Transl Clin Pharmacol.
2018 Mar;26(1):32-38. 10.12793/tcp.2018.26.1.32.
Quantitative analysis of acetylsalicylic acid in human blood using volumetric absorptive microsampling
- Affiliations
-
- 1Center for Clinical Pharmacology and Biomedical Research Institute, Chonbuk National University Hospital, Jeonju 54907, Republic of Korea. mgkim@jbnu.ac.kr
- 2Department of Pharmacology, School of Medicine, Chonbuk National University, Jeonju 54907, Republic of Korea.
- KMID: 2420305
- DOI: http://doi.org/10.12793/tcp.2018.26.1.32
Abstract
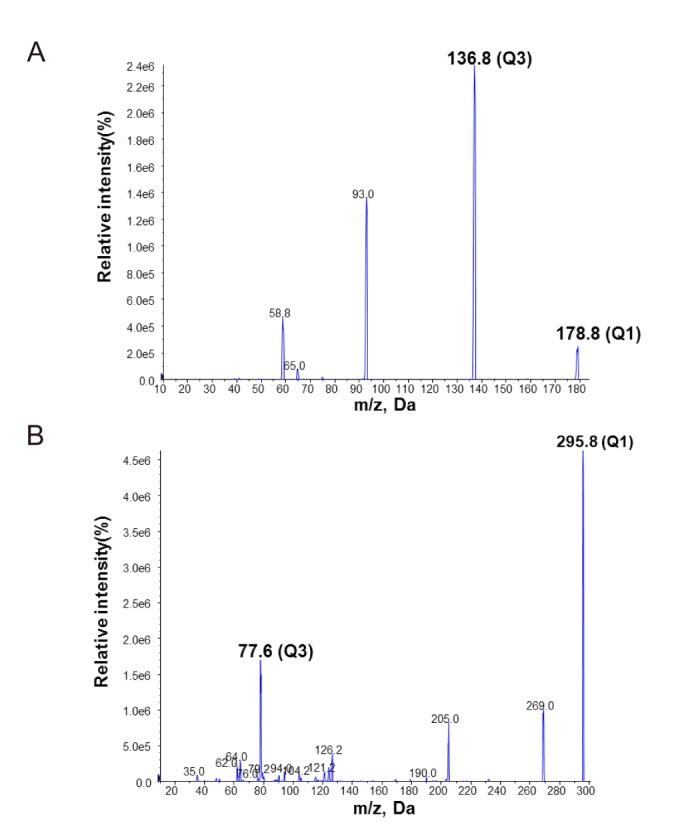
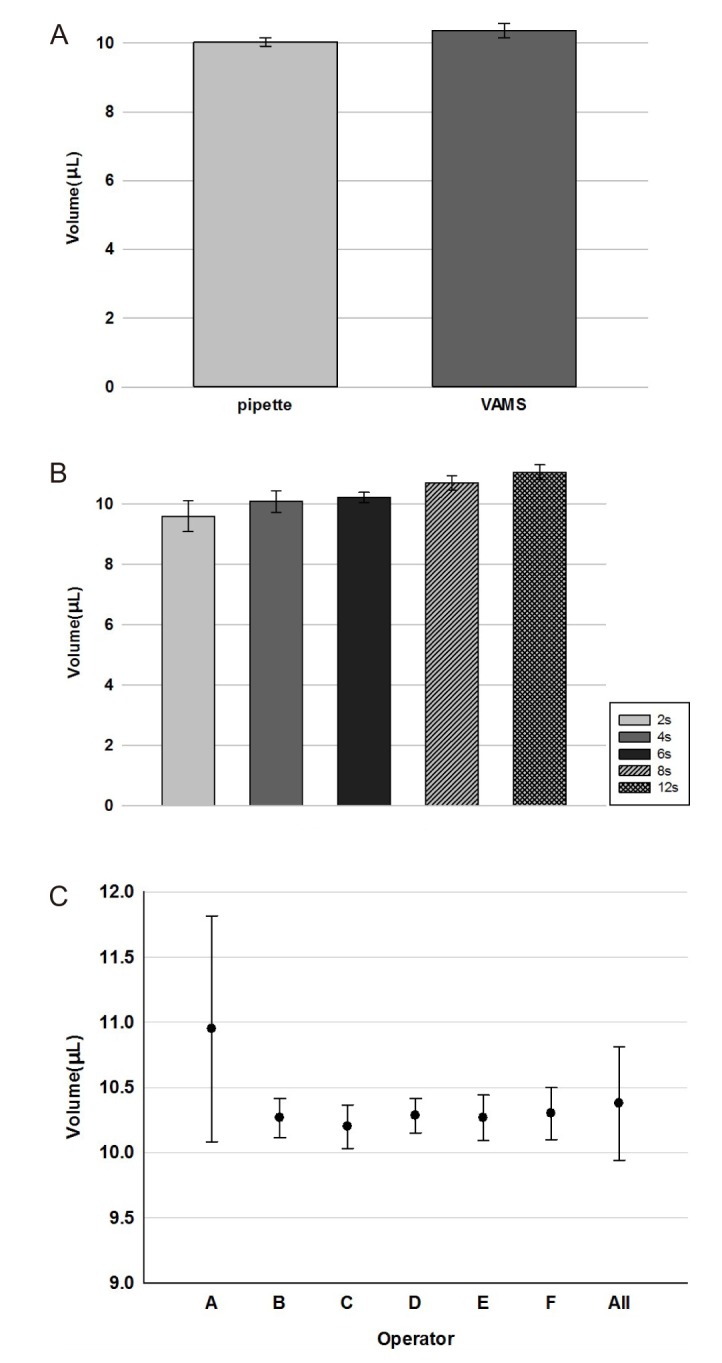
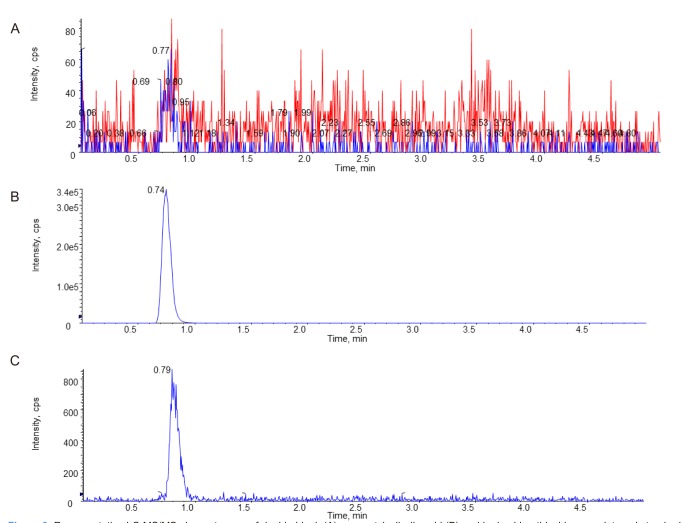
- Volumetric absorptive microsampling (VAMS) is a novel sampling technique that allows for the collection of an accurate volume of blood by dipping a microsampler tip. The purpose of this study is to compare the requirement of a stabilizing reagent for the conventional venous blood sampling method versus VAMS in the analytical measurement of the concentration of acetylsalicylic acid. A high-performance liquid chromatography with mass spectrometry (LC-MS/MS) method was developed and validated for the accurate determination of acetylsalicylic acid in human blood. The blood samples spiked with acetylsalicylic acid with and without stabilizing reagent were absorbed into VAMS tips. In the whole blood sample, the same concentration was shown regardless of the addition of the stabilizing reagent, but the concentration decreased when the stabilizing reagent was not added to the VAMS sample. To apply the VAMS technology as a new blood sampling method, stabilizing reagents should be added before the analysis of acetylsalicylic acid concentration.
MeSH Terms
Figure
Reference
-
1. Denniff P, Parry S, Dopson W, Spooner N. Quantitative bioanalysis of paracetamol in rats using volumetric absorptive microsampling (VAMS). J Pharm Biomed Anal. 2015; 108:61–69. DOI: 10.1016/j.jpba.2015.01.052. PMID: 25710904.
Article2. De Kesel PM, Lambert WE, Stove CP. Does volumetric absorptive microsampling eliminate the hematocrit bias for caffeine and paraxanthine in dried blood samples? A comparative study. Anal Chim Acta. 2015; 881:65–73. DOI: 10.1016/j.aca.2015.04.056. PMID: 26041521.
Article3. Denniff P, Spooner N. Volumetric absorptive microsampling: a dried sample collection technique for quantitative bioanalysis. Anal Chem. 2014; 86:8489–8495. DOI: 10.1021/ac5022562. PMID: 25058158.
Article4. Kip AE, Kiers KC, Rosing H, Schellens JHM, Beijnen JH, Dorlo TPC. Volumetric absorptive microsampling (VAMS) as an alternative to conventional dried blood spots in the quantification of miltefosine in dried blood samples. J Pharm Biomed Anal. 2017; 135:160–166. DOI: 10.1016/j.jpba.2016.12.012. PMID: 28033553.
Article5. Mano Y, Kita K, Kusano K. Hematocrit-independent recovery is a key for bioanalysis using volumetric absorptive microsampling devices, Mitra. Bioanalysis. 2015; 7:1821–1829. DOI: 10.4155/bio.15.111. PMID: 26295984.6. Buchanan MR, Rischke JA, Hirsh J. Aspirin inhibits platelet function independent of the acetylation of cyclo-oxygenase. Thromb Res. 1982; 25:363–373. PMID: 7071811.7. Lemos Silva R, Carvalho de Sousa J, Calisto C, Braz Nogueira JM, Ravara L. Oral anticoagulant therapy. Fundamentals, clinical practice and recommendations. Rev Port Cardiol. 2007; 26:769–788. PMID: 17939586.8. Jeong KH, Kim JY, Choi YS, Lee MY, Kim SY. Influence of aspirin on pilocarpine-induced epilepsy in mice. Korean J Physiol Pharmacol. 2013; 17:15–21. DOI: 10.4196/kjpp.2013.17.1.15. PMID: 23439794.
Article9. Singh GB, Leach GD, Atal CK. Antiinflammatory actions of methyl- and phenyl-3-methoxy-4-hydroxy styryl ketones. Arzneimittelforschung. 1987; 37:435–440. PMID: 3496890.10. Ali MA, Routh JI. The protein binding of acetylsalicylic acid and salicylic acid. Clin Chem. 1969; 15:1027–1038. PMID: 5357059.
Article11. Ibrahim H, Boyer A, Bouajila J, Couderc F, Nepveu F. Determination of non-steroidal anti-inflammatory drugs in pharmaceuticals and human serum by dual-mode gradient HPLC and fluorescence detection. J Chromatogr B Analyt Technol Biomed Life Sci. 2007; 857:59–66.
Article12. Feldman M, Shewmake K, Cryer B. Time course inhibition of gastric and platelet COX activity by acetylsalicylic acid in humans. Am J Physiol Gastrointest Liver Physiol. 2000; 279:G1113–G1120. PMID: 11053009.
Article13. Castillo-García ML, Aguilar-Caballos MP, Gómez-Hens A. Determination of acetylsalicylic acid and its major metabolites in bovine urine using ultra performance liquid chromatography. J Chromatogr B Analyt Technol Biomed Life Sci. 2015; 985:85–90. DOI: 10.1016/j.jchromb.2015.01.026.
Article14. Fogel J, Epstein P, Chen P. Simultaneous high-performance liquid chromatography assay of acetylsalicylic acid and salicylic acid in film-coated aspirin tablets. J Chromatogr. 1984; 317:507–511. PMID: 6530452.
Article15. O'Kruk RJ, Adams MA, Philp RB. Rapid and sensitive determination of acetylsalicylic acid and its metabolites using reversed-phase high-performance liquid chromatography. J Chromatogr. 1984; 310:343–352. PMID: 6511852.16. Zaugg S, Zhang X, Sweedler J, Thormann W. Determination of salicylate, gentisic acid and salicyluric acid in human urine by capillary electrophoresis with laser-induced fluorescence detection. J Chromatogr B Biomed Sci Appl. 2001; 752:17–31. PMID: 11254191.
Article17. Gu N, Kim BH, Chung YJ, Lim KS, Seo HB, Yim DS, et al. Influence of Simvastatin on Pharmacokinetics/Pharmacodynamics of Aspirin after Oral Co-administration in Healthy Volunteers. J Korean Soc Clin Pharmacol Ther. 2011; 19:73–83.
Article18. Nagelschmitz J, Blunck M, Kraetzschmar J, Ludwig M, Wensing G, Hohlfeld T. Pharmacokinetics and pharmacodynamics of acetylsalicylic acid after intravenous and oral administration to healthy volunteers. Clin Pharmacol. 2014; 6:51–59. DOI: 10.2147/CPAA.S47895. PMID: 24672263.
Article19. Bae SK, Seo KA, Jung EJ, Kim HS, Yeo CW, Shon JH, et al. Determination of acetylsalicylic acid and its major metabolite, salicylic acid, in human plasma using liquid chromatography-tandem mass spectrometry: application to pharmacokinetic study of Astrix in Korean healthy volunteers. Biomed Chromatogr. 2008; 22:590–595. DOI: 10.1002/bmc.973. PMID: 18254152.
Article20. Halder D, Dan S, Biswas E, Sarkar P, Halder UC, Pal TK. A Rapid LCESI-MS/MS Method for the Quantitation of Salicylic Acid, an Active Metabolite of Acetylsalicylic Acid. Appl Clin Res Clin Trials Regul Aff. 2015; 2:90–102.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Stability of acetylsalicylic acid in human blood collected using volumetric absorptive microsampling (VAMS) under various drying conditions
- Hematocrit Determination using a Volumetric Absorptive Microsampling Technique in Patients with Pancreatic Cancer
- Validation of LC-MS/MS method for determination of rosuvastatin concentration in human blood collected by volumetric absorptive microsampling (VAMS)
- Three Casesof Livedo Vasculitis Cleared by Combined Therapy of Acetylsalicylic Acid , Dipyridamole and Nifedipine
- A Case of Atropie Blanche Accompanied by Cryoglobulinemia