J Vet Sci.
2018 Sep;19(5):635-642. 10.4142/jvs.2018.19.5.635.
Clostridium botulinum spores in Polish honey samples
- Affiliations
-
- 1Department of Hygiene of Animal Feedingstuffs, National Veterinary Research Institute, 24-100 Pulawy, Poland. tomasz.grenda@piwet.pulawy.pl
- 2Department of Honey Bee Diseases, National Veterinary Research Institute, 24-100 Pulawy, Poland.
- KMID: 2420932
- DOI: http://doi.org/10.4142/jvs.2018.19.5.635
Abstract
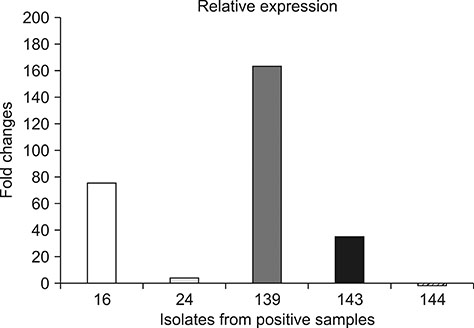
- The aim of this study was an examination of 240 multifloral honey samples collected from Polish apiaries to determine Clostridium botulinum occurrence. Honey was collected from apiaries directly after the extraction process. Samples were inoculated by using the dilution and centrifugation method. Suspected isolates were examined by using mouse bioassay, polymerase chain reaction (PCR), and real-time PCR methods. C. botulinum type A and B strains were detected in 5 of 240 examined honey samples (2.1%). Bacterial strains were also detected that were phenotypically similar to C. botulinum but that did not exhibit the ability to produce botulinum toxins and did not show the presence of the botulinum cluster (ntnh and bont genes) or expression of the ntnh gene. The methods used in the examination, especially the expression analysis of ntnh gene, enabled specific analysis of suspected strains and could be used routinely in environmental isolate analyses of C. botulinum occurrence.
Keyword
MeSH Terms
Figure
Reference
-
1. Arnon SS, Midura TF, Damus K, Thompson B, Wood RM, Chin J. Honey and other environmental risk factors for infant botulism. J Pediatr. 1979; 94:331–336.
Article2. Austin JW. Clostridium botulinum. In : Beuchat LR, Doyle MP, Montville TJ, editors. Food Microbiology: Fundamentals and Frontiers. 2nd ed. Washington: ASM Press;2001. p. 329–349.3. Carter AT, Peck MW. Genomes, neurotoxins and biology of Clostridium botulinum Group I and Group II. Res Microbiol. 2015; 166:303–317.
Article4. Collins MD, East AK. Phylogeny and taxonomy of the food-borne pathogen Clostridium botulinum and its neurotoxins. J Appl Microbiol. 1998; 84:5–17.
Article5. De Medici D, Anniballi F, Wyatt GM, Lindström M, Messelhäusser U, Aldus CF, Delibato E, Korkeala H, Peck MW, Fenicia L. Multiplex PCR for detection of botulinum neurotoxin-producing clostridia in clinical, food, and environmental samples. Appl Environ Microbiol. 2009; 75:6457–6461.
Article6. Dover N, Barash JR, Hill KK, Xie G, Arnon SS. Molecular characterization of a novel botulinum neurotoxin type H gene. J Infect Dis. 2014; 209:192–202.
Article7. Glass K, Marshall K. Clostridium botulinum. In : Morris JG, Potter ME, editors. Foodborne Infections and Intoxications. 4th ed. Boston: Elsevier;2013. p. 371–387.8. Grenda T, Grabczak M, Kwiatek K, Bober A. Prevalence of C. botulinum and C. perfringens spores in food products available on Polish market. J Vet Res. 2017; 61:287–291.
Article9. Grenda T, Kukier E, Kwiatek K. Methods and difficulties in detection of Clostridium botulinum and its toxins. Pol J Vet Sci. 2014; 17:195–205.
Article10. Gücükoğlu A, Terzi G, Çadirci Ö, Alişarli M, Kevenk O, Uyanik T. Detection of C. botulinum types in honey by mPCR. J Food Sci. 2014; 79:M600–M603.11. Hatheway CL. Botulism: the present status of the disease. Curr Top Microbiol Immunol. 1995; 195:55–75.
Article12. Hatheway CL. Toxigenic clostridia. Clin Microbiol Rev. 1990; 3:66–98.
Article13. Küplülü Ö, Göncüoğlu M, Özdemir H, Koluman A. Incidence of Clostridium botulinum spores in honey in Turkey. Food Control. 2006; 17:222–224.
Article14. Lindström M, Korkeala H. Laboratory diagnostics of botulism. Clin Microbiol Rev. 2006; 19:298–314.
Article15. Midura TF. Update: infant botulism. Clin Microbiol Rev. 1996; 9:119–125.
Article16. Mustafina R, Maikanov B, Wiśniewski J, Tracz M, Anusz K, Grenda T, Kukier E, Goldsztejn M, Kwiatek K. Contamination of honey produced in the Republic of Kazakhstan with Clostridium botulinum. Bull Vet Inst Pulawy. 2015; 59:241–246.
Article17. Nakano H, Okabe T, Hashimoto H, Sakaguchi G. Incidence of Clostridium botulinum in honey of various origins. Jpn J Med Sci Biol. 1990; 43:183–195.18. Nakano H, Sakaguchi G. An unusually heavy contamination of honey products by Clostridium botulinum type F and Bacillus alvei. FEMS Microbiol Lett. 1991; 63:171–177.19. Nevas M, Lindström M, Hautamäki K, Puoskari S, Korkeala H. Prevalence and diversity of Clostridium botulinum types A, B, E and F in honey produced in the Nordic countries. Int J Food Microbiol. 2005; 105:145–151.
Article20. Pickett J, Berg B, Chaplin E, Brunstetter-Shafer MA. Syndrome of botulism in infancy: clinical and electrophysiologic study. N Engl J Med. 1976; 295:770–772.
Article21. Raphael BH, Andreadis JD. Real-time PCR detection of the nontoxic nonhemagglutinin gene as a rapid screening method for bacterial isolates harboring the botulinum neurotoxin (A-G) gene complex. J Microbiol Methods. 2007; 71:343–346.
Article22. Schmittgen TD, Livak KJ. Analyzing real-time PCR data by the comparative CT method. Nat Protoc. 2008; 3:1101–1108.
Article23. Solomon HM, Lilly T Jr. Clostridium botulinum. In : Merker RI, editor. Bacteriological Analytical Manual Online, Revision A. Maryland: Center for Food Safety and Applied Nutrition, Food and Drug Administration;2001.24. Tanzi MG, Gabay MP. Association between honey consumption and infant botulism. Pharmacotherapy. 2002; 22:1479–1483.
Article25. Vaneechoutte M, Cartwright CP, Williams EC, Jäger B, Tichy HV, De Baere T, De Rouck A, Verschraegen G. Evaluation of 16S rRNA gene restriction analysis for the identification of cultured organisms of clinically important Clostridium species. Anaerobe. 1996; 2:249–256.
Article26. Wojtacka J, Wysok B, Kabašinškienė A, Wiszniewska-Łaszczych A, Gomółka-Pawlicka M, Szteyn J, Malakauskas M, Migowska-Calik A. Prevalence of Clostridium botulinum type A, B, E and F isolated from directly sold honey in Lithuania. J Agr Sci Tech. 2017; 19:335–343.27. Wojtacka J, Wysok B, Lipiński Z, Gomółka-Pawlicka M, Rybak-Chmielewska H, Wiszniewska-Łaszczych A. Clostridium botulinum spores found in honey from small apiaries in Poland. J Apic Sci. 2016; 60:89–100.
Article28. Zhang S, Masuyer G, Zhang J, Shen Y, Lundin D, Henriksson L, Miyashita SI, Martínez-Carranza M, Dong M, Stenmark P. Identification and characterization of a novel botulinum neurotoxin. Nat Commun. 2017; 8:14130.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Molecular Mechanism of the Action of Clostridium botulinum Type B Neurotoxin
- Efficient Method for the Rapid Purification of Nosema ceranae Spores
- The First Outbreak of Botulism in Korea
- Prevalence of Nosema and Virus in Honey Bee (Apis mellifera L.) Colonies on Flowering Period of Acacia in Korea
- A clinical study on the use of botulinum toxin type a in maxillofacial area