Lab Anim Res.
2017 Jun;33(2):76-83. 10.5625/lar.2017.33.2.76.
Pine bark extract (Pycnogenol®) suppresses cigarette smoke-induced fibrotic response via transforming growth factor-β1/Smad family member 2/3 signaling
- Affiliations
-
- 1College of Veterinary Medicine (BK21 Plus Project Team), Chonnam National University, 77 Yongbongro, Buk-gu, Gwangju 500-757, Korea. dvmmk79@gmail.com
- 2Research Center, Dongnam Institute of Radiological and Medical Sciences (DIRAMS), Jwadong-gil 40, Gijang-gun, Busan 619-953, Korea.
- 3College of Health Sciences, Cheongju University, 298 Daesungro, Sangdang-gu, Cheongju, Chungbuk 360-764, Korea.
- KMID: 2407419
- DOI: http://doi.org/10.5625/lar.2017.33.2.76
Abstract
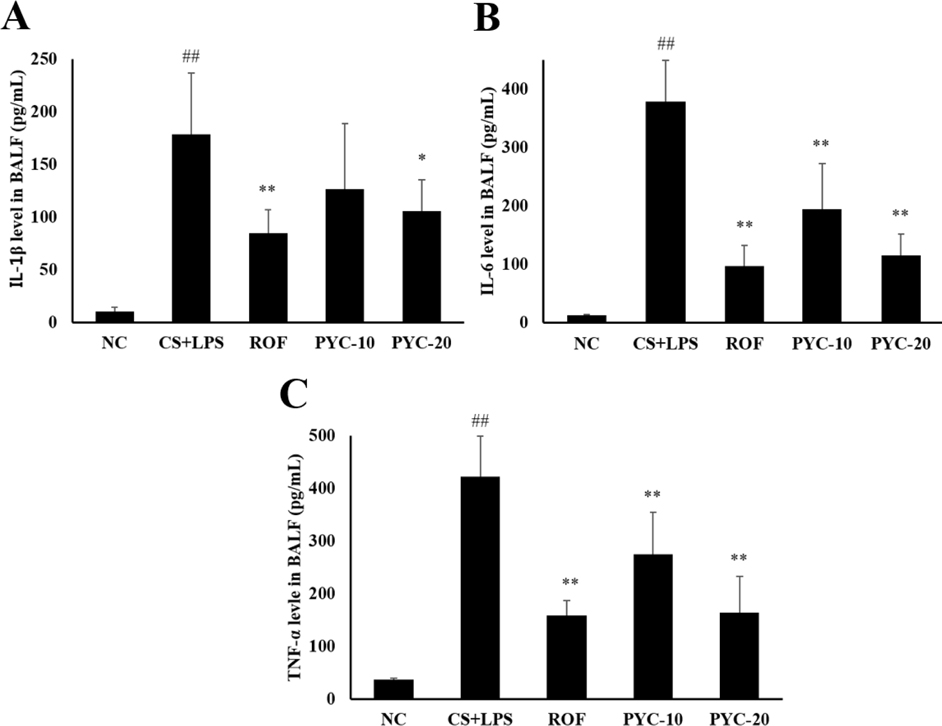
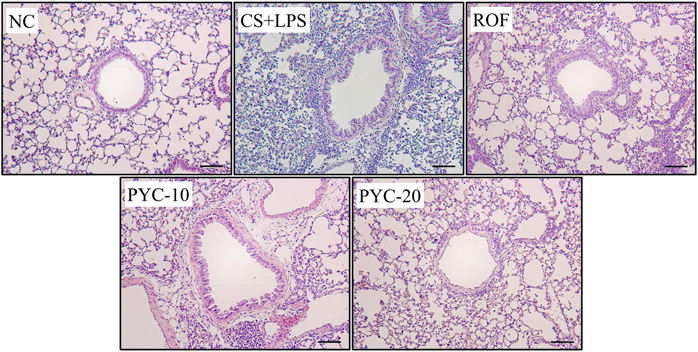
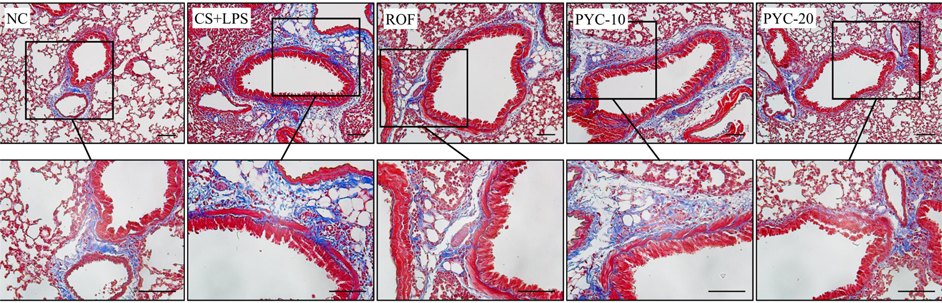
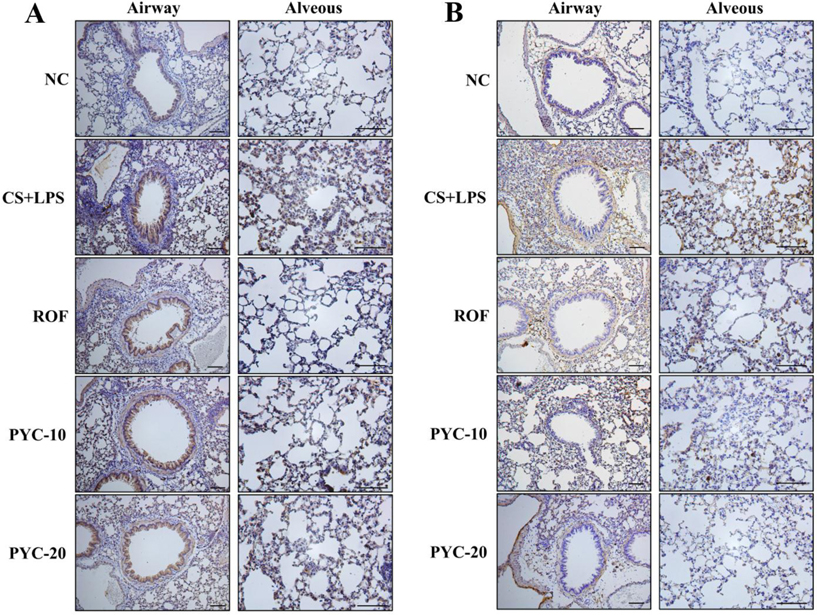
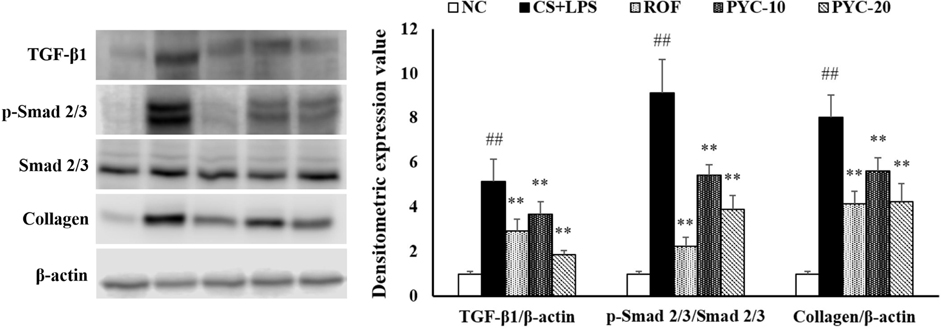
- Chronic obstructive pulmonary diseases (COPD) is an important disease featured as intense inflammation, protease imbalance, and air flow limitation and mainly induced by cigarette smoke (CS). In present study, we explored the effects of Pycnogenol® (PYC, pine bark extract) on pulmonary fibrosis caused by CS+lipopolysaccharide (LPS) exposure. Mice were treated with LPS intranasally on day 12 and 26, followed by CS exposure for 1 h/day (8 cigarettes per day) for 4 weeks. One hour before CS exposure, 10 and 20 mg/kg of PYC were administered by oral gavage for 4 weeks. PYC effectively reduced the number of inflammatory cells and proinflammatory mediators caused by CS+LPS exposure in bronchoalveolar lavage fluid. PYC inhibited the collagen deposition on lung tissue caused by CS+LPS exposure, as evidenced by Masson's trichrome stain. Furthermore, transforming growth factor-β1 (TGF-β1) expression and Smad family member 2/3 (Smad 2/3) phosphorylation were effectively suppressed by PYC treatment. PYC markedly reduced the collagen deposition caused by CS+LPS exposure, which was closely involved in TGF-β1/Smad 2/3 signaling, which is associated with pulmonary fibrotic change. These findings suggest that treatment with PYC could be a therapeutic strategy for controlling COPD progression.
Keyword
MeSH Terms
Figure
Reference
-
1. Churg A, Tai H, Coulthard T, Wang R, Wright JL. Cigarette smoke drives small airway remodeling by induction of growth factors in the airway wall. Am J Respir Crit Care Med. 2006; 174(12):1327–1334.2. Jeffery PK. Remodeling in asthma and chronic obstructive lung disease. Am J Respir Crit Care Med. 2001; 164:S28–S38.3. Milara J, Peiró T, Serrano A, Cortijo J. Epithelial to mesenchymal transition is increased in patients with COPD and induced by cigarette smoke. Thorax. 2013; 68(5):410–420.4. Dhainaut JF, Charpentier J, Chiche JD. Transforming growth factor-beta: a mediator of cell regulation in acute respiratory distress syndrome. Crit Care Med. 2003; 31(4):S258–S264.5. Xu F, Liu C, Zhou D, Zhang L. TGF-â/SMAD Pathway and Its Regulation in Hepatic Fibrosis. J Histochem Cytochem. 2016; 64(3):157–167.6. Brandsma CA, Timens W, Jonker MR, Rutgers B, Noordhoek JA, Postma DS. Differential effects of fluticasone on extracellular matrix production by airway and parenchymal fibroblasts in severe COPD. Am J Physiol Lung Cell Mol Physiol. 2013; 305(8):L582–L589.7. Farid M, Kanaji N, Nakanishi M, Gunji Y, Michalski J, Iwasawa S, Ikari J, Wang X, Basma H, Nelson AJ, Liu X, Rennard SI. Smad3 mediates cigarette smoke extract (CSE) induction of VEGF release by human fetal lung fibroblasts. Toxicol Lett. 2013; 220(2):126–134.8. Ko JW, Lee IC, Park SH, Moon C, Kang SS, Kim SH, Kim JC. Protective effects of pine bark extract against cisplatin-induced hepatotoxicity and oxidative stress in rats. Lab Anim Res. 2014; 30(4):174–180.9. Matsumori A, Higuchi H, Shimada M. French maritime pine bark extract inhibits viral replication and prevents development of viral myocarditis. J Card Fail. 2007; 13(9):785–791.10. Mei L, Mochizuki M, Hasegawa N. Hepatoprotective effects of pycnogenol in a rat model of non-alcoholic steatohepatitis. Phytother Res. 2012; 26(10):1572–1574.11. Cho HS, Lee MH, Lee JW, No KO, Park SK, Lee HS, Kang S, Cho WG, Park HJ, Oh KW, Hong JT. Anti-wrinkling effects of the mixture of vitamin C, vitamin E, pycnogenol and evening primrose oil, and molecular mechanisms on hairless mouse skin caused by chronic ultraviolet B irradiation. Photodermatol Photoimmunol Photomed. 2007; 23(5):155–162.12. Shin NR, Ryu HW, Ko JW, Park JW, Kwon OK, Oh SR, Kim JC, Shin IS, Ahn KS. A standardized bark extract of Pinus pinaster Aiton (Pycnogenol®) attenuated chronic obstructive pulmonary disease via Erk-sp1 signaling pathway. J Ethnopharmacol. 2016; 194:412–420.13. Tashkin DP. Roflumilast: the new orally active, selective phophodiesterase-4 inhibitor, for the treatment of COPD. Expert Opin Pharmacother. 2014; 15(1):85–96.14. Shin IS, Shin NR, Park JW, Jeon CM, Hong JM, Kwon OK, Kim JS, Lee IC, Kim JC, Oh SR, Ahn KS. Melatonin attenuates neutrophil inflammation and mucus secretion in cigarette smoke-induced chronic obstructive pulmonary diseases via the suppression of Erk-Sp1 signaling. J Pineal Res. 2015; 58(1):50–60.15. Takizawa H, Tanaka M, Takami K, Ohtoshi T, Ito K, Satoh M, Okada Y, Yamasawa F, Nakahara K, Umeda A. Increased expression of transforming growth factor-beta1 in small airway epithelium from tobacco smokers and patients with chronic obstructive pulmonary disease (COPD). Am J Respir Crit Care Med. 2001; 163(6):1476–1483.16. Eltom S, Belvisi MG, Stevenson CS, Maher SA, Dubuis E, Fitzgerald KA, Birrell MA. Role of the inflammasome-caspase1/11-IL-1/18 axis in cigarette smoke driven airway inflammation: an insight into the pathogenesis of COPD. PLoS One. 2014; 9(11):e112829.17. Barnes PJ. Alveolar macrophages as orchestrators of COPD. COPD. 2004; 1(1):59–70.18. Ryu HW, Song HH, Shin IS, Cho BO, Jeong SH, Kim DY, Ahn KS, Oh SR. Suffruticosol A isolated from paeonia lactiflora seedcases attecuates airway inflammation in mice induced by cigarette smoke and LPS exposure. J Funct Foods. 2015; 17:774–784.19. Kumar V, Abbas AK, Fausto N, Robbins SL, Cotran RS. Tissue renewal and repair: regeneration, healing, and fibrosis. Pathologic basis of disease. 2005; 7:87–118.20. Wynn TA. Cellular and molecular mechanisms of fibrosis. J Pathol. 2008; 214(2):199–210.21. Peng YJ, Lee CH, Wang CC, Salter DM, Lee HS. Pycnogenol attenuates the inflammatory and nitrosative stress on joint inflammation induced by urate crystals. Free Radic Biol Med. 2012; 52(4):765–774.22. Khan MM, Kempuraj D, Thangavel R, Zaheer A. Protection of MPTP-induced neuroinflammation and neurodegeneration by Pycnogenol. Neurochem Int. 2013; 62(4):379–388.23. Shin IS, Shin NR, Jeon CM, Hong JM, Kwon OK, Kim JC, Oh SR, Hahn KW, Ahn KS. Inhibitory effects of Pycnogenol® (French maritime pine bark extract) on airway inflammation in ovalbumin-induced allergic asthma. Food Chem Toxicol. 2013; 62:681–686.24. Xia YF, Zhang JH, Xu ZF, Deng XM. Pycnogenol, a compound isolated from the bark of pinus maritime mill, attenuates ventilator-induced lung injury through inhibiting NF-κB-mediated inflammatory response. Int J Clin Exp Med. 2015; 8(2):1824–1833.25. Barkauskas CE, Noble PW. Cellular mechanisms of tissue fibrosis. 7. New insights into the cellular mechanisms of pulmonary fibrosis. Am J Physiol Cell Physiol. 2014; 306(11):C987–C996.26. Gao W, Li L, Wang Y, Zhang S, Adcock IM, Barnes PJ, Huang M, Yao X. Bronchial epithelial cells: The key effector cells in the pathogenesis of chronic obstructive pulmonary disease? Respirology. 2015; 20(5):722–729.27. Spurzem JR, Rennard SI. Pathogenesis of COPD. Semin Respir Crit Care Med. 2005; 26(2):142–153.28. Zhang H, Liu H, Borok Z, Davies KJ, Ursini F, Forman HJ. Cigarette smoke extract stimulates epithelial-mesenchymal transition through Src activation. Free Radic Biol Med. 2012; 52(8):1437–1442.29. Vietti G, Lison D, van den Brule S. Mechanisms of lung fibrosis induced by carbon nanotubes: towards an Adverse Outcome Pathway (AOP). Part Fibre Toxicol. 2016; 13(1):11.30. Zandvoort A, Postma DS, Jonker MR, Noordhoek JA, Vos JT, van der Geld YM, Timens W. Altered expression of the Smad signalling pathway: implications for COPD pathogenesis. Eur Respir J. 2006; 28(3):533–541.31. Adcock IM, Caramori G, Barnes PJ. Chronic obstructive pulmonary disease and lung cancer: new molecular insights. Respiration. 2011; 81(4):265–284.32. Mahmood MQ, Reid D, Ward C, Muller HK, Knight DA, Sohal SS, Walters EH. Transforming growth factor (TGF) β1 and Smad signalling pathways: A likely key to EMT-associated COPD pathogenesis. Respirology. 2017; 22(1):133–140.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Hypoxia-Inducible Factor 1α Regulates the Transforming Growth Factor β1/SMAD Family Member 3 Pathway to Promote Breast Cancer Progression
- The Role of Tripartite Motif Family Proteins in TGF-β Signaling Pathway and Cancer
- TGF-beta-activated kinase-1: New insights into the mechanism of TGF-beta signaling and kidney disease
- The Effects of Retinoic Acid and MAPK Inhibitors on Phosphorylation of Smad2/3 Induced by Transforming Growth Factor β1
- Hydrogen sulfide alleviates hypothyroidism-induced myocardial fibrosis in rats through stimulating autophagy and inhibiting TGF-β1/Smad2 pathway