J Vet Sci.
2017 Mar;18(1):81-88. 10.4142/jvs.2017.18.1.81.
Alginate encapsulation preserves the quality and fertilizing ability of Mediterranean Italian water buffalo (Bubalus bubalis) and Holstein Friesian (Bos taurus) spermatozoa after cryopreservation
- Affiliations
-
- 1Department of Drug Sciences, University of Pavia, 27100 Pavia, Italy. theodora.chlapanidas@unipv.it
- 2Italian Experimental Institute Lazzaro Spallanzani, 26027 Rivolta D'Adda, Italy.
- 3Department of Veterinary Science and Public Health, University of Milan, 20133 Milan, Italy.
- KMID: 2412591
- DOI: http://doi.org/10.4142/jvs.2017.18.1.81
Abstract
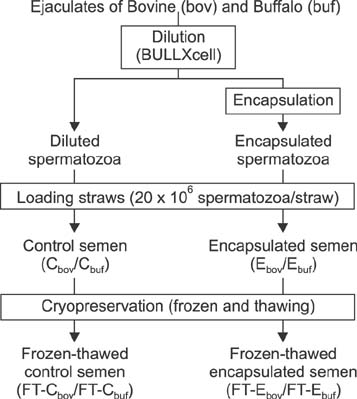
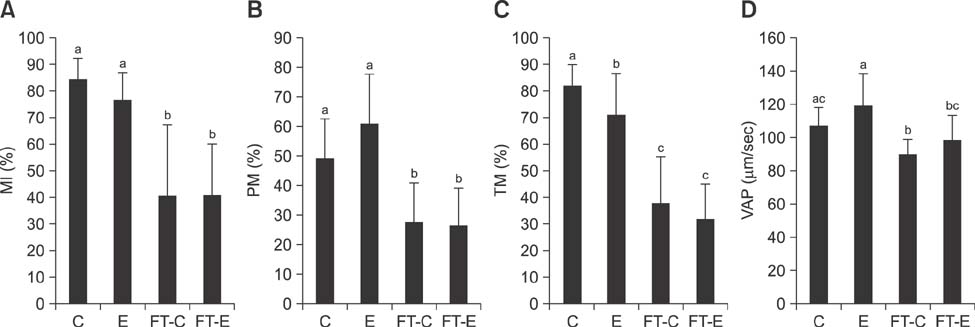
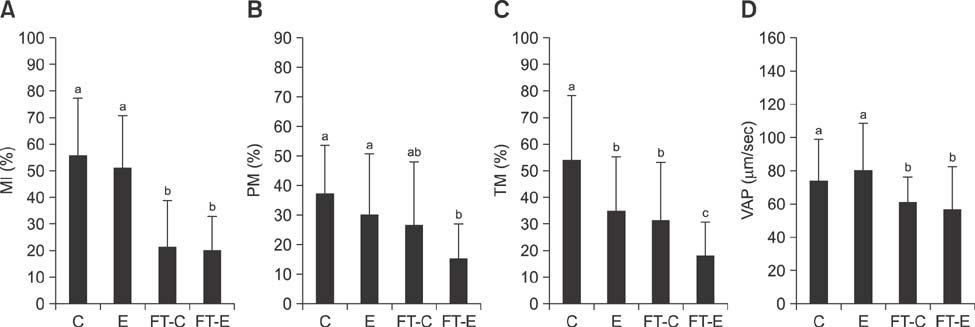
- The use of artificial insemination (AI) in buffalo (Bubalus bubalis) is limited by poor ovarian activity during the hot season, seasonal qualitative patterns in semen, low resistance of sperm cells in the female tract, difficulties in estrus detection, and variable estrus duration. Although AI procedures are commonly used in bovine, use of AI has been limited in buffalo. In the zootechnical field, different studies have been conducted to develop techniques for improvement of fertilizing ability of buffalo spermatozoa after AI. In this study, for the first time, the use of alginate encapsulation and cryopreservation of buffalo spermatozoa is described, and the same procedure was performed with Holstein Friesian (Bos taurus) semen. Results obtained from in vitro analyses indicate that the encapsulation process does not have detrimental effects (compared to controls) on quality parameters (membrane integrity, progressive motility, path average velocity) in either species. Similarly, there were no detrimental effects after cryopreservation in either species. The fertilizing potential of encapsulated and cryopreserved semen was evaluated after AI in 25 buffalo and 113 bovine females. Pregnancy rates were not affected in either species. The results of this study show proof of concept for the use of frozen semen controlled-release devices in buffalo.
Keyword
MeSH Terms
-
Alginates/*pharmacology
Animals
Biocompatible Materials/*pharmacology
Buffaloes/*physiology
Cattle
Cryopreservation/veterinary
Female
Glucuronic Acid/pharmacology
Hexuronic Acids/pharmacology
Insemination, Artificial/*veterinary
Italy
Male
Pregnancy
Pregnancy Rate
Semen Preservation/*veterinary
Spermatozoa/*chemistry
Alginates
Biocompatible Materials
Hexuronic Acids
Glucuronic Acid
Figure
Reference
-
1. Amiridis GS, Cseh S. Assisted reproductive technologies in the reproductive management of small ruminants. Anim Reprod Sci. 2012; 130:152–161.
Article2. Bahga CS, Khokar BS. Effect of different seasons on concentration of plasma luteinizing hormone and seminal quality vis-à-vis freezability of buffalo bulls (Bubalus bubalis). Int J Biometeorol. 1991; 35:222–224.
Article3. Ballester J, Johannisson A, Saravia F, Håård M, Gustafsson H, Bajramovic D, Rodriguez-Martinez H. Post-thaw viability of bull AI-doses with low-sperm numbers. Theriogenology. 2007; 68:934–943.
Article4. Baruselli PS, Mucciolo RG, Visintin JA, Viana WG, Arruda RP, Madureira EH, Oiveira CA, Molero-Filho JR. Ovarian follicular dynamics during the estrous cycle in buffalo (Bubalus bubalis). Theriogenology. 1997; 47:1531–1547.
Article5. De Rensis F, López-Gatius F. Protocols for synchronizing estrus and ovulation in buffalo (Bubalus bubalis): a review. Theriogenology. 2007; 67:209–216.
Article6. De Rensis F, Ronci G, Guarneri P, Nguyen BX, Presicce GA, Huszenicza G, Scaramuzzi RJ. Conception rate after fixed time insemination following ovsynch protocol with and without progesterone supplementation in cyclic and noncyclic Mediterranean Italian buffaloes (Bubalus bubalis). Theriogenology. 2005; 63:1824–1831.
Article7. Faustini M, Bucco M, Galeati G, Spinaci M, Villani S, Chlapanidas T, Ghidoni I, Vigo D, Torre ML. Boar sperm encapsulation reduces in vitro polyspermy. Reprod Domest Anim. 2010; 45:359–362.8. Faustini M, Vigo D, Spinaci M, Galeati G, Torre ML. Enhancing insemination performance in pigs through controlled release of encapsulated spermatozoa. Reprod Domest Anim. 2012; 47:Suppl 4. 353–358.
Article9. Forero-Gonzalez RA, Celeghini ECC, Raphael CF, Andrade AFC, Bressan FF, Arruda RP. Effects of bovine sperm cryopreservation using different freezing techniques and cryoprotective agents on plasma, acrosomal and mitochondrial membranes. Andrologia. 2012; 44:Suppl 1. 154–159.
Article10. Garner DL, Seidel GE Jr. History of commercializing sexed semen for cattle. Theriogenology. 2008; 69:886–895.
Article11. Gaviraghi A, Puglisi R, Balduzzi D, Severgnini A, Bornaghi V, Bongioni G, Frana A, Gandini LM, Lukaj A, Bonacina C, Galli A. Minimum number of spermatozoa per dose in Mediterranean Italian buffalo (Bubalus bubalis) using sexed frozen semen and conventional artificial insemination. Theriogenology. 2013; 79:1171–1176.
Article12. Herrler A, Eisner S, Bach V, Weissenborn U, Beier HM. Cryopreservation of spermatozoa in alginic acid capsules. Fertil Steril. 2006; 85:208–213.
Article13. Kemmer C, Fluri DA, Witschi U, Passeraub A, Gutzwiller A, Fussenegger M. A designer network coordinating bovine artificial insemination by ovulation-triggered release of implanted sperms. J Control Release. 2011; 150:23–29.
Article14. Kumar R, Atreja SK. Effect of incorporation of additives in tris-based egg yolk extender on buffalo (Bubalus bubalis) sperm tyrosine phosphorylation during cryopreservation. Reprod Domest Anim. 2012; 47:485–490.
Article15. Leite TG, do Vale Filho VR, de Arruda RP, de Andrade AFC, Emerick LL, Zaffalon FG, Martins JAM, de Andrade VJ. Effects of extender and equilibration time on post-thaw motility and membrane integrity of cryopreserved Gyr bull semen evaluated by CASA and flow cytometry. Anim Reprod Sci. 2010; 120:31–38.
Article16. Minervini F, Guastamacchia R, Pizzi F, Dell’Aquila ME, Barile VL. Assessment of different functional parameters of frozen-thawed buffalo spermatozoa by using cytofluorimetric determinations. Reprod Domest Anim. 2013; 48:317–324.
Article17. Monteiro BM, de Souza DC, Vasconcellos GSFM, Corrêa TB, Vecchio D, de Sá MF, de Carvalho NAT, Baruselli PS. Ovarian responses of dairy buffalo cows to timed artificial insemination protocol, using new or used progesterone devices, during the breeding season (autumn-winter). Anim Sci J. 2016; 87:13–20.
Article18. Nebel RL, Bame JH, Saacke RG, Lim F. Microencapsulation of bovine spermatozoa. J Anim Sci. 1985; 60:1631–1639.
Article19. Nebel RL, Vishwanath R, McMillan WH, Saacke RG. Microencapsulation of bovine spermatozoa for use in artificial insemination: a review. Reprod Fertil Dev. 1993; 5:701–712.
Article20. Orgal S, Zeron Y, Elior N, Biran D, Friedman E, Druker S, Roth Z. Season-induced changes in bovine sperm motility following a freeze-thaw procedure. J Reprod Dev. 2012; 58:212–218.
Article21. Perera BMAO. Reproduction in domestic buffalo. Reprod Domest Anim. 2008; 43:Suppl 2. 200–206.
Article22. Perera BMAO. Reproductive cycles of buffalo. Anim Reprod Sci. 2011; 124:194–199.
Article23. Perteghella S, Vigani V, Crivelli B, Spinaci M, Galeati G, Bucci D, Vigo D, Torre ML, Chlapanidas T. Sperm encapsulation from 1985 to date: technology evolution and new challenges in swine reproduction. Reprod Domest Anim. 2015; 50:Suppl 2. 98–102.
Article24. Rodriguez-Martinez H. Assisted reproductive techniques for cattle breeding in developing countries: a critical appraisal of their value and limitations. Reprod Domest Anim. 2012; 47:Suppl 1. 21–26.
Article25. Seidel GE Jr, Schenk JL. Pregnancy rates in cattle with cryopreserved sexed sperm: effects of sperm numbers per inseminate and site of sperm deposition. Anim Reprod Sci. 2008; 105:129–138.
Article26. Shah S, Otsuki T, Fujimura C, Yamamoto N, Yamashita Y, Higaki S, Hishinuma M. Cryopreservation of microencapsulated canine sperm. Theriogenology. 2011; 75:679–686.
Article27. Silva PFN, Gadella BM. Detection of damage in mammalian sperm cells. Theriogenology. 2006; 65:958–978.
Article28. Singh J, Nanda AS, Adams GP. The reproductive pattern and efficiency of female buffaloes. Anim Reprod Sci. 2000; 60-61:593–604.29. Spinaci M, Bucci D, Chlapanidas T, Vallorani C, Perteghella S, Communod R, Vigo D, Tamanini C, Galeati G, Faustini M, Torre ML. Boar sperm changes after sorting and encapsulation in barium alginate membranes. Theriogenology. 2013; 80:526–532.
Article30. Spinaci M, Chlapanidas T, Bucci D, Vallorani C, Perteghella S, Lucconi G, Communod R, Vigo D, Galeati G, Faustini M, Torre ML. Encapsulation of sex sorted boar semen: sperm membrane status and oocyte penetration parameters. Theriogenology. 2013; 79:575–581.
Article31. Spinaci M, Perteghella S, Chlapanidas T, Galeati G, Vigo D, Tamanini C, Bucci D. Storage of sexed boar spermatozoa: limits and perspectives. Theriogenology. 2016; 85:65–73.
Article32. Standerholen FB, Waterhouse KE, Larsgard AG, Garmo RT, Myromslien FD, Sunde J, Ropstad E, Klinkenberg G, Kommisrud E. Use of immobilized cryopreserved bovine semen in a blind artificial insemination trial. Theriogenology. 2015; 84:413–420.
Article33. Vigo D, Faustini M, Torre ML, Pecile A, Villani S, Asti A, Norbert R, Maggi L, Conte U, Cremonesi F, Stacchezzini S, Maffeo G. Boar semen controlled-delivery system: morphological investigation and in vitro fertilization test. Reprod Fertil Dev. 2002; 14:307–314.
Article34. Vigo D, Faustini M, Villani S, Orsini F, Bucco M, Chlapanidas T, Conte U, Ellis K, Torre ML. Semen controlled-release capsules allow a single artificial insemination in sows. Theriogenology. 2009; 72:439–444.
Article35. Vishwanath R, Nebel RL, McMillan WH, Pitt CJ, Macmillan KL. Selected times of insemination with microencapsulated bovine spermatozoa affect pregnancy rates of synchronized heifers. Theriogenology. 1997; 48:369–376.
Article36. Weber W, Rimann M, Schafroth T, Witschi U, Fussenegger M. Design of high-throughput-compatible protocols for microencapsulation, cryopreservation and release of bovine spermatozoa. J Biotechnol. 2006; 123:155–163.
Article37. World Health Organization. Semen analysis: standard procedures. WHO laboratory manual for the examination and processing of human semen. 5th ed. Geneva: WHO Press;2010. p. 7–115. Chapt. 2.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The use of crossreactive monoclonal antibodies to characterize the immune system of the water buffalo (Bubalus bubalis)
- Estimation of Damage to Human Spermatozoa During Cryopreservation by Measurement of Release of Glutamic-Oxalacetic Transaminase
- High level of estradiol in a Holstein-Friesian cow with sacroiliac luxation after calving
- An outbreak of neonatal enteritis in buffalo calves associated with astrovirus
- Efficacy and Fertilizing Ability of Frozen-thawed Testicular Spermatozoa and Spermatozoa Extracted from the Seminiferous Tubule with Intracytoplasmic Sperm Injection (ICSI)