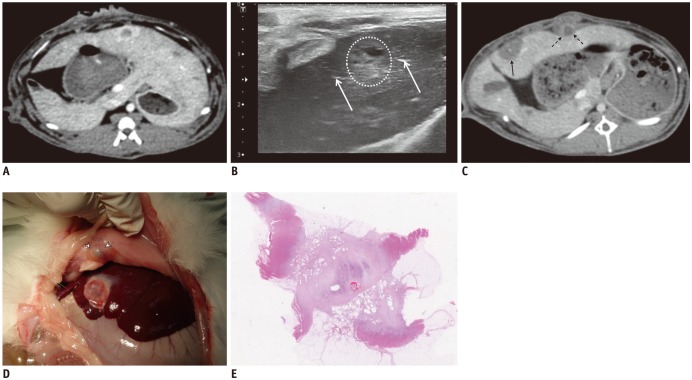
In a proof of concept study, we compared no-touch radiofrequency ablation (NtRFA) in bipolar mode with conventional direct tumor puncture (DTP) in terms of local tumor control (LTC), peritoneal seeding, and tumorigenic factors, in the rabbit VX2 subcapsular hepatic tumor model.
MATERIALS AND METHODS
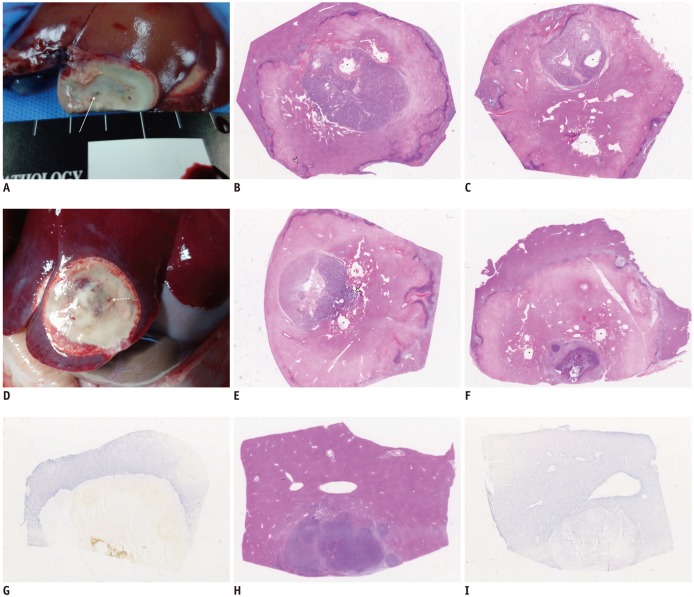
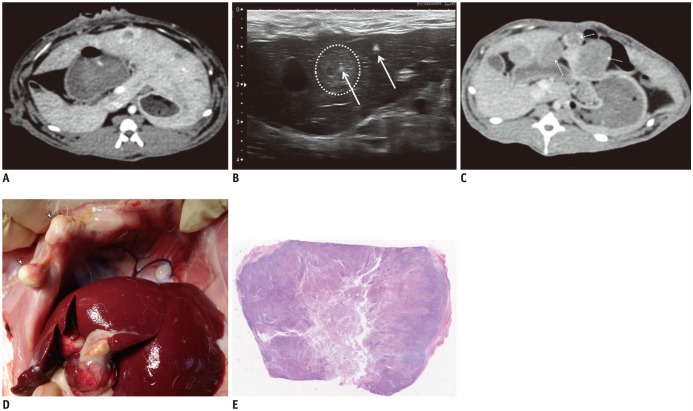
Sixty-two rabbits with VX2 subcapsular hepatic tumors were divided into three groups according to the procedure: DTP-RFA (n = 25); NtRFA (n = 25); and control (n = 12). Each of the three groups was subdivided into two sets for pathologic analysis (n = 24) or computed tomography (CT) follow-up for 6 weeks after RFA (n = 38). Ultrasonography-guided DTP-RFA and NtRFA were performed nine days after tumor implantation. LTC was defined by either achievement of complete tumor necrosis on histopathology or absence of local tumor progression on follow-up CT and autopsy. Development of peritoneal seeding was also compared among the groups. Serum hepatocyte growth factor (HGF), vascular endothelial growth factor (VEGF) and interleukin-6 (IL-6) were measured via ELISA (Elabscience Biotechnology Co.) after RFA for tumorigenic factor evaluation.
RESULTS
Regarding LTC, there was a trend in NtRFA (80%, 20/25) toward better ablation than in DTP-RFA (56%, 14/25) (p = 0.069). Complete tumor necrosis was achieved in 54.5% of DTP-RFA (6/11) and 90.9% of NtRFA (10/11). Peritoneal seeding was significantly more common in DTP-RFA (71.4%, 10/14) than in NtRFA (21.4%, 3/14) (p = 0.021) or control (0%). Elevations of HGF, VEGF or IL-6 were not detected in any group.
CONCLUSION
No-touch radiofrequency ablation led to lower rates of peritoneal seeding and showed a tendency toward better LTC than DTP-RFA.
Keyword
MeSH Terms
Figure
Reference
-
1. Bruix J, Sherman M. American Association for the Study of Liver Diseases. Management of hepatocellular carcinoma: an update. Hepatology. 2011; 53:1020–1022. PMID: 21374666.
Article2. Hocquelet A, Aubé C, Rode A, Cartier V, Sutter O, Manichon AF, et al. Comparison of no-touch multi-bipolar vs. monopolar radiofrequency ablation for small HCC. J Hepatol. 2017; 66:67–74. PMID: 27422750.
Article3. Lu DS, Yu NC, Raman SS, Limanond P, Lassman C, Murray K, et al. Radiofrequency ablation of hepatocellular carcinoma: treatment success as defined by histologic examination of the explanted liver. Radiology. 2005; 234:954–960. PMID: 15681691.
Article4. Brillet PY, Paradis V, Brancatelli G, Rangheard AS, Consigny Y, Plessier A, et al. Percutaneous radiofrequency ablation for hepatocellular carcinoma before liver transplantation: a prospective study with histopathologic comparison. AJR Am J Roentgenol. 2006; 186(5 Suppl):S296–S305. PMID: 16632691.
Article5. Seror O, N'Kontchou G, Nault JC, Rabahi Y, Nahon P, Ganne-Carrié N, et al. Hepatocellular carcinoma within Milan criteria: no-touch multibipolar radiofrequency ablation for treatment-long-term results. Radiology. 2016; 280:981.
Article6. Dodd GD 3rd, Frank MS, Aribandi M, Chopra S, Chintapalli KN. Radiofrequency thermal ablation: computer analysis of the size of the thermal injury created by overlapping ablations. AJR Am J Roentgenol. 2001; 177:777–782. PMID: 11566672.7. Lee J, Lee JM, Yoon JH, Lee JY, Kim SH, Lee JE, et al. Percutaneous radiofrequency ablation with multiple electrodes for medium-sized hepatocellular carcinomas. Korean J Radiol. 2012; 13:34–43. PMID: 22247634.
Article8. Seror O, N'Kontchou G, Van Nhieu JT, Rabahi Y, Nahon P, Laurent A, et al. Histopathologic comparison of monopolar versus no-touch multipolar radiofrequency ablation to treat hepatocellular carcinoma within Milan criteria. J Vasc Interv Radiol. 2014; 25:599–607. PMID: 24529547.
Article9. Patel PA, Ingram L, Wilson ID, Breen DJ. No-touch wedge ablation technique of microwave ablation for the treatment of subcapsular tumors in the liver. J Vasc Interv Radiol. 2013; 24:1257–1262. PMID: 23885917.
Article10. Lee MW, Raman SS, Asvadi NH, Siripongsakun S, Hicks RM, Chen J, et al. Radiofrequency ablation of hepatocellular carcinoma as bridge therapy to liver transplantation: a 10-year intention-to-treat analysis. Hepatology. 2017; 65:1979–1990. PMID: 28170115.
Article11. Pompili M, Francica G, Ponziani FR, Iezzi R, Avolio AW. Bridging and downstaging treatments for hepatocellular carcinoma in patients on the waiting list for liver transplantation. World J Gastroenterol. 2013; 19:7515–7530. PMID: 24282343.
Article12. Kumar N, Gaba RC, Knuttinen MG, Omene BO, Martinez BK, Owens CA, et al. Tract seeding following radiofrequency ablation for hepatocellular carcinoma: prevention, detection, and management. Semin Intervent Radiol. 2011; 28:187–192. PMID: 22654260.
Article13. Wang S, Mei XG, Goldberg SN, Ahmed M, Lee JC, Gong W, et al. Does thermosensitive liposomal vinorelbine improve end-point survival after percutaneous radiofrequency ablation of liver tumors in a mouse model? Radiology. 2016; 279:762–772. PMID: 26785043.
Article14. Bulvik BE, Rozenblum N, Gourevich S, Ahmed M, Andriyanov AV, Galun E, et al. Irreversible electroporation versus radiofrequency ablation: a comparison of local and systemic effects in a small-animal model. Radiology. 2016; 280:413–424. PMID: 27429143.
Article15. Ahmed M, Kumar G, Moussa M, Wang Y, Rozenblum N, Galun E, et al. Hepatic radiofrequency ablation-induced stimulation of distant tumor growth is suppressed by c-Met inhibition. Radiology. 2016; 279:103–117. PMID: 26418615.
Article16. Rozenblum N, Zeira E, Scaiewicz V, Bulvik B, Gourevitch S, Yotvat H, et al. Oncogenesis: an “off-target” effect of radiofrequency ablation. Radiology. 2015; 3. 30. [Epub ahead of print]. DOI: 10.1148/radiol.15141695.
Article17. Rozenblum N, Zeira E, Bulvik B, Gourevitch S, Yotvat H, Galun E, et al. Radiofrequency ablation: inflammatory changes in the periablative zone can induce global organ effects, including liver regeneration. Radiology. 2015; 276:416–425. PMID: 25822472.
Article18. Maruyama H, Matsutani S, Saisho H, Kamiyama N, Mine Y, Hirata T, et al. Sonographic shift of hypervascular liver tumor on blood pool harmonic images with definity: time-related changes of contrast-enhanced appearance in rabbit VX2 tumor under extra-low acoustic power. Eur J Radiol. 2005; 56:60–65. PMID: 16168266.
Article19. Galasko CS, Muckle DS. Intrasarcolemmal proliferation of the VX2 carcinoma. Br J Cancer. 1974; 29:59–65. PMID: 4820946.
Article20. Bimonte S, Leongito M, Piccirillo M, de Angelis C, Pivonello C, Granata V, et al. Radio-frequency ablation-based studies on VX2rabbit models for HCC treatment. Infect Agent Cancer. 2016; 11:38. PMID: 27525037.
Article21. Behm B, Di Fazio P, Michl P, Neureiter D, Kemmerling R, Hahn EG, et al. Additive antitumour response to the rabbit VX2 hepatoma by combined radio frequency ablation and toll like receptor 9 stimulation. Gut. 2016; 65:134–143. PMID: 25524262.
Article22. Jiang T, Zhang X, Ding J, Duan B, Lu S. Inflammation and cancer: inhibiting the progression of residual hepatic VX2 carcinoma by anti-inflammatory drug after incomplete radiofrequency ablation. Int J Clin Exp Pathol. 2015; 8:13945–13956. PMID: 26823706.23. Ke S, Ding XM, Kong J, Gao J, Wang SH, Cheng Y, et al. Low temperature of radiofrequency ablation at the target sites can facilitate rapid progression of residual hepatic VX2 carcinoma. J Transl Med. 2010; 8:73. PMID: 20667141.
Article24. Ahmed M, Solbiati L, Brace CL, Breen DJ, Callstrom MR, Charboneau JW, et al. International Working Group on Image-Guided Tumor Ablation. Interventional Oncology Sans Frontières Expert Panel. Technology Assessment Committee of the Society of Interventional Radiology. Standard of Practice Committee of the Cardiovascular and Interventional Radiological Society of Europe. Image-guided tumor ablation: standardization of terminology and reporting criteria--a 10-year update. J Vasc Interv Radiol. 2014; 25:1691–1705.e4. PMID: 25442132.
Article25. Imamura J, Tateishi R, Shiina S, Goto E, Sato T, Ohki T, et al. Neoplastic seeding after radiofrequency ablation for hepatocellular carcinoma. Am J Gastroenterol. 2008; 103:3057–3062. PMID: 19086957.
Article26. Wu H, Wilkins LR, Ziats NP, Haaga JR, Exner AA. Real-time monitoring of radiofrequency ablation and postablation assessment: accuracy of contrast-enhanced US in experimental rat liver model. Radiology. 2014; 270:107–116. PMID: 23912621.
Article27. Chang W, Lee JM, Lee SM, Han JK. No-touch radiofrequency ablation: a comparison of switching bipolar and switching monopolar ablation in ex vivo bovine liver. Korean J Radiol. 2017; 18:279–288. PMID: 28246508.28. Yoon JH, Lee JM, Hwang EJ, Hwang IP, Baek J, Han JK, et al. Monopolar radiofrequency ablation using a dual-switching system and a separable clustered electrode: evaluation of the in vivo efficiency. Korean J Radiol. 2014; 15:235–244. PMID: 24643383.29. Chang S, Kim SH, Lim HK, Kim SH, Lee WJ, Choi D, et al. Needle tract implantation after percutaneous interventional procedures in hepatocellular carcinomas: lessons learned from a 10-year experience. Korean J Radiol. 2008; 9:268–274. PMID: 18525230.
Article30. Livraghi T, Lazzaroni S, Meloni F, Solbiati L. Risk of tumour seeding after percutaneous radiofrequency ablation for hepatocellular carcinoma. Br J Surg. 2005; 92:856–858. PMID: 15892154.
Article31. Mulier S, Mulier P, Ni Y, Miao Y, Dupas B, Marchal G, et al. Complications of radiofrequency coagulation of liver tumours. Br J Surg. 2002; 89:1206–1222. PMID: 12296886.
Article32. Tateishi R, Shiina S, Teratani T, Obi S, Sato S, Koike Y, et al. Percutaneous radiofrequency ablation for hepatocellular carcinoma. An analysis of 1000 cases. Cancer. 2005; 103:1201–1209. PMID: 15690326.33. Jaskolka JD, Asch MR, Kachura JR, Ho CS, Ossip M, Wong F, et al. Needle tract seeding after radiofrequency ablation of hepatic tumors. J Vasc Interv Radiol. 2005; 16:485–491. PMID: 15802448.
Article34. Llovet JM, Vilana R, Brú C, Bianchi L, Salmeron JM, Boix L, et al. Barcelona Clínic Liver Cancer (BCLC) Group. Increased risk of tumor seeding after percutaneous radiofrequency ablation for single hepatocellular carcinoma. Hepatology. 2001; 33:1124–1129. PMID: 11343240.
Article35. Park SI, Kim IJ, Lee SJ, Shin MW, Shin WS, Chung YE, et al. Angled cool-tip electrode for radiofrequency ablation of small superficial subcapsular tumors in the liver: a feasibility study. Korean J Radiol. 2016; 17:742–749. PMID: 27587963.
Article36. Shirai K, Tamai H, Shingaki N, Mori Y, Moribata K, Enomoto S, et al. Clinical features and risk factors of extrahepatic seeding after percutaneous radiofrequency ablation for hepatocellular carcinoma. Hepatol Res. 2011; 41:738–745. PMID: 21699637.
Article37. Poon RT, Ng KK, Lam CM, Ai V, Yuen J, Fan ST. Radiofrequency ablation for subcapsular hepatocellular carcinoma. Ann Surg Oncol. 2004; 11:281–289. PMID: 14993023.
Article38. Wu LW, Chen CY, Liu CJ, Chen MY, Liu PC, Liu PF, et al. Multipolar radiofrequency ablation with non-touch technique for hepatocellular carcinoma ≤ 3 cm: a preliminary report. Advances in Digestive Medicine. 2014; 1:80–85.39. Li Y, Xu KS, Li JS, Jia WD, Liu WB, He XD, et al. The research of no-touch isolation technique on the prevention of postoperative recurrence and metastasis of hepatocellular carcinoma after hepatectomy. Hepatogastroenterology. 2014; 61:784–791. PMID: 26176074.40. Miyazono F, Takao S, Natsugoe S, Uchikura K, Kijima F, Aridome K, et al. Molecular detection of circulating cancer cells during surgery in patients with biliary-pancreatic cancer. Am J Surg. 1999; 177:475–479. PMID: 10414697.
Article41. Hayashi N, Egami H, Kai M, Kurusu Y, Takano S, Ogawa M. No-touch isolation technique reduces intraoperative shedding of tumor cells into the portal vein during resection of colorectal cancer. Surgery. 1999; 125:369–374. PMID: 10216526.
Article42. Liu CL, Fan ST, Cheung ST, Lo CM, Ng IO, Wong J. Anterior approach versus conventional approach right hepatic resection for large hepatocellular carcinoma: a prospective randomized controlled study. Ann Surg. 2006; 244:194–203. PMID: 16858181.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Radiofrequency Ablation of Unilateral Kidney VX2 Tumors in the Rabbit Model
- Percutaneous Radiofrequency Thermal Ablation of Lung VX2 Tumors in a Rabbit Model: Evaluation with Helical CT Findings for the Complete and Partal Ablation
- Radiofrequency Ablation of Hepatic Cysts: Case Report
- Effects of Arsenic Trioxide on Radiofrequency Ablation of VX2 Liver Tumor: Intraarterial versus Intravenous Administration
- Radiofrequency Thermal Ablation of Hepatocellular Carcinomas