Obstet Gynecol Sci.
2018 Jan;61(1):14-22. 10.5468/ogs.2018.61.1.14.
Effects of corticotropin-releasing hormone on the expression of adenosine triphosphate-sensitive potassium channels (Kir6.1/SUR2B) in human term pregnant myometrium
- Affiliations
-
- 1Department of Obstetrics and Gynecology, Eulji General Hospital, Eulji University, Seoul, Korea. obdrseo@eulji.ac.kr
- 2Department of Obstetrics and Gynecology, Eulji Medi-Bio Research Institute, Eulji University, Daejeon, Korea.
- KMID: 2420147
- DOI: http://doi.org/10.5468/ogs.2018.61.1.14
Abstract
OBJECTIVE
Corticotropin-releasing hormone (CRH) is a crucial regulator of human pregnancy and parturition. Adenosine triphosphate (ATP)-sensitive potassium (KATP) channels are important for regulating myometrial quiescence during pregnancy. We investigated regulatory effects of different concentrations of CRH on KATP channel expression in human myometrial smooth muscle cells (HSMCs) in in vitro conditions.
METHODS
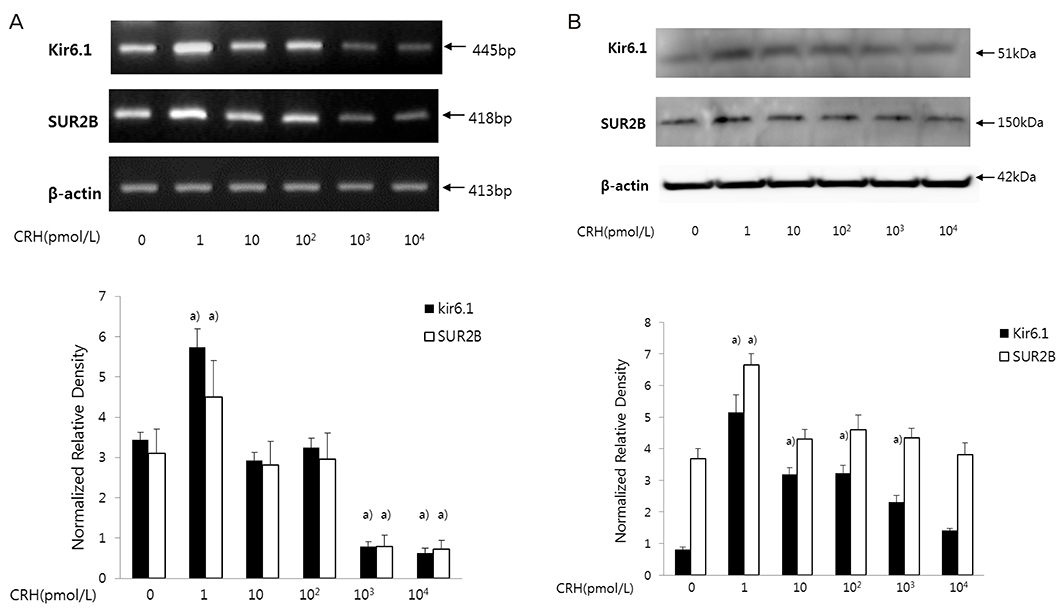
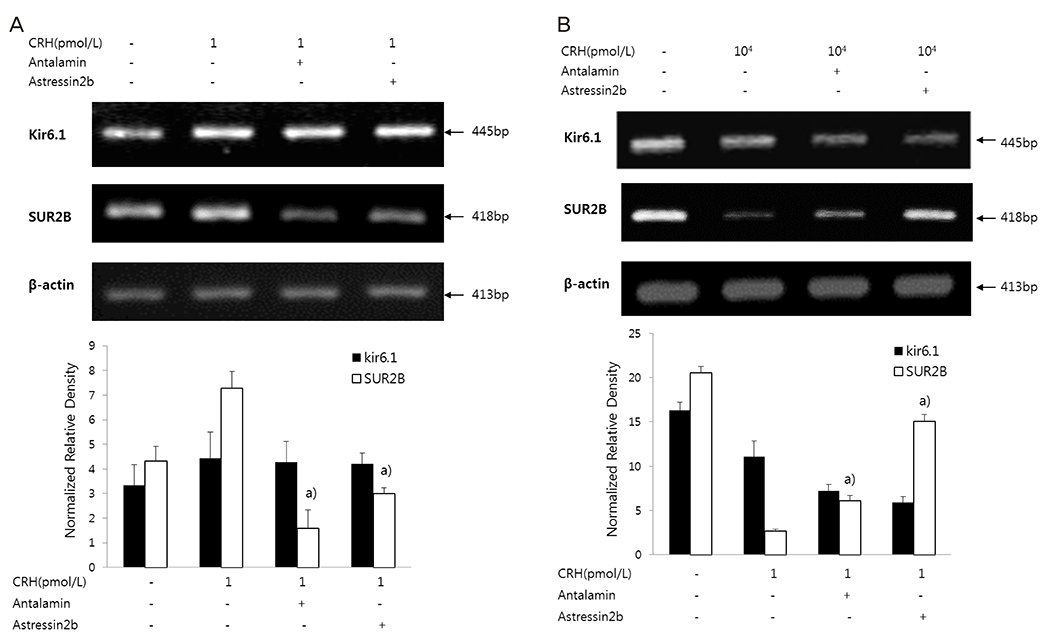
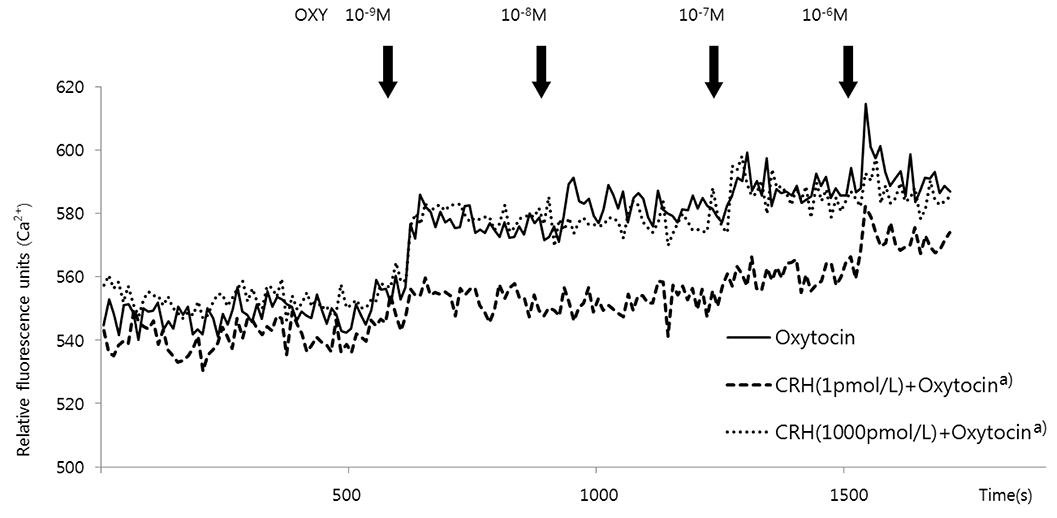
After treating HSMCs with different concentrations of CRH (1, 10, 102, 103, 104 pmol/L), mRNA and protein expression of KATP channel subunits (Kir6.1 and SUR2B) was analyzed by reverse transcription-polymerase chain reaction and western blot. We investigated which CRH receptor was involved in the reaction and measured the effects of CRH on intracellular Ca2+ concentration when oxytocin was administered in HSMCs using Fluo-8 AM ester.
RESULTS
When HSMCs were treated with low (1 pmol/L) and high (103, 104 pmol/L) CRH concentrations, KATP channel expression significantly increased and decreased, respectively. SUR2B mRNA expression at low and high CRH concentrations was significantly antagonized by antalarmin (CRH receptor-1 antagonist) and astressin 2b (CRH receptor-2 antagonist), respectively; however, Kir6.1 mRNA expression was not affected. After oxytocin treatment, the intracellular Ca2+ concentration in CRH-treated HSMCs was significantly lowered in low concentration of CRH (1 pmol/L), but not in high concentration of CRH (103 pmol/L), compared to control.
CONCLUSION
Our data demonstrated the regulatory effect was different when HSMCs were treated with low (early pregnancy-like) and high (labor-like) CRH concentrations and the KATP channel expression showed significant increase and decrease. This could cause inhibition and activation, respectively, of uterine muscle contraction, demonstrating opposite dual actions of CRH.
MeSH Terms
-
Adenosine Triphosphate
Adenosine*
Animals
Blotting, Western
Corticotropin-Releasing Hormone*
Female
Humans*
In Vitro Techniques
KATP Channels
Mice
Myocytes, Smooth Muscle
Myometrium*
Oxytocin
Parturition
Potassium Channels*
Potassium*
Pregnancy
Receptors, Corticotropin-Releasing Hormone
RNA, Messenger
Adenosine
Adenosine Triphosphate
Corticotropin-Releasing Hormone
KATP Channels
Oxytocin
Potassium
Potassium Channels
RNA, Messenger
Receptors, Corticotropin-Releasing Hormone
Figure
Reference
-
1. McCormick MC. The contribution of low birth weight to infant mortality and childhood morbidity. N Engl J Med. 1985; 312:82–90.
Article2. Khan RN, Matharoo-Ball B, Arulkumaran S, Ashford ML. Potassium channels in the human myometrium. Exp Physiol. 2001; 86:255–264.
Article3. Brainard AM, Korovkina VP, England SK. Potassium channels and uterine function. Semin Cell Dev Biol. 2007; 18:332–339.
Article4. Morrison JJ, Ashford ML, Khan RN, Smith SK. The effects of potassium channel openers on isolated pregnant human myometrium before and after the onset of labor: potential for tocolysis. Am J Obstet Gynecol. 1993; 169:1277–1285.
Article5. Cheuk JM, Hollingsworth M, Hughes SJ, Piper IT, Maresh MJ. Inhibition of contractions of the isolated human myometrium by potassium channel openers. Am J Obstet Gynecol. 1993; 168:953–960.
Article6. Okawa T, Vedernikov YP, Saade GR, Longo M, Olson GL, Chwalisz K, et al. Roles of potassium channels and nitric oxide in modulation of uterine contractions in rat pregnancy. Am J Obstet Gynecol. 1999; 181:649–655.
Article7. Sadlonova V, Franova S, Dokus K, Janicek F, Visnovsky J, Sadlonova J. Participation of BKCa2+ and KATP potassium ion channels in the contractility of human term pregnant myometrium in in vitro conditions. J Obstet Gynaecol Res. 2011; 37:215–221.8. Novakovic R, Milovanovic S, Protic D, Djokic J, Heinle H, Gojkovic-Bukarica L. The effect of potassium channel opener pinacidil on the non-pregnant rat uterus. Basic Clin Pharmacol Toxicol. 2007; 101:181–186.
Article9. Mandi G, Sarkar SN, Mishra SK, Raviprakash V. Effects of calcium channel blocker, mibefradil, and potassium channel opener, pinacidil, on the contractile response of mid-pregnant goat myometrium. Indian J Exp Biol. 2005; 43:795–801.10. Aguilar-Bryan L, Clement JP 4th, Gonzalez G, Kunjilwar K, Babenko A, Bryan J. Toward understanding the assembly and structure of KATP channels. Physiol Rev. 1998; 78:227–245.
Article11. Curley M, Cairns MT, Friel AM, McMeel OM, Morrison JJ, Smith TJ. Expression of mRNA transcripts for ATP-sensitive potassium channels in human myometrium. Mol Hum Reprod. 2002; 8:941–945.
Article12. Xu C, You X, Gao L, Zhang L, Hu R, Hui N, et al. Expression of ATP-sensitive potassium channels in human pregnant myometrium. Reprod Biol Endocrinol. 2011; 9:35.
Article13. Sasaki A, Tempst P, Liotta AS, Margioris AN, Hood LE, Kent SB, et al. Isolation and characterization of a corticotropin-releasing hormone-like peptide from human placenta. J Clin Endocrinol Metab. 1988; 67:768–773.
Article14. Goland RS, Wardlaw SL, Blum M, Tropper PJ, Stark RI. Biologically active corticotropin-releasing hormone in maternal and fetal plasma during pregnancy. Am J Obstet Gynecol. 1988; 159:884–890.
Article15. Sasaki A, Shinkawa O, Margioris AN, Liotta AS, Sato S, Murakami O, et al. Immunoreactive corticotropin-releasing hormone in human plasma during pregnancy, labor, and delivery. J Clin Endocrinol Metab. 1987; 64:224–229.
Article16. Goland RS, Wardlaw SL, Stark RI, Brown LS Jr, Frantz AG. High levels of corticotropin-releasing hormone immunoactivity in maternal and fetal plasma during pregnancy. J Clin Endocrinol Metab. 1986; 63:1199–1203.
Article17. McLean M, Bisits A, Davies J, Woods R, Lowry P, Smith R. A placental clock controlling the length of human pregnancy. Nat Med. 1995; 1:460–463.
Article18. Korebrits C, Ramirez MM, Watson L, Brinkman E, Bocking AD, Challis JR. Maternal corticotropin-releasing hormone is increased with impending preterm birth. J Clin Endocrinol Metab. 1998; 83:1585–1591.
Article19. Warren WB, Patrick SL, Goland RS. Elevated maternal plasma corticotropin-releasing hormone levels in pregnancies complicated by preterm labor. Am J Obstet Gynecol. 1992; 166:1198–1204.
Article20. Makrigiannakis A, Semmler M, Briese V, Eckerle H, Minas V, Mylonas I, et al. Maternal serum corticotropin-releasing hormone and ACTH levels as predictive markers of premature labor. Int J Gynaecol Obstet. 2007; 97:115–119.
Article21. Sawada K, Morishige K, Hashimoto K, Tasaka K, Kurachi H, Murata Y, et al. Gestational change of K+ channel opener effect is correlated with the expression of uterine KATP channel subunits. Eur J Obstet Gynecol Reprod Biol. 2005; 122:49–56.
Article22. Campbell EA, Linton EA, Wolfe CD, Scraggs PR, Jones MT, Lowry PJ. Plasma corticotropin-releasing hormone concentrations during pregnancy and parturition. J Clin Endocrinol Metab. 1987; 64:1054–1059.
Article23. Guendelman S, Kosa JL, Pearl M, Graham S, Kharrazi M. Exploring the relationship of second-trimester corticotropin releasing hormone, chronic stress and preterm delivery. J Matern Fetal Neonatal Med. 2008; 21:788–795.
Article24. Zhang LM, Wang YK, Hui N, Sha JY, Chen X, Guan R, et al. Corticotropin-releasing hormone acts on CRH-R1 to inhibit the spontaneous contractility of non-labouring human myometrium at term. Life Sci. 2008; 83:620–624.
Article25. Simpkin JC, Kermani F, Palmer AM, Campa JS, Tribe RM, Linton EA, et al. Effects of corticotrophin releasing hormone on contractile activity of myometrium from pregnant women. Br J Obstet Gynaecol. 1999; 106:439–445.
Article26. Tyson EK, Smith R, Read M. Evidence that corticotropin-releasing hormone modulates myometrial contractility during human pregnancy. Endocrinology. 2009; 150:5617–5625.
Article27. Hillhouse EW, Grammatopoulos DK. Control of intracellular signalling by corticotropin-releasing hormone in human myometrium. Front Horm Res. 2001; 27:66–74.
Article28. Aggelidou E, Hillhouse EW, Grammatopoulos DK. Up-regulation of nitric oxide synthase and modulation of the guanylate cyclase activity by corticotropin-releasing hormone but not urocortin II or urocortin III in cultured human pregnant myometrial cells. Proc Natl Acad Sci USA. 2002; 99:3300–3305.
Article29. Grammatopoulos DK. The role of CRH receptors and their agonists in myometrial contractility and quiescence during pregnancy and labour. Front Biosci. 2007; 12:561–571.
Article30. Quartero HW, Fry CH. Placental corticotrophin releasing factor may modulate human parturition. Placenta. 1989; 10:439–443.
Article31. Mignot TM, Paris B, Carbonne B, Vauge C, Ferré F, Vaiman D. Corticotropin-releasing hormone effects on human pregnant vs. nonpregnant myometrium explants estimated from a mathematical model of uterine contraction. J Appl Physiol. 1985; 2005:1157–1163.
Article32. Grammatopoulos D, Thompson S, Hillhouse EW. The human myometrium expresses multiple isoforms of the corticotropin-releasing hormone receptor. J Clin Endocrinol Metab. 1995; 80:2388–2393.
Article33. Hillhouse EW, Randeva H, Ladds G, Grammatopoulos D. Corticotropin-releasing hormone receptors. Biochem Soc Trans. 2002; 30:428–432.
Article34. Li C, Chen P, Vaughan J, Blount A, Chen A, Jamieson PM, et al. Urocortin III is expressed in pancreatic beta-cells and stimulates insulin and glucagon secretion. Endocrinology. 2003; 144:3216–3224.35. Karteris E, Hillhouse EW, Grammatopoulos D. Urocortin II is expressed in human pregnant myometrial cells and regulates myosin light chain phosphorylation: potential role of the type-2 corticotropin-releasing hormone receptor in the control of myometrial contractility. Endocrinology. 2004; 145:890–900.
Article36. Jovanović A, Jovanović S. SURA2 targeting for cardioprotection? Curr Opin Pharmacol. 2009; 9:189–193.
Article37. Du Q, Jovanović S, Sukhodub A, Jovanović A. Infection with AV-SUR2A protects H9C2 cells against metabolic stress: a mechanism of SUR2A-mediated cytoprotection independent from the K(ATP) channel activity. Biochim Biophys Acta. 2010; 1803:405–415.
Article38. Ballantyne T, Du Q, Jovanović S, Neemo A, Holmes R, Sinha S, et al. Testosterone protects female embryonic heart H9c2 cells against severe metabolic stress by activating estrogen receptors and up-regulating IES SUR2B. Int J Biochem Cell Biol. 2013; 45:283–291.
Article39. Jovanović S, Ballantyne T, Du Q, Blagojević M, Jovanović A. Phenylephrine preconditioning in embryonic heart H9c2 cells is mediated by up-regulation of SUR2B/Kir6.2: a first evidence for functional role of SUR2B in sarcolemmal KATP channels and cardioprotection. Int J Biochem Cell Biol. 2016; 70:23–28.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Differential Effect of Intracellular ATP on Skeletal-and Smooth Muscle-Type KATP Channel Activities
- Neonatal Diabetes Caused by Activating Mutations in the Sulphonylurea Receptor
- Identification of ATP-sensitive K+ Channel in an Uterine Leiomyoma Cell
- Expression of ATP-sensitive potassium channel and sulfonylurea receptor in neonate and adult rat tissues
- Role of potassium channels in female reproductive system