J Vet Sci.
2016 Sep;17(3):413-419. 10.4142/jvs.2016.17.3.413.
Establishment of hydrochloric acid/lipopolysaccharide-induced pelvic inflammatory disease model
- Affiliations
-
- 1College of Veterinary Medicine & Institute of Veterinary Science, Kangwon National University, Chuncheon 24341, Korea. bdjung@kangwon.ac.kr
- 2College of Pharmacy, Kangwon National University, Chuncheon 24341, Korea.
- 3Animal Reproduction & Biotechnology Center, Myung-Poom Hanwoo Consulting, Hoengseong 25218, Korea.
- 4Department of Biotechnology, College of Engineering, Daegu University, Gyeongsan 38453, Korea.
- 5Department of Biomedical Laboratory Science, College of Health Sciences, Yonsei University, Wonju 26493, Korea.
- KMID: 2413143
- DOI: http://doi.org/10.4142/jvs.2016.17.3.413
Abstract
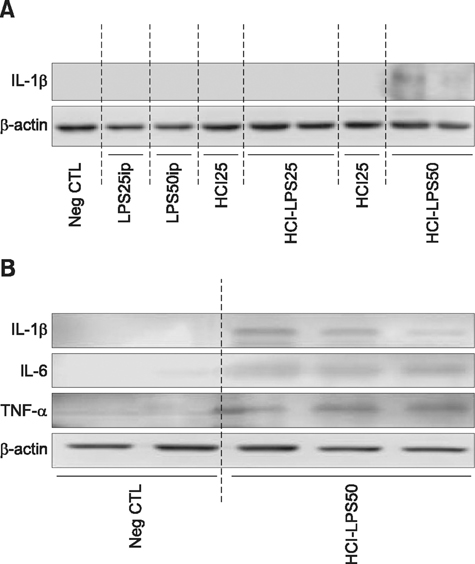
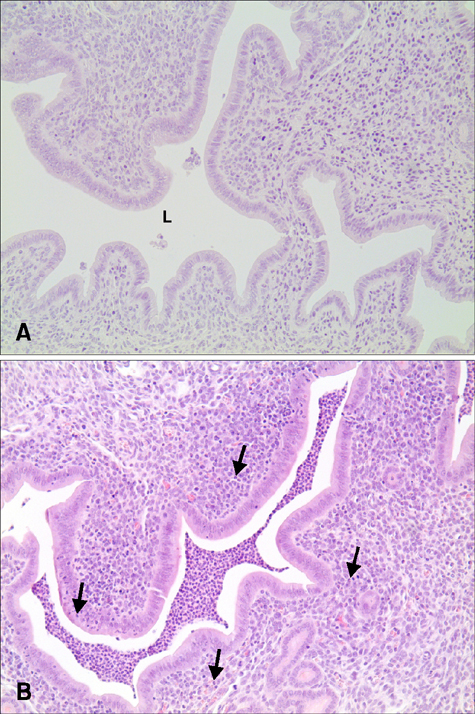
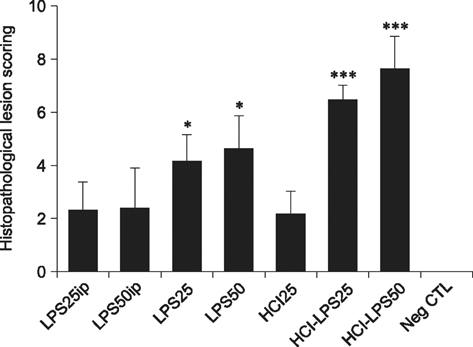
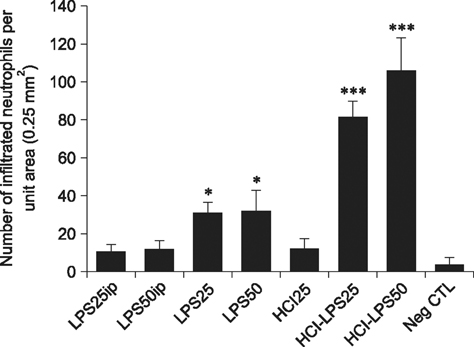
- Pelvic inflammatory disease (PID), which is one of the most problematic complications experienced by women with sexually transmitted diseases, frequently causes secondary infections after reproductive abnormalities in veterinary animals. Although the uterus is self-protective, it becomes fragile during periods or pregnancy. To investigate PID, bacteria or lipopolysaccharide (LPS) extracted from gram negative bacteria has been used to induce the disease in several animal models. However, when LPS is applied to the peritoneum, it often causes systemic sepsis leading to death and the PID was not consistently demonstrated. Hydrochloric acid (HCl) has been used to induce inflammation in the lungs and stomach but not tested for reproductive organs. In this study, we developed a PID model in mice by HCl and LPS sequential intracervical (i.c.) administration. The proinflammatory cytokines, interleukin (IL)-1β, IL-6 and tumor necrosis factor-α, were detected in the mouse uterus by western blot analysis and cytokine enzyme-linked immunosorbent assay after HCl (25 mg/kg) administration i.c. followed by four LPS (50 mg/kg) treatments. Moreover, mice exhibited increased infiltration of neutrophils in the endometrium and epithelial layer. These results suggest that ic co-administration of HCl and LPS induces PID in mice. This new model may provide a consistent and reproducible PID model for future research.
Keyword
MeSH Terms
-
Animals
Blotting, Western/veterinary
Cytokines/*metabolism
*Disease Models, Animal
Female
Humans
*Hydrochloric Acid
Inflammation/chemically induced/*veterinary
*Lipopolysaccharides
*Mice
Mice, Inbred C57BL
Pelvic Inflammatory Disease/*chemically induced
Uterus/immunology/pathology
Cytokines
Lipopolysaccharides
Hydrochloric Acid
Figure
Reference
-
1. Abdullah FFJ, Adamu L, Ismael NA, Osman AY, Haron AW, Saad MZ, Saharee AA. Clinical responses and reproductive pathological changes associated with Brucella melitensis and it's lipopolysaccharides in female mice. Int J Anim Vet Adv. 2014; 6:15–22.
Article2. Ando H, Takamura T, Ota T, Nagai Y, Kobayashi K. Cerivastatin improves survival of mice with lipopolysaccharide-induced sepsis. J Pharmacol Exp Ther. 2000; 294:1043–1046.3. Aronoff DM, Hao Y, Chung J, Coleman N, Lewis C, Peres CM, Serezani CH, Chen GH, Flamand N, Brock TG, Peters-Golden M. Misoprostol impairs female reproductive tract innate immunity against Clostridium sordellii. J Immunol. 2008; 180:8222–8230.
Article4. Bernstein-Hanley I, Coers J, Balsara ZR, Taylor GA, Starnbach MN, Dietrich WF. The p47 GTPases Igtp and Irgb10 map to the Chlamydia trachomatis susceptibility locus Ctrq-3 and mediate cellular resistance in mice. Proc Natl Acad Sci U S A. 2006; 103:14092–14097.
Article5. Brecchia G, Menchetti L, Cardinali R, Castellini C, Polisca A, Zerani M, Maranesi M, Boiti C. Effects of a bacterial lipopolysaccharide on the reproductive functions of rabbit does. Anim Reprod Sci. 2014; 147:128–134.
Article6. Chen G, Li J, Qing X, Czura CJ, Ochani M, Ochani K, Ulloa L, Yang H, Tracey KJ, Wang P, Sama AE, Wang H. Suppression of HMGB1 release by stearoyl lysophosphatidylcholine: an additional mechanism for its therapeutic effects in experimental sepsis. J Lipid Res. 2005; 46:623–627.
Article7. Deb K, Chaturvedi MM, Jaiswal YK. Gram-negative bacterial LPS induced poor uterine receptivity and implantation failure in mouse: alterations in IL-1β expression in the preimplantation embryo and uterine horns. Infect Dis Obstet Gynecol. 2005; 13:125–133.
Article8. Fahey JV, Wright JA, Shen L, Smith JM, Ghosh M, Rossoll RM, Wira CR. Estradiol selectively regulates innate immune function by polarized human uterine epithelial cells in culture. Nucosal Immunol. 2008; 1:317–325.
Article9. Fang L, Nowicki BJ, Dong YL, Yallampalli C. Localized increase in nitric oxide production and the expression of nitric oxide synthase isoforms in rat uterus with experimental intrauterine infection. Am J Obstet Gynecol. 1999; 181:601–609.
Article10. Jiang Y, Wang MH. Ethanol extract of Synurus deltoids (Aiton) Nakai suppresses in vitro LPS-induced cytokine production in RAW 264.7 macrophages and in vivo acute inflammatory symptoms. Nutr Res Pract. 2014; 8:11–19.
Article11. Laven RA, Peters AR. Bovine retained placenta: aetiology, pathogenesis and economic loss. Vet Rec. 1996; 139:465–471.
Article12. Luo J, Xu Y, Zhang M, Gao L, Fang C, Zhou C. Magnolol inhibits LPS-induced inflammatory response in uterine epithelial cells. Inflammation. 2013; 36:997–1003.
Article13. Moore CC, Martin EN, Lee G, Taylor C, Dondero R, Reznikov LL, Dinarello C, Thompson J, Scheld WM. Eukaryotic translation initiation factor 5A small interference RNA-liposome complexes reduce inflammation and increase survival in murine models of severe sepsis and acute lung injury. J Infect Dis. 2008; 198:1407–1414.
Article14. Nylander O, Holm L, Wilander E, Hällgren A. Exposure of the duodenum to high concentrations of hydrochloric acid: effects on mucosal permeability, alkaline secretion, and blood flow. Scand J Gastroenterol. 1994; 29:437–444.
Article15. Ross JDC. An update on pelvic inflammatory disease. Sex Transm Infect. 2002; 78:18–19.
Article16. Schlafer DH, Miller RB. Female genital system. In : Maxie MG, editor. Jubb, Kennedy, and Palmer's Pathology of Domestic Animals. 5th ed. London: Elsevier;2007. p. 466–474.17. Sheldon IM, Lewis GS, LeBlanc S, Gilbert RO. Defining postpartum uterine disease in cattle. Theriogenology. 2006; 65:1516–1530.
Article18. Sheldon IM, Rycroft AN, Dogan B, Craven M, Bromfield JJ, Chandler A, Roberts MH, Price SB, Gilbert RO, Simpson KW. Specific strains of Escherichia coli are pathogenic for the endometrium of cattle and cause pelvic inflammatory disease in cattle and mice. PLoS One. 2010; 5:e9192.19. Yamashiro O, Morimoto A, Sakata Y, Watanabe T, Murakami N. Febrile and metabolic tolerance to endotoxin and human recombinant interleukin-1 beta in rabbits. Am J Physiol. 1993; 264:R1180–R1185.
Article20. Yu PJ, Li JR, Zhu ZG, Kong HY, Jin H, Zhang JY, Tian YX, Li ZH, Wu XY, Zhang JJ, Wu SG. Praeruptorin D and E attenuate lipopolysaccharide/hydrochloric acid induced acute lung injury in mice. Eur J Pharmacol. 2013; 710:39–48.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- White Esophageal Mucosa and Black Gastric Mucosa: Upper Gastrointestinal Injury Due to Hydrochloric Acid Ingestion
- A Case of Corrosive Gastritis Caused by Hydrochloric Acid
- The Effect of Beta-Ray on the Course of Experimental Acid Burn of Rabbit's Cornea
- A Case of Hydrochloric Acid Induced Hyperkalemia
- The Effects of Acute Variations in Plasma pH and Pco2 on Renal Bicarbonate Reabsorption in the Dog