Predictors of Malignancies in Patients with Inconclusive or Negative Results of Endoscopic Ultrasound-guided Fine-needle Aspiration for Solid Pancreatic Masses
- Affiliations
-
- 1Department of Internal Medicine, Chungbuk National University Hospital, Cheongju, Korea. smpark@chungbuk.ac.kr
- 2Department of Internal Medicine, Chungbuk National University College of Medicine, Cheongju, Korea.
- KMID: 2407672
- DOI: http://doi.org/10.4166/kjg.2018.71.3.153
Abstract
- BACKGROUND/AIMS
This study analyzed the diagnostic accuracy of endoscopic ultrasound-guided fine needle aspiration (EUS-FNA) for pancreatic solid masses in patients with or without chronic pancreatitis as well as the clinical parameters relevant to a malignancy when EUS-FNA was negative or inconclusive.
METHODS
A total of 97 patients, who underwent EUS-FNA for solid pancreatic masses over 2 years at a single institution, were evaluated. All patients underwent EUS-FNA for 3-5 passes with 22 or 25 G needles without an on-site cytopathologist. The final diagnosis was obtained by surgery or compatible clinical outcomes for a more than 12 month follow-up. The diagnostic yields in the patients with or without chronic pancreatitis were compared and the histories and laboratory data relevant to pancreatic ductal adenocarcinoma (PDAC) or pseudo-tumor were analyzed.
RESULTS
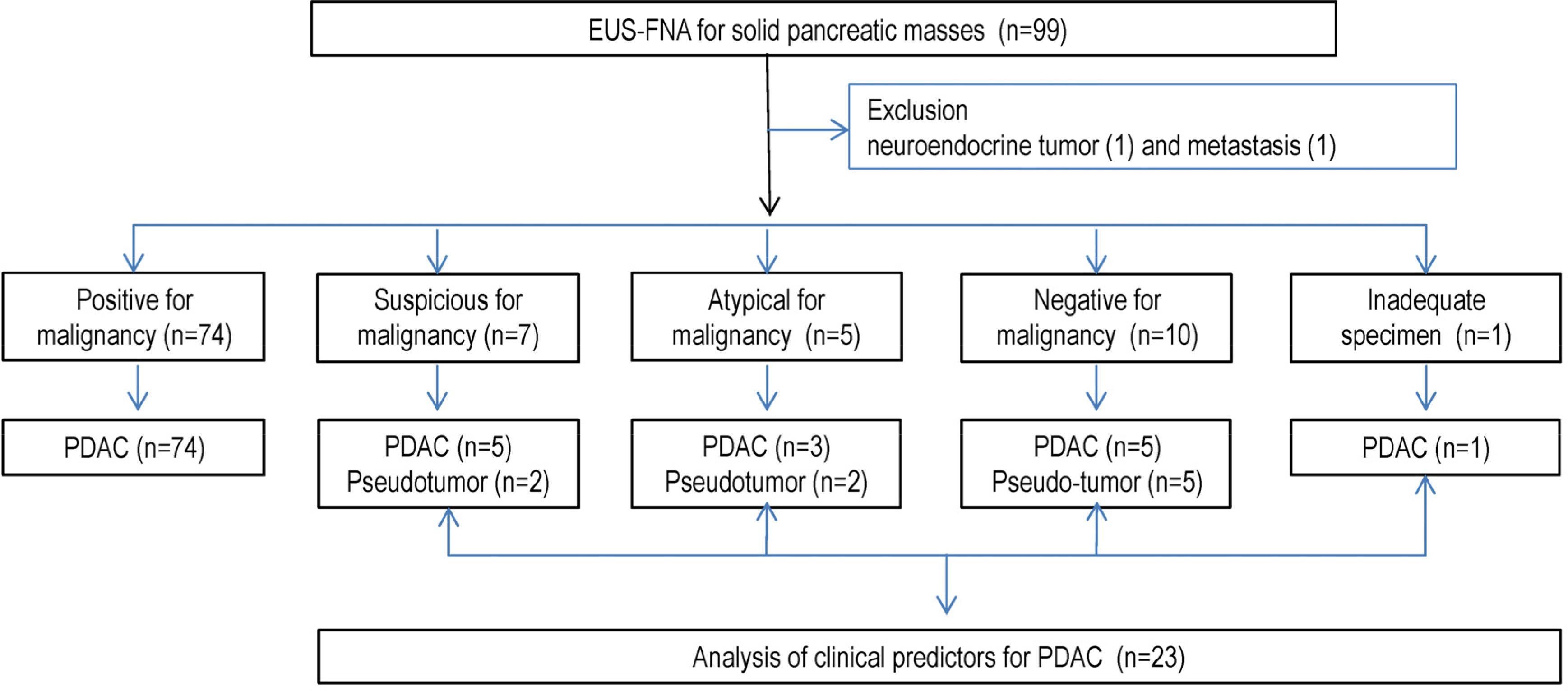
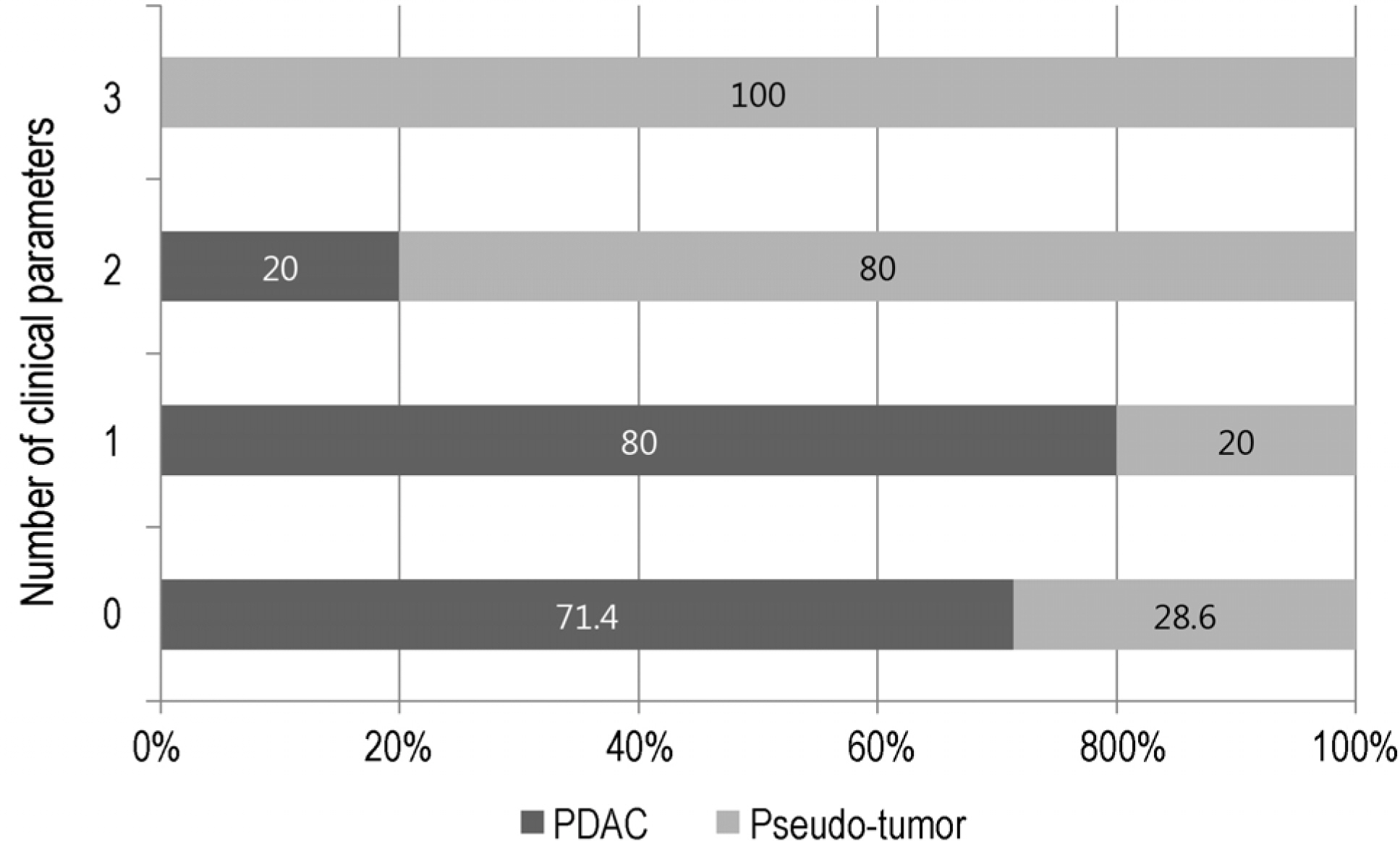
The final diagnoses were adenocarcinoma in 88 patients (90.7%) and inflammatory pseudo-tumor in 9 (9.3%). The results of EUS-FNA were adenocarcinoma (74), suspicious (7), atypical (5), negative (10), and inadequate specimen (1). The diagnostic accuracies were 76.9% and 91.6% in patients with or without chronic pancreatitis, respectively. Among the 23 cases with non-diagnostic results of EUS-FNA, PDAC was finally diagnosed in 5 out of 7 suspicious, 3 out of 5 atypical, and 5 out of 10 negative cytology cases. The clinical parameters related to a pseudo-tumor were a history of alcohol consumption and pancreatitis, and normal alkaline phosphatase levels.
CONCLUSIONS
The diagnostic accuracy of pancreatic masses in the background of chronic pancreatitis was low. When EUS-FNA produced inconclusive results, the histories of alcohol consumption, pancreatitis, and serum levels of alkaline phosphatase are useful for making a final diagnosis.
MeSH Terms
Figure
Cited by 3 articles
-
Differential Diagnosis of Pancreatic Cancer by Endoscopic Ultrasound-guided Fine-needle Aspiration in Solid Mass of Pancreas
Sung Bum Kim
Korean J Gastroenterol. 2018;71(3):113-115. doi: 10.4166/kjg.2018.71.3.113.The Risk Factors for Acute Pancreatitis after Endoscopic Ultrasound Guided Biopsy
Afonso Ribeiro, Akash Goel
Korean J Gastroenterol. 2018;72(3):135-140. doi: 10.4166/kjg.2018.72.3.135.Comparison of the Diagnostic Performances of Same-session Endoscopic Ultrasound- and Endoscopic Retrograde Cholangiopancreatography-guided Tissue Sampling for Suspected Biliary Strictures at Different Primary Tumor Sites
Seong Jae Yeo, Chang Min Cho, Min Kyu Jung, An Na Seo, Han Ik Bae
Korean J Gastroenterol. 2019;73(4):213-218. doi: 10.4166/kjg.2019.73.4.213.
Reference
-
References
1. Farrell JJ. Diagnosing pancreatic malignancy in the setting of chronic pancreatitis: is there room for improvement? Gastrointest Endosc. 2005; 62:737–741.
Article2. Krishna NB, Mehra M, Reddy AV, Agarwal B. EUS/EUS-FNA for abdominal pancreatic cancer: influence of chronic pancreatitis and clinical presentation with or without obstructive jaundice on abdominal characteristics. Gastrointest Endosc. 2009; 70:70–79.3. Puli SR, Bechtold ML, Buxbaum JL, Eloubeidi MA. How good is abdominal ultrasound-guided fine-needle aspiration in abdominal the correct etiology for a solid pancreatic mass?: a metaanalysis and systematic review. Pancreas. 2013; 42:20–26.4. Fritscher-Ravens A, Brand L, Knöfel WT, et al. Comparison of abdominal ultrasound-guided fine needle aspiration for focal abdominal lesions in patients with normal parenchyma and chronic pancreatitis. Am J Gastroenterol. 2002; 97:2768–2775.5. Varadarajulu S, Tamhane A, Eloubeidi MA. Yield of EUS-guided FNA of pancreatic masses in the presence or the absence of chronic pancreatitis. Gastrointest Endosc. 2005; 62:728–736.
Article6. Eloubeidi MA, Varadarajulu S, Desai S, et al. A prospective abdominal of an algorithm incorporating routine preoperative abdominal ultrasound-guided fine needle aspiration in suspected pancreatic cancer. J Gastrointest Surg. 2007; 11:813–819.7. Sun B, Yang X, Ping B, He Y, Zhang Z. Impact of inconclusive abdominal ultrasound-guided fine-needle aspiration results in the management and outcome of patients with solid pancreatic masses. Dig Endosc. 2015; 27:130–136.8. Bang JY, Varadarajulu S. Neoplasia in chronic pancreatitis: how to maximize the yield of endoscopic ultrasound-guided fine abdominal aspiration. Clin Endosc. 2014; 47:420–424.9. Bloomston M, Bekaii-Saab TS, Kosuri K, et al. Preoperative abdominal antigen 19–9 is most predictive of malignancy in older jaundiced patients undergoing pancreatic resection. Pancreas. 2006; 33:246–249.10. Tessler DA, Catanzaro A, Velanovich V, Havstad S, Goel S. Predictors of cancer in patients with suspected pancreatic abdominal without a tissue diagnosis. Am J Surg. 2006; 191:191–197.11. Lee H, Lee JK, Kang SS, et al. Is there any clinical or radiologic feature as a preoperative marker for differentiating mass-forming pancreatitis from early-stage pancreatic adenocarcinoma? Hepatogastroenterology. 2007; 54:2134–2140.12. Cai QC, Chen Y, Xiao Y, et al. A prediction rule for estimating abdominal cancer risk in chronic pancreatitis patients with focal abdominal mass lesions with prior negative EUS-FNA cytology. Scand J Gastroenterol. 2011; 46:464–470.13. Alston EA, Bae S, Eltoum IA. Suspicious cytologic diagnostic abdominal in endoscopic ultrasound-guided FNA of the pancreas: fol-low-up and outcomes. Cancer Cytopathol. 2016; 124:53–57.14. Lee HS, Kim TN, Lee DH, et al. Korean guideline for chronic pancreatitis. Korean J Pancreas Biliary Tract. 2008; 13:38–48.15. Siddiqui UD, Rossi F, Rosenthal LS, Padda MS, Murali-Dharan V, Aslanian HR. EUS-guided FNA of solid pancreatic masses: a abdominal, randomized trial comparing 22-gauge and 25-gauge needles. Gastrointest Endosc. 2009; 70:1093–1097.16. Pitman MB, Centeno BA, Ali SZ, et al. Standardized terminology and nomenclature for pancreatobiliary cytology: the Papanicolaou Society of Cytopathology guidelines. Diagn Cytopathol. 2014; 42:338–350.
Article17. Hewitt MJ, McPhail MJ, Possamai L, Dhar A, Vlavianos P, Monahan KJ. EUS-guided FNA for diagnosis of solid pancreatic neoplasms: a metaanalysis. Gastrointest Endosc. 2012; 75:319–331.
Article18. Ardengh JC, Lopes CV, Campos AD, Pereira de Lima LF, Venco F, Módena JL. Endoscopic ultrasound and fine needle aspiration in chronic pancreatitis: differential diagnosis between pseudoabdominalal masses and pancreatic cancer. JOP. 2007; 8:413–421.19. Savides TJ, Donohue M, Hunt G, et al. EUS-guided FNA diagnostic yield of malignancy in solid pancreatic masses: a benchmark for quality performance measurement. Gastrointest Endosc. 2007; 66:277–282.20. Abdelgawwad MS, Alston E, Eltoum IA. The frequency and cancer risk associated with the atypical cytologic diagnostic category in endoscopic ultrasound-guided fine-needle aspiration abdominal of solid pancreatic lesions: a metaanalysis and argument for a Bethesda system for reporting cytopathology of the pancreas. Cancer Cytopathol. 2013; 121:620–628.21. Layfield LJ, Schmidt RL, Hirschowitz SL, Olson MT, Ali SZ, Dodd LL. Significance of the diagnostic categories “atypical” and “suspicious for malignancy” in the cytologic diagnosis of solid pancreatic masses. Diagn Cytopathol. 2014; 42:292–296.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Role of Repeated Endoscopic Ultrasound-Guided Fine Needle Aspiration for Inconclusive Initial Cytology Result
- How to optimize the diagnostic yield of endoscopic ultrasoundguided fine-needle sampling in solid pancreatic lesions from a technical perspective
- Endoscopic Ultrasound-Guided Direct Intervention for Solid Pancreatic Tumors
- Endoscopic Ultrasound-Guided Fine Needle Aspiration in Cystic Pancreatic Lesions
- How Can We Get the Best Results with Endoscopic Ultrasound-Guided Fine Needle Aspiration?