Blood Res.
2018 Mar;53(1):61-70. 10.5045/br.2018.53.1.61.
Expression of adhesion molecules on CD34+ cells from steady-state bone marrow before and after mobilization and their association with the yield of CD34+ cells
- Affiliations
-
- 1Oncologia ClÃnica e Experimental, Universidade Federal de São Paulo – UNIFESP, Sao Paulo, Brazil. kcecyn@gmail.com
- KMID: 2414366
- DOI: http://doi.org/10.5045/br.2018.53.1.61
Abstract
- BACKGROUND
Cell adhesion molecules (CAMs) expressed on hematopoietic progenitor cells (HPCs), endothelial cells, and stromal cells play a pivotal role in the mobilization of CD34+ cells. Herein, we conducted a non-randomized peripheral blood stem cell (PBSC) mobilization study aimed to compare the potential differences in the expressions of several CAMs and chemokines on CD34+ cells obtained from bone marrow aspirate before and after HPC mobilization from patients with hematologic malignancies and healthy donors.
METHODS
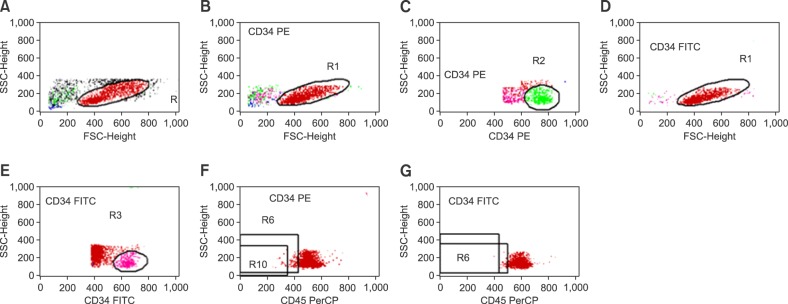
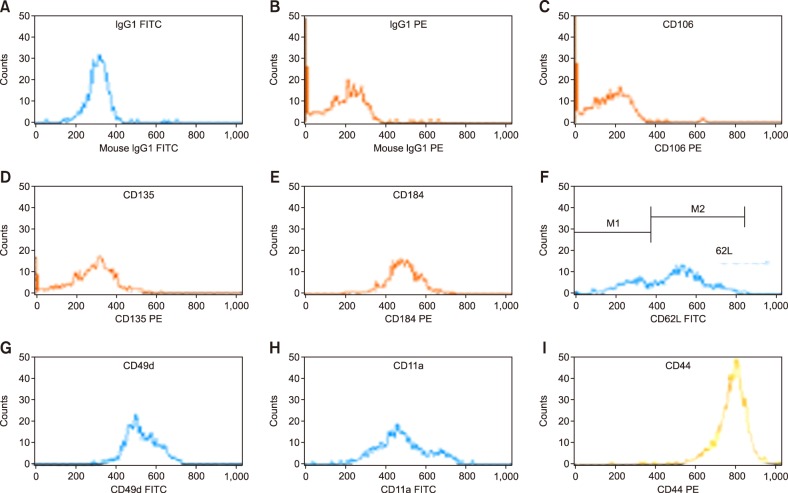
Three-color cytofluorometric analysis was used to compare the expressions of CAMs and chemokines in the bone marrow before and after mobilization.
RESULTS
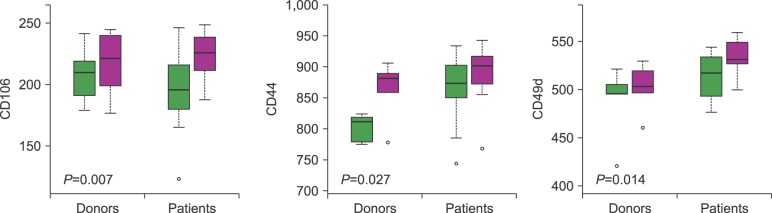
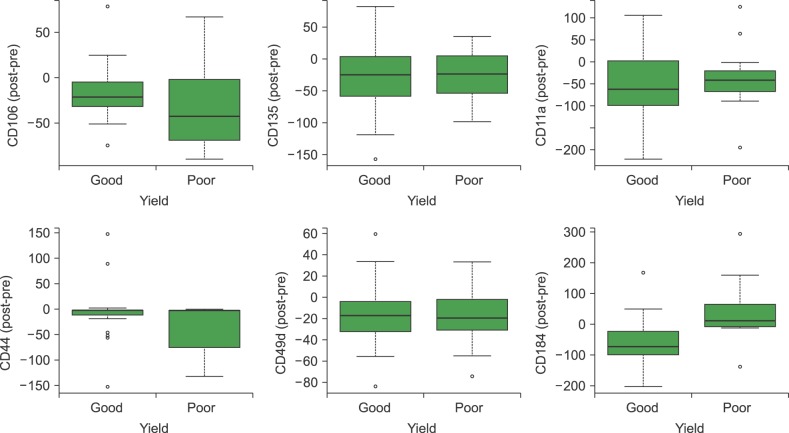
For all studied groups, CAM expression among those with good and poor yields of CD34+ cells was significantly correlated with VCAM-1 (P=0.007), CD44 (P=0.027), and VLA-4 (P=0.014) expressions. VCAM-1 (P=0.001), FLT-3 (P=0.001), CD44 (P=0.011), VLA-4 (P=0.001), and LFA-1 (P=0.001) expressions were higher before HPC mobilization than after HPC mobilization. By contrast, the expression of CXCR4 significantly varied before and after mobilization only among those with successful PBSC mobilization (P=0.002).
CONCLUSION
We attempted to identify particular aspects of CAMs involved in CD34+ cell mobilization, which is a highly complex mechanism that involves adhesion molecules and matrix metalloproteases. The mechanism by which CD34+ cell mobilization is activated through proteolytic enzymes is not fully understood. We believe that CXCR4, VLA-4, CD44, and VCAM-1 are the most important molecules implicated in HPC mobilization, particularly because they show a correlation with the yield of CD34+ cells collected via large volume leukapheresis.
Keyword
MeSH Terms
-
Bone Marrow*
Cell Adhesion Molecules
Chemokines
Endothelial Cells
Hematologic Neoplasms
Hematopoietic Stem Cells
Humans
Integrin alpha4beta1
Leukapheresis
Lymphocyte Function-Associated Antigen-1
Lymphoma, Non-Hodgkin
Metalloproteases
Multiple Myeloma
Peptide Hydrolases
Stem Cells
Stromal Cells
Tissue Donors
Vascular Cell Adhesion Molecule-1
Cell Adhesion Molecules
Chemokines
Integrin alpha4beta1
Lymphocyte Function-Associated Antigen-1
Metalloproteases
Peptide Hydrolases
Vascular Cell Adhesion Molecule-1
Figure
Reference
-
1. Arslan O, Moog R. Mobilization of peripheral blood stem cells. Transfus Apher Sci. 2007; 37:179–185. PMID: 17980665.
Article2. Ikeda K, Kozuka T, Harada M. Factors for PBPC collection efficiency and collection predictors. Transfus Apher Sci. 2004; 31:245–259. PMID: 15556472.
Article3. Haverkos BM, McBride A, O'Donnell L, et al. An effective mobilization strategy for lymphoma patients after failed upfront mobilization with plerixafor. Bone Marrow Transplant. 2014; 49:1052–1055. PMID: 24797182.
Article4. Angelopoulou MK, Tsirkinidis P, Boutsikas G, Vassilakopoulos TP, Tsirigotis P. New insights in the mobilization of hematopoietic stem cells in lymphoma and multiple myeloma patients. Biomed Res Int. 2014; 2014:835138. PMID: 25197663.
Article5. Yuan S, Wang S. How do we mobilize and collect autologous peripheral blood stem cells? Transfusion. 2017; 57:13–23. PMID: 27731496.
Article6. Olivieri A, Marchetti M, Lemoli R, et al. Proposed definition of ‘poor mobilizer’ in lymphoma and multiple myeloma: an analytic hierarchy process by ad hoc working group Gruppo ItalianoTrapianto di Midollo Osseo. Bone Marrow Transplant. 2012; 47:342–351. PMID: 21625224.
Article7. Fruehauf S, Seggewiss R. It's moving day: factors affecting peripheral blood stem cell mobilization and strategies for improvement. Br J Haematol. 2003; 122:360–375. PMID: 12877663.8. Kröger N, Zeller W, Hassan HT, Dierlamm J, Zander AR. Difference between expression of adhesion molecules on CD34+ cells from bone marrow and G-CSF-stimulated peripheral blood. Stem Cells. 1998; 16:49–53. PMID: 9474747.9. Chan JY, Watt SM. Adhesion receptors on haematopoietic progenitor cells. Br J Haematol. 2001; 112:541–557. PMID: 11260052.
Article10. Ford CD, Greenwood J, Anderson J, Snow G, Petersen FB. CD34+ cell adhesion molecule profiles differ between patients mobilized with granulocyte-colony-stimulating factor alone and chemotherapy followed by granulocyte-colony-stimulating factor. Transfusion. 2006; 46:193–198. PMID: 16441594.
Article11. Oostendorp RA, Reisbach G, Spitzer E, et al. VLA-4 and VCAM-1 are the principal adhesion molecules involved in the interaction between blast colony-forming cells and bone marrow stromal cells. Br J Haematol. 1995; 91:275–284. PMID: 8547062.
Article12. Lichterfeld M, Martin S, Burkly L, Haas R, Kronenwett R. Mobilization of CD34+ haematopoietic stem cells is associated with a functional inactivation of the integrin very late antigen 4. Br J Haematol. 2000; 110:71–81. PMID: 10930981.13. Lévesque JP, Hendy J, Takamatsu Y, Simmons PJ, Bendall LJ. Disruption of the CXCR4/CXCL12 chemotactic interaction during hematopoietic stem cell mobilization induced by GCSF or cyclophosphamide. J Clin Invest. 2003; 111:187–196. PMID: 12531874.
Article14. Devine SM, Flomenberg N, Vesole DH, et al. Rapid mobilization of CD34+ cells following administration of the CXCR4 antagonist AMD3100 to patients with multiple myeloma and non-Hodgkin's lymphoma. J Clin Oncol. 2004; 22:1095–1102. PMID: 15020611.
Article15. Girbl T, Lunzer V, Greil R, Namberger K, Hartmann TN. The CXCR4 and adhesion molecule expression of CD34+ hematopoietic cells mobilized by “on-demand” addition of plerixafor to granulocyte-colony-stimulating factor. Transfusion. 2014; 54:2325–2335. PMID: 24673458.16. Avigdor A, Goichberg P, Shivtiel S, et al. CD44 and hyaluronic acid cooperate with SDF-1 in the trafficking of human CD34+ stem/progenitor cells to bone marrow. Blood. 2004; 103:2981–2989. PMID: 15070674.
Article17. Szmigielska-Kaplon A, Szemraj J, Hamara K, et al. Polymorphism of CD44 influences the efficacy of CD34(+) cells mobilization in patients with hematological malignancies. Biol Blood Marrow Transplant. 2014; 20:986–991. PMID: 24680978.
Article18. Bojko P, Pawloski D, Stellberg W, Schröder JK, Seeber S. Flt3 ligand and thrombopoietin serum levels during peripheral blood stem cell mobilization with chemotherapy and recombinant human glycosylated granulocyte colony-stimulating factor (rhu-G-CSF, lenograstim) and after high-dose chemotherapy. Ann Hematol. 2002; 81:522–528. PMID: 12373354.19. Papayannopoulou T, Nakamoto B, Andrews RG, Lyman SD, Lee MY. In vivo effects of Flt3/Flk2 ligand on mobilization of hematopoietic progenitors in primates and potent synergistic enhancement with granulocyte colony-stimulating factor. Blood. 1997; 90:620–629. PMID: 9226162.
Article20. He S, Chu J, Vasu S, et al. FLT3L and plerixafor combination increases hematopoietic stem cell mobilization and leads to improved transplantation outcome. Biol Blood Marrow Transplant. 2014; 20:309–313. PMID: 24365795.
Article21. Zubair AC, Rymer R, Young J, et al. Multiple myeloma patients receiving large volume leukapheresis efficiently yield enough CD34+ cells to allow double transplants. J Clin Apher. 2009; 24:6–11. PMID: 19156756.
Article22. Sutherland DR, Anderson L, Keeney M, Nayar R, Chin-Yee I. The ISHAGE guidelines for CD34+ cell determination by flow cytometry. International Society of Hematotherapy and Graft Engineering. J Hematother. 1996; 5:213–226. PMID: 8817388.23. Petty JM, Lenox CC, Weiss DJ, Poynter ME, Suratt BT. Crosstalk between CXCR4/stromal derived factor-1 and VLA-4/VCAM-1 pathways regulates neutrophil retention in the bone marrow. J Immunol. 2009; 182:604–612. PMID: 19109194.
Article24. Südhoff T, Söhngen D. Circulating endothelial adhesion molecules (sE-selectin, sVCAM-1 and sICAM-1) during rHuG-CSF-stimulated stem cell mobilization. J Hematother Stem Cell Res. 2002; 11:147–151. PMID: 11847011.
Article25. Papayannopoulou T, Priestley GV, Nakamoto B, Zafiropoulos V, Scott LM. Molecular pathways in bone marrow homing: dominant role of alpha(4)beta(1) over beta(2)-integrins and selectins. Blood. 2001; 98:2403–2411. PMID: 11588037.26. Lévesque JP, Takamatsu Y, Nilsson SK, Haylock DN, Simmons PJ. Vascular cell adhesion molecule-1 (CD106) is cleaved by neutrophil proteases in the bone marrow following hematopoietic progenitor cell mobilization by granulocyte colony-stimulating factor. Blood. 2001; 98:1289. PMID: 11520773.27. Velders GA, Pruijt JF, Verzaal P, et al. Enhancement of G-CSF-induced stem cell mobilization by antibodies against the beta 2 integrins LFA-1 and Mac-1. Blood. 2002; 100:327–333. PMID: 12070044.28. Gunji Y, Nakamura M, Hagiwara T, et al. Expression and function of adhesion molecules on human hematopoietic stem cells: CD34+ LFA-1- cells are more primitive than CD34+ LFA-1+ cells. Blood. 1992; 80:429–436. PMID: 1378320.
Article29. Zöller M. CD44, hyaluronan, the hematopoietic stem cell, and leukemia-initiating cells. Front Immunol. 2015; 6:235. PMID: 26074915.
Article30. Lapidot T, Petit I. Current understanding of stem cell mobilization: the roles of chemokines, proteolytic enzymes, adhesion molecules, cytokines, and stromal cells. Exp Hematol. 2002; 30:973–981. PMID: 12225788.31. Lee S, Im SA, Yoo ES, et al. Mobilization kinetics of CD34(+) cells in association with modulation of CD44 and CD31 expression during continuous intravenous administration of G-CSF in normal donors. Stem Cells. 2000; 18:281–286. PMID: 10924094.32. Fukuda S, Broxmeyer HE, Pelus LM. Flt3 ligand and the Flt3 receptor regulate hematopoietic cell migration by modulating the SDF-1alpha(CXCL12)/CXCR4 axis. Blood. 2005; 105:3117–3126. PMID: 15618475.33. He S, Chu J, Vasu S, et al. FLT3L and plerixafor combination increases hematopoietic stem cell mobilization and leads to improved transplantation outcome. Biol Blood Marrow Transplant. 2014; 20:309–313. PMID: 24365795.
Article34. Anandasabapathy N, Hurley A, Breton G, et al. A phase 1 trial of the hematopoietic growth factor CDX301 (rhuFlt3L) in healthy volunteers. Biol Blood Marrow Transplant. 2013; 19(Suppl):S112. (BMT Tandem Meetings Abstracts).
Article35. Cheng M, Qin G. Progenitor cell mobilization and recruitment: SDF-1, CXCR4, α4-integrin, and c-kit. Prog Mol Biol Transl Sci. 2012; 111:243–264. PMID: 22917234.
Article36. Carion A, Benboubker L, Hérault O, et al. Stromal-derived factor 1 and matrix metalloproteinase 9 levels in bone marrow and peripheral blood of patients mobilized by granulocyte colony-stimulating factor and chemotherapy. Relationship with mobilizing capacity of haematopoietic progenitor cells. Br J Haematol. 2003; 122:918–926. PMID: 12956762.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Homing-Associated Cell Adhesion Molecules and Cell Cycle Status on the Nucleated Cells in the Bone Marrow, Mobilized Peripheral Blood and Cord Blood
- Modulation of CD44/CD31 Expression and Apoptotic Status of Mobilized CD34+ Cells during Continuous Intravenous Administration of Granulocyte-Colony Stimulating Factor in Normal Donors
- Regulation of Adhesion Molecule Expression and Stromal Cell-Derived Factor-1 Production in Human Bone Marrow Cells by Interferon-gamma, Tumor Necrosis Factor-alpha, and Transforming Growth Factor-beta1: Implications in Bone Marrow Homing of Hematopoietic Cells
- The Frequency of CD34 and CD34 CD38 Hematopoietic Stem/Progenitor Cells in the Cord Blood and Adult Peripheral Blood
- Proportions of Cells Expressing CD38-/CD34+, CD38+/CD34+, CD19+/CD34+, or CD13,33+/CD34+ in the Regenerating Bone Marrows During Complete Remission of Acute Leukemia or After Bone Marrow Transplantation