J Pathol Transl Med.
2018 Sep;52(5):349-353. 10.4132/jptm.2018.07.16.
Abrupt Dyskeratotic and Squamoid Cells in Poorly Differentiated Carcinoma: Case Study of Two Thoracic NUT Midline Carcinomas with Cytohistologic Correlation
- Affiliations
-
- 1Department of Pathology, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea. hanjho@skku.edu
- 2Division of Pulmonary and Critical Care Medicine, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- KMID: 2422103
- DOI: http://doi.org/10.4132/jptm.2018.07.16
Abstract
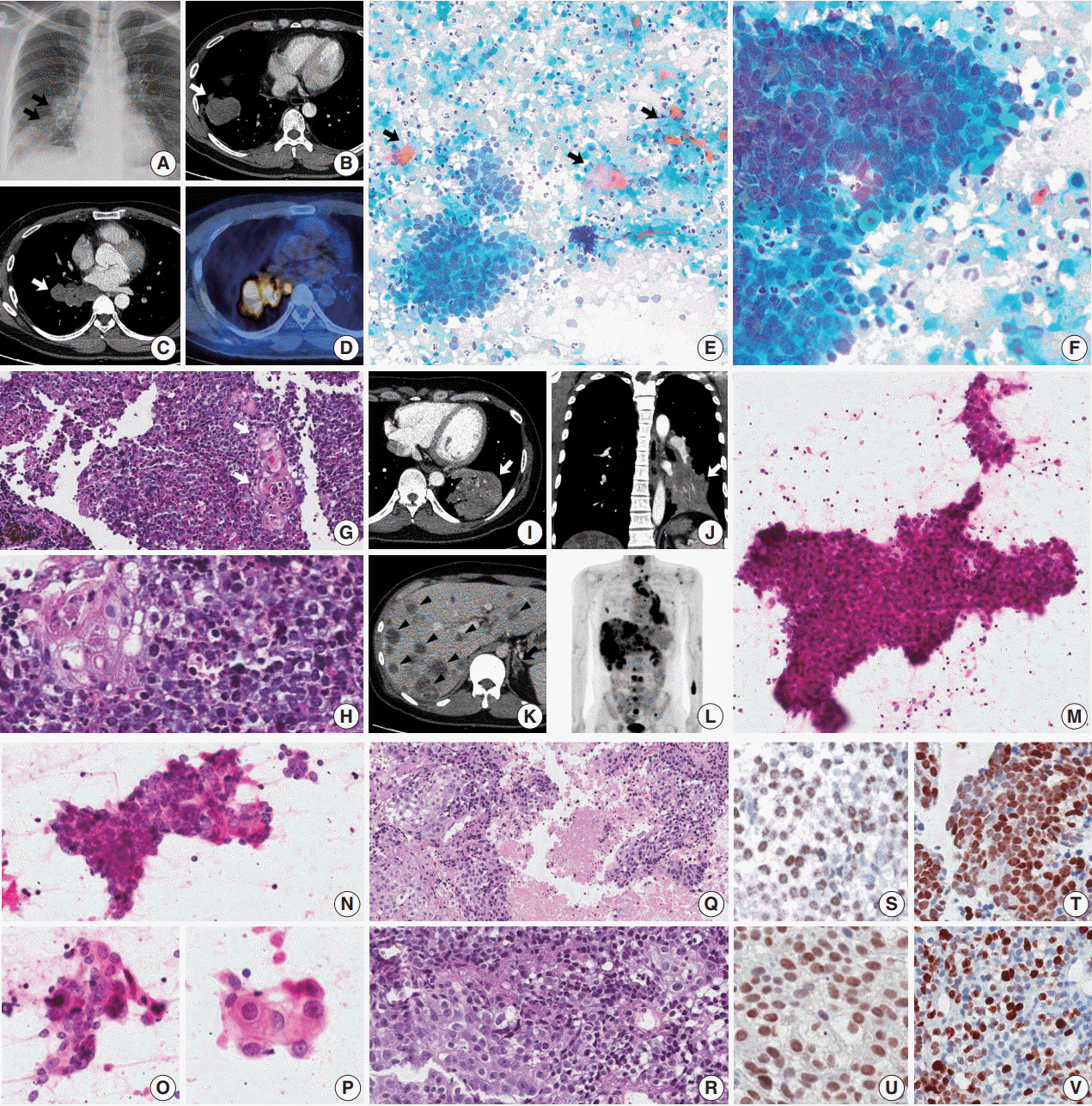
- Cytologic diagnosis of nuclear protein in testis (NUT) midline carcinoma (NMC) is important due to its aggressive behavior and miserable prognosis. Early diagnosis of NMC can facilitate proper management, and here we report two rare cases of thoracic NMC with cytohistologic correlation. In aspiration cytology, the tumor presented with mixed cohesive clusters and dispersed single cells, diffuse background necrosis and many neutrophils. Most of the tumor cells had scanty cytoplasm and medium-sized irregular nuclei, which had fine to granular nuclear chromatin. Interestingly, a few dyskeratotic cells or squamoid cell clusters were present in each case. Biopsy specimen histology revealed more frequent squamous differentiation, and additional immunohistochemistry tests showed nuclear expression of NUT. Because this tumor has a notorious progression and has been previously underestimated in terms of its prevalence, awareness of characteristic findings and proper ancillary tests should be considered in all suspicious cases.
MeSH Terms
Figure
Reference
-
1. French CA. Pathogenesis of NUT midline carcinoma. Annu Rev Pathol. 2012; 7:247–65.
Article2. Stelow EB. A review of NUT midline carcinoma. Head Neck Pathol. 2011; 5:31–5.
Article3. Park HS, Bae YS, Yoon SO, et al. Usefulness of nuclear protein in testis (NUT) immunohistochemistry in the cytodiagnosis of NUT midline carcinoma: a brief case report. Korean J Pathol. 2014; 48:335–8.
Article4. Policarpio-Nicolas ML, de Leon EM, Jagirdar J. Cytologic findings of NUT midline carcinoma in the hilum of the lung. Diagn Cytopathol. 2015; 43:739–42.
Article5. Bishop JA, French CA, Ali SZ. Cytopathologic features of NUT midline carcinoma: a series of 26 specimens from 13 patients. Cancer Cytopathol. 2016; 124:901–8.
Article6. French CA, Ramirez CL, Kolmakova J, et al. BRD-NUT oncoproteins: a family of closely related nuclear proteins that block epithelial differentiation and maintain the growth of carcinoma cells. Oncogene. 2008; 27:2237–42.
Article7. Schwartz BE, Hofer MD, Lemieux ME, et al. Differentiation of NUT midline carcinoma by epigenomic reprogramming. Cancer Res. 2011; 71:2686–96.
Article8. Sturgis CD, Nassar DL, D'Antonio JA, Raab SS. Cytologic features useful for distinguishing small cell from non-small cell carcinoma in bronchial brush and wash specimens. Am J Clin Pathol. 2000; 114:197–202.
Article9. Marks RA, Cramer HM, Wu HH. Fine-needle aspiration cytology of basaloid squamous cell carcinoma and small cell carcinoma-a comparison study. Diagn Cytopathol. 2013; 41:81–4.
Article10. Gray W, Kocjan G. Diagnostic cytopathology. Philadelphia: Elsevier;2010. p. 68–9. 3rd.11. Evans AG, French CA, Cameron MJ, et al. Pathologic characteristics of NUT midline carcinoma arising in the mediastinum. Am J Surg Pathol. 2012; 36:1222–7.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Case Report of 4-Years Old Patient With Nuclear Protein in Testis Midline Carcinoma of Larynx
- Two Cases of Nuclear Protein in Testis Midline Carcinomas of Sinonasal Tract
- Radiologic Manifestations of Pulmonary Nuclear Protein in Testis Midline Carcinoma: A Case Report
- Expression of HLA-DR antigen in large bowel carcinoma
- Poorly Differentiated Carcinoma of the Thyroid Retrospective Clinical and mMrphologic Evaluation