Cancer Res Treat.
2018 Apr;50(2):538-550. 10.4143/crt.2017.106.
Declined Preoperative Aspartate Aminotransferase to Neutrophil Ratio Index Predicts Poor Prognosis in Patients with Intrahepatic Cholangiocarcinoma after Hepatectomy
- Affiliations
-
- 1Department of Hepatic Surgery, The First Affiliated Hospital of Sun Yat-sen University, Guangzhou, China. pengbaogang@medmail.com.cn, shunlishen@163.com
- 2Department of Medical Ultrasonics, Institute of Diagnostic and Interventional Ultrasound, The First Affiliated Hospital of Sun Yat-sen University, Guangzhou, China.
- 3Department of Oncology, The First Affiliated Hospital of Sun Yat-sen University, Guangzhou, China.
- KMID: 2411142
- DOI: http://doi.org/10.4143/crt.2017.106
Abstract
- PURPOSE
Various inflammation-based prognostic biomarkers such as the platelet to lymphocyte ratio and neutrophil to lymphocyte ratio, are related to poor survival in patients with intrahepatic cholangiocarcinoma (ICC). This study aims to investigate the prognostic value of the aspartate aminotransferase to neutrophil ratio index (ANRI) in ICC after hepatic resection.
MATERIALS AND METHODS
Data of 184 patients with ICC after hepatectomy were retrospectively reviewed. The cut-off value of ANRI was determined by a receiver operating characteristic curve. Preoperative ANRI and clinicopathological variables were analyzed. The predictive value of preoperative ANRI for prognosis of ICC was identified by univariate and multivariate analyses.
RESULTS
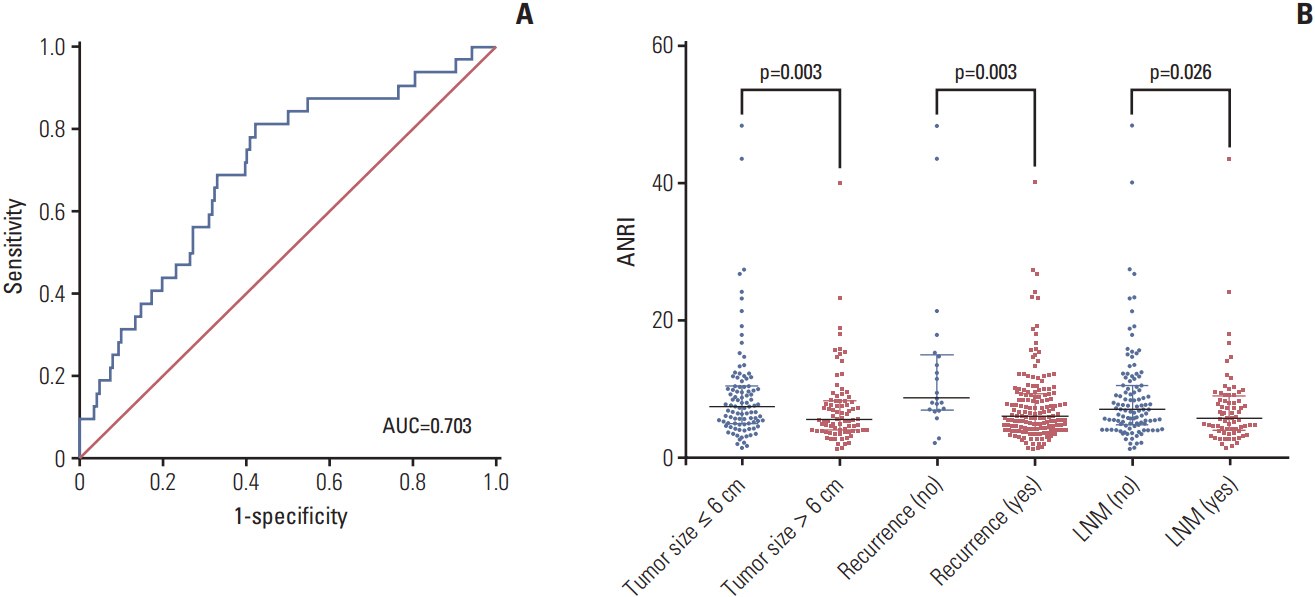
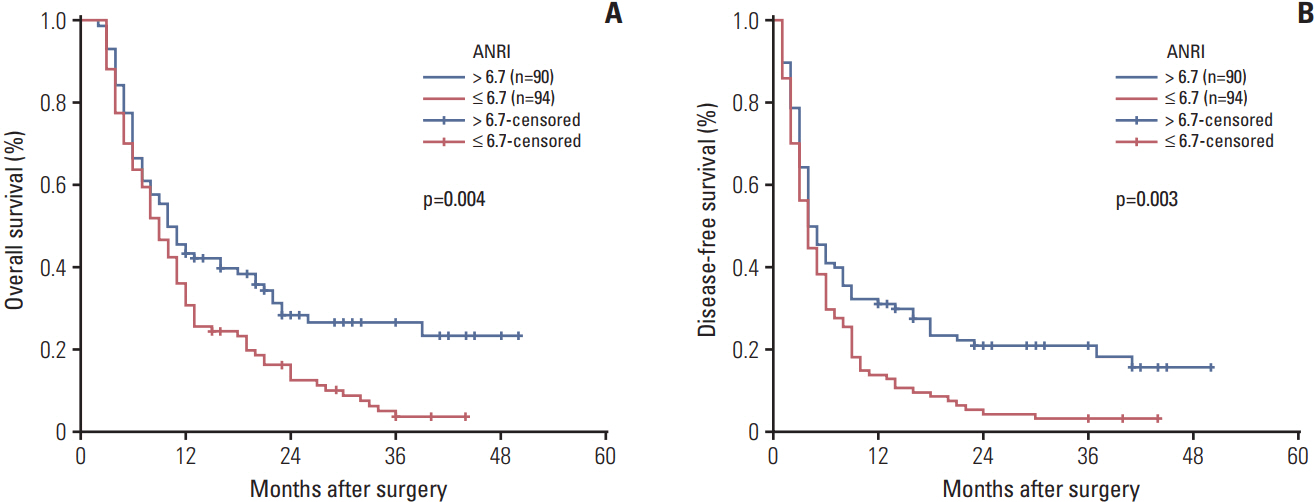
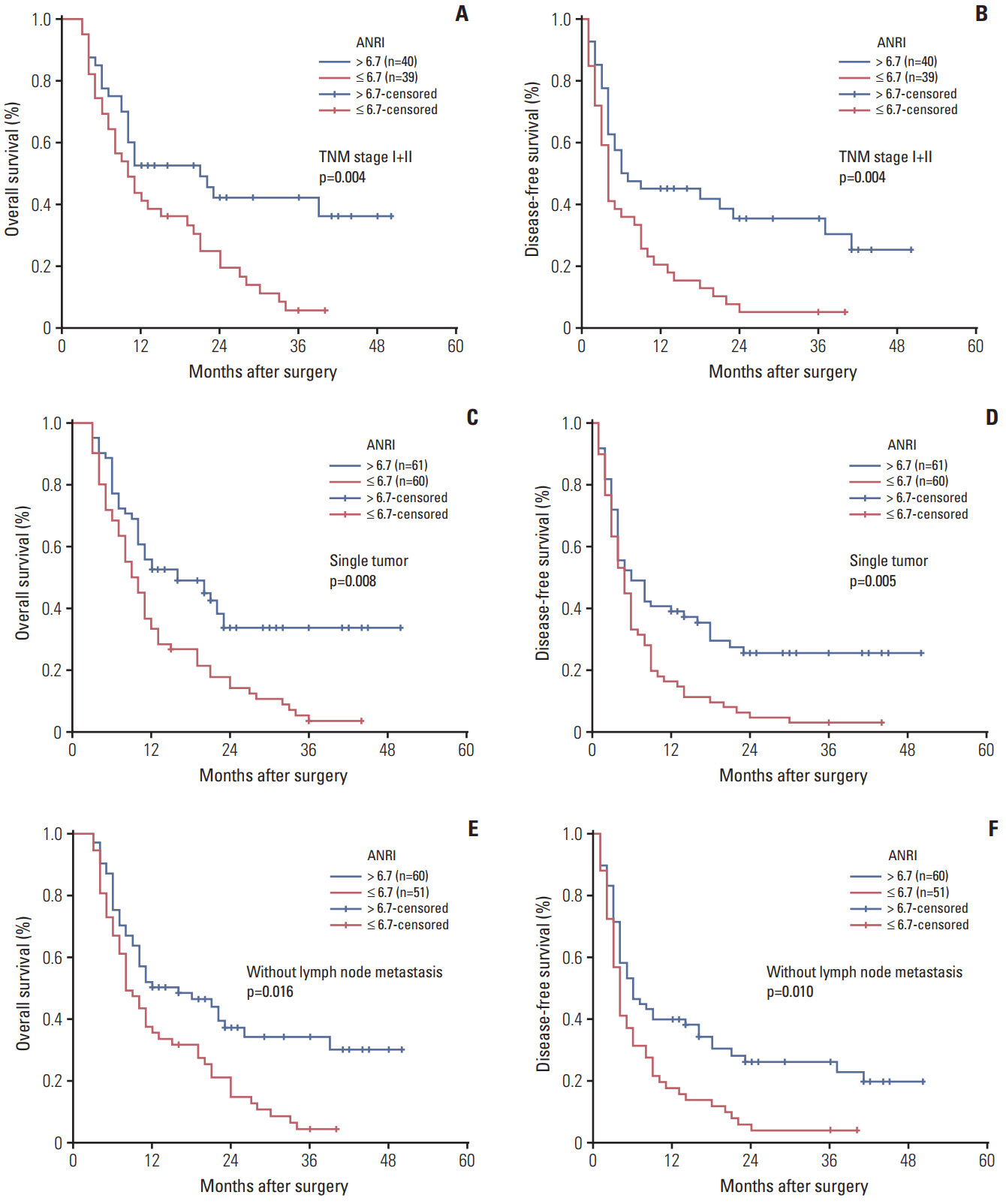
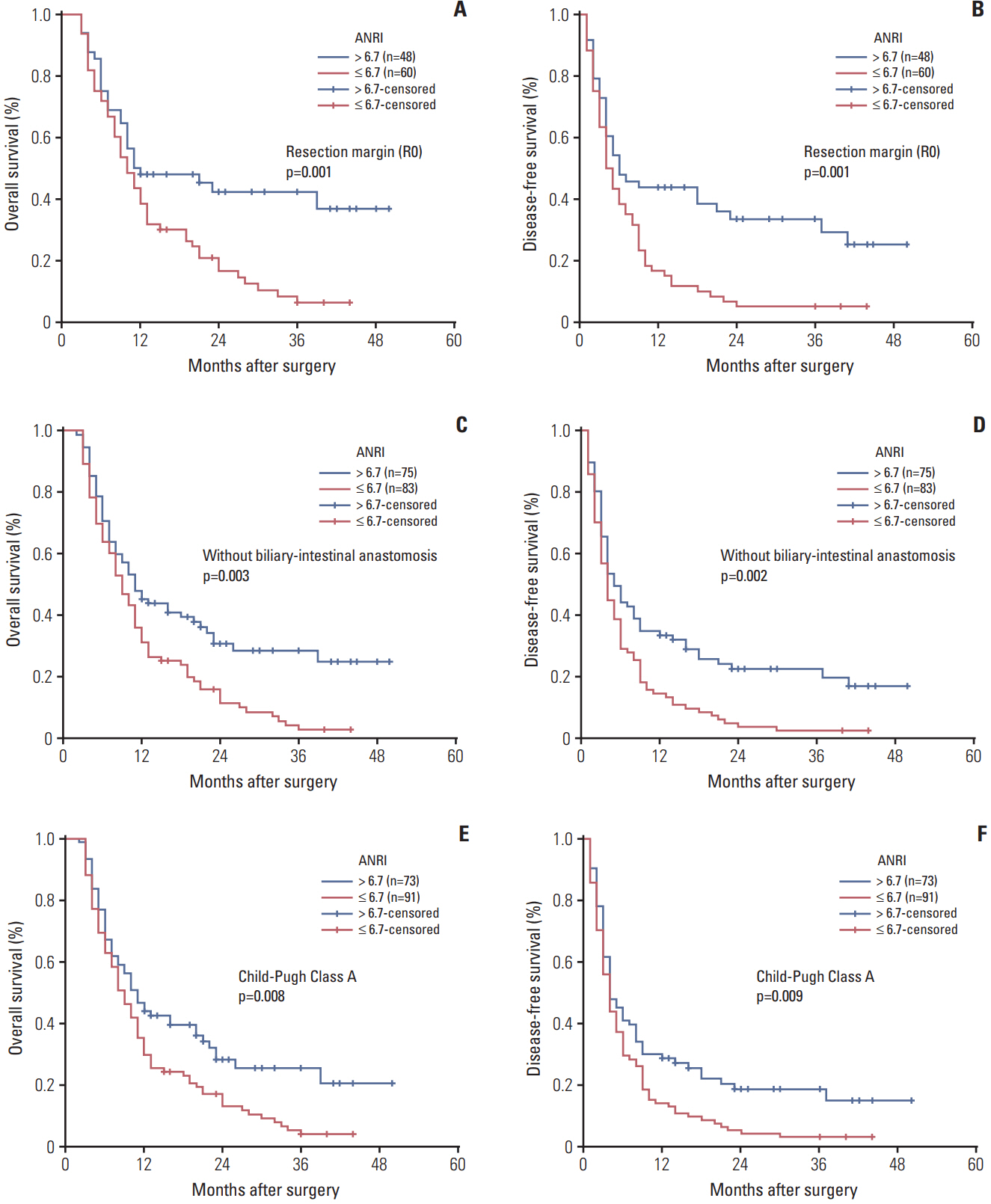
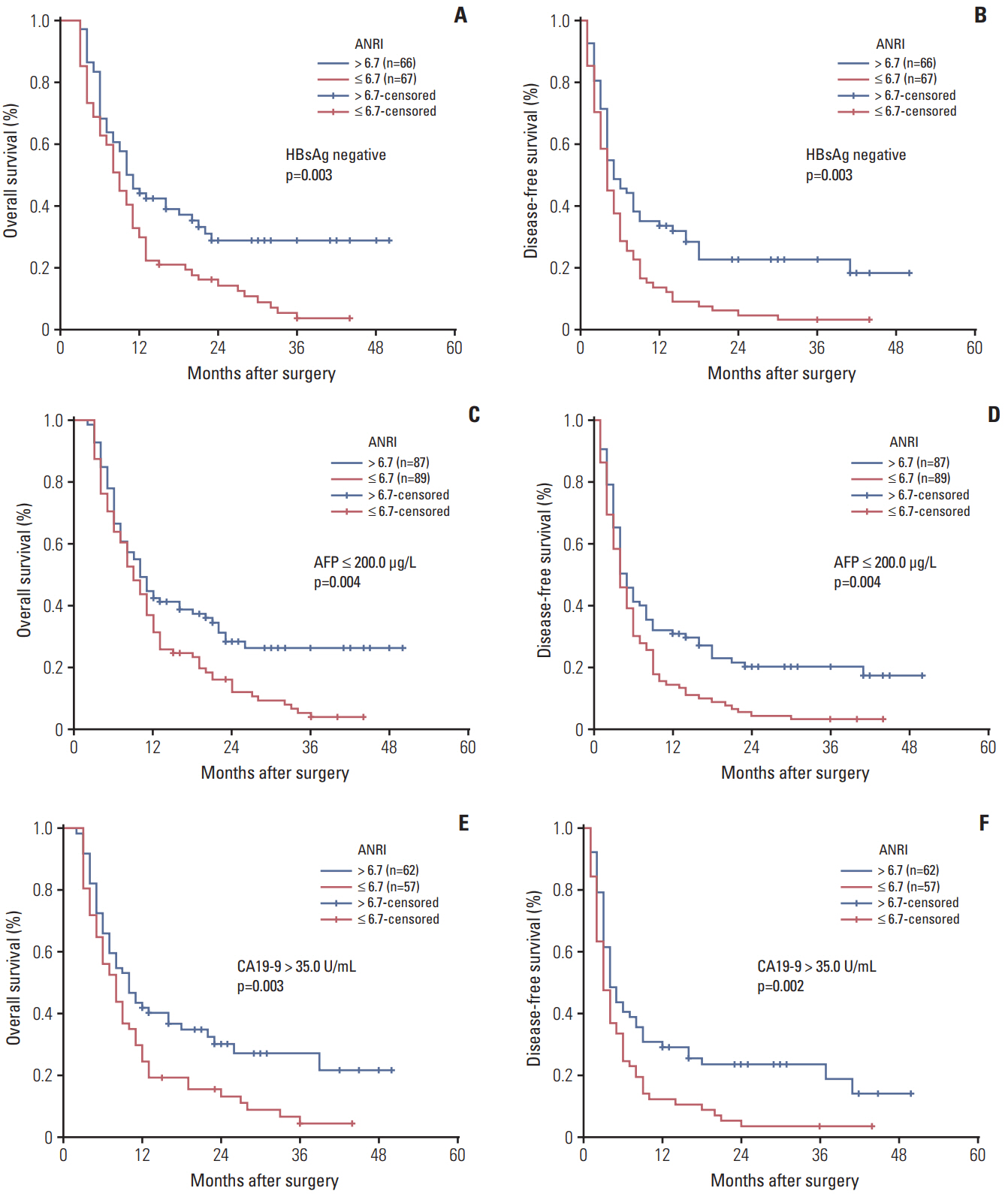
The optimal cut-off value of ANRI was 6.7. ANRI was associated with tumor size, tumor recurrence, white blood cell, neutrophil count, aspartate aminotransferase, and alanine transaminase. Univariate analysis showed that ANRI, sex, tumor number, tumor size, tumor differentiation, lymph node metastasis, resection margin, clinical TNM stage, neutrophil count, and carcinoembryonic antigen were markedly correlated with overall survival (OS) and disease-free survival (DFS) in patients with ICC. Multivariable analyses revealed that ANRI, a tumor size > 6 cm, poor tumor differentiation, and an R1 resection margin were independent prognostic factors for both OS and DFS. Additionally, preoperative ANRI also had a significant value to predict prognosis in various subgroups of ICC, including serum hepatitis B surface antigen"’negative and preoperative elevated carbohydrate antigen 19-9 patients.
CONCLUSION
Preoperative declined ANRI is a noninvasive, simple, and effective predictor of poor prognosis in patients with ICC after hepatectomy.
MeSH Terms
-
Alanine Transaminase
Aspartate Aminotransferases*
Aspartic Acid*
Biomarkers
Blood Platelets
Carcinoembryonic Antigen
Cholangiocarcinoma*
Disease-Free Survival
Hepatectomy*
Hepatitis B
Humans
Leukocytes
Lymph Nodes
Lymphocytes
Multivariate Analysis
Neoplasm Metastasis
Neutrophils*
Prognosis*
Recurrence
Retrospective Studies
ROC Curve
Alanine Transaminase
Aspartate Aminotransferases
Aspartic Acid
Biomarkers
Carcinoembryonic Antigen
Figure
Reference
-
References
1. Moeini A, Sia D, Bardeesy N, Mazzaferro V, Llovet JM. Molecular pathogenesis and targeted therapies for intrahepatic cholangiocarcinoma. Clin Cancer Res. 2016; 22:291–300.
Article2. Bridgewater J, Galle PR, Khan SA, Llovet JM, Park JW, Patel T, et al. Guidelines for the diagnosis and management of intrahepatic cholangiocarcinoma. J Hepatol. 2014; 60:1268–89.
Article3. Jutric Z, Johnston WC, Hoen HM, Newell PH, Cassera MA, Hammill CW, et al. Impact of lymph node status in patients with intrahepatic cholangiocarcinoma treated by major hepatectomy: a review of the National Cancer Database. HPB (Oxford). 2016; 18:79–87.4. Spolverato G, Kim Y, Alexandrescu S, Marques HP, Lamelas J, Aldrighetti L, et al. Management and outcomes of patients with recurrent intrahepatic cholangiocarcinoma following previous curative-intent surgical resection. Ann Surg Oncol. 2016; 23:235–43.
Article5. Poultsides GA, Zhu AX, Choti MA, Pawlik TM. Intrahepatic cholangiocarcinoma. Surg Clin North Am. 2010; 90:817–37.
Article6. Kim HC, Nam CM, Jee SH, Han KH, Oh DK, Suh I. Normal serum aminotransferase concentration and risk of mortality from liver diseases: prospective cohort study. BMJ. 2004; 328:983.
Article7. Fu SJ, Shen SL, Li SQ, Hua YP, Hu WJ, Liang LJ, et al. Prognostic value of preoperative peripheral neutrophil-to-lymphocyte ratio in patients with HBV-associated hepatocellular carcinoma after radical hepatectomy. Med Oncol. 2013; 30:721.
Article8. Jin J, Zhu P, Liao Y, Li J, Liao W, He S. Elevated preoperative aspartate aminotransferase to lymphocyte ratio index as an independent prognostic factor for patients with hepatocellular carcinoma after hepatic resection. Oncotarget. 2015; 6:19217–27.
Article9. Ji F, Fu S, Guo Z, Pang H, Chen D, Wang X, et al. Prognostic significance of preoperative aspartate aminotransferase to neutrophil ratio index in patients with hepatocellular carcinoma after hepatic resection. Oncotarget. 2016; 7:72276–89.
Article10. Bosman FT, Carneiro F, Hruban RH, Theise ND. WHO classification of tumours of the digestive system. 4th ed. Lyon: IARC Press;2010.11. Mantovani A, Allavena P, Sica A, Balkwill F. Cancer-related inflammation. Nature. 2008; 454:436–44.
Article12. Nakashima H, Matsuoka Y, Yoshida R, Nagata M, Hirosue A, Kawahara K, et al. Pre-treatment neutrophil to lymphocyte ratio predicts the chemoradiotherapy outcome and survival in patients with oral squamous cell carcinoma: a retrospective study. BMC Cancer. 2016; 16:41.
Article13. Park HS, Lee HS, Park JS, Park JS, Lee DK, Lee SJ, et al. Prognostic scoring index for patients with metastatic pancreatic adenocarcinoma. Cancer Res Treat. 2016; 48:1253–63.
Article14. Zhang L, Cai JQ, Zhao JJ, Bi XY, Tan XG, Yan T, et al. Impact of hepatitis B virus infection on outcome following resection for intrahepatic cholangiocarcinoma. J Surg Oncol. 2010; 101:233–8.
Article15. Wislez M, Rabbe N, Marchal J, Milleron B, Crestani B, Mayaud C, et al. Hepatocyte growth factor production by neutrophils infiltrating bronchioloalveolar subtype pulmonary adenocarcinoma: role in tumor progression and death. Cancer Res. 2003; 63:1405–12.16. Houghton AM, Rzymkiewicz DM, Ji H, Gregory AD, Egea EE, Metz HE, et al. Neutrophil elastase-mediated degradation of IRS-1 accelerates lung tumor growth. Nat Med. 2010; 16:219–23.
Article17. Teramukai S, Kitano T, Kishida Y, Kawahara M, Kubota K, Komuta K, et al. Pretreatment neutrophil count as an independent prognostic factor in advanced non-small-cell lung cancer: an analysis of Japan Multinational Trial Organisation LC00-03. Eur J Cancer. 2009; 45:1950–8.
Article18. Zhou SL, Dai Z, Zhou ZJ, Chen Q, Wang Z, Xiao YS, et al. CXCL5 contributes to tumor metastasis and recurrence of intrahepatic cholangiocarcinoma by recruiting infiltrative intratumoral neutrophils. Carcinogenesis. 2014; 35:597–605.
Article19. Kusumanto YH, Dam WA, Hospers GA, Meijer C, Mulder NH. Platelets and granulocytes, in particular the neutrophils, form important compartments for circulating vascular endothelial growth factor. Angiogenesis. 2003; 6:283–7.
Article20. Peng H, Zhang Q, Li J, Zhang N, Hua Y, Xu L, et al. Apatinib inhibits VEGF signaling and promotes apoptosis in intrahepatic cholangiocarcinoma. Oncotarget. 2016; 7:17220–9.
Article21. Khan SA, Thomas HC, Davidson BR, Taylor-Robinson SD. Cholangiocarcinoma. Lancet. 2005; 366:1303–14.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Robotic-assisted right hepatectomy via anterior approach for intrahepatic cholangiocarcinoma
- Surgical Treatment for Intrahepatic Cholangiocarcinoma
- Fibrosis-4 index, a predictor for prognosis of hepatocellular carcinoma patients after curative hepatectomy even in hepatitis B virus dominant populations
- Prognostic Factors for Intrahepatic Cholangiocarcinoma Treated with Surgical Resection
- Preoperative neutrophil-to-lymphocyte ratio is prognostic for early recurrence after curative intrahepatic cholangiocarcinoma resection