Anat Cell Biol.
2017 Dec;50(4):275-283. 10.5115/acb.2017.50.4.275.
Ameliorative effects of type-A procyanidins polyphenols from cinnamon bark in compound 48/80-induced mast cell degranulation
- Affiliations
-
- 1Department of Scientific Affairs, Indus Biotech Private Limited, Pune, India. prasad@indusbiotech.com
- 2Department of Pharmacology, Sinhgad Institute of Pharmacy, Pune, India.
- KMID: 2399896
- DOI: http://doi.org/10.5115/acb.2017.50.4.275
Abstract
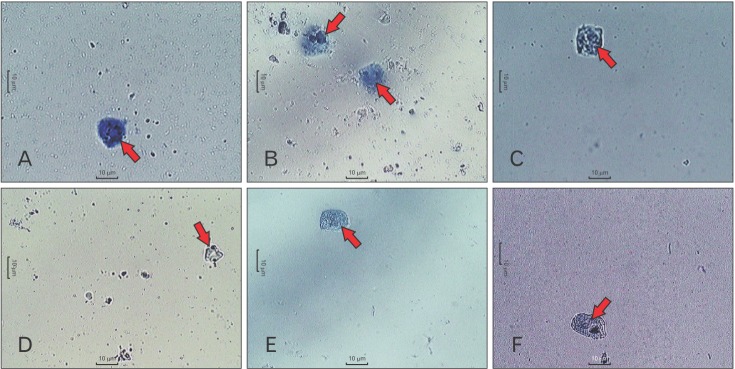
- Allergic diseases are a significant health concern in developing countries. Type-A procyanidin polyphenols from cinnamon (Cinnamomum zeylanicum Blume) bark (TAPP-CZ) possesses antiasthmatic and antiallergic potential. The present study was aimed at the possible anti-allergic mechanism of TAPP-CZ against the compound 48/80 (C48/80)-induced mast cell degranulation in isolated rat peritoneal mast cells (RPMCs). TAPP-CZ (1, 3, 10, and 30 µg/ml) was incubated for 3 hours with isolated, purified RPMCs. The C48/80 (1 µg/ml) was used to induce mast cell degranulation. The mast cell viability was assessed using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay whereas histamine, β-hexosaminidase (β-HEX), and interleukin-4 (IL-4) levels were determined in RPMCs. TAPP-CZ (3, 10, and 30 µg/ml) showed significant and dose-dependent decrease in a number of degranulated cells and levels of markers (histamine, β-HEX, and IL-4) as compared with C48/80 control. In conclusion, TAPP-CZ stabilizes mast cell and cause inhibition of the allergic markers such as histamine, IL-4, and β-HEX in IgE-mediated manner. The present study supports mast cell stabilization as a possible mechanism of action of TAPP-CZ against immune respiratory disorders such as asthma and allergic rhinitis.
MeSH Terms
Figure
Cited by 1 articles
-
Tumor associated mast cells: biological roles and therapeutic applications
Shikha Saxena, Anil Singh, Priyanka Singh
Anat Cell Biol. 2020;53(3):245-251. doi: 10.5115/acb.19.181.
Reference
-
1. Takizawa H. Impact of air pollution on allergic diseases. Korean J Intern Med. 2011; 26:262–273. PMID: 22016586.2. Kay AB. Overview of ‘allergy and allergic diseases: with a view to the future’. Br Med Bull. 2000; 56:843–864. PMID: 11359624.3. Galli SJ, Kalesnikoff J, Grimbaldeston MA, Piliponsky AM, Williams CM, Tsai M. Mast cells as “tunable” effector and immunoregulatory cells: recent advances. Annu Rev Immunol. 2005; 23:749–786. PMID: 15771585.4. Puxeddu I, Piliponsky AM, Bachelet I, Levi-Schaffer F. Mast cells in allergy and beyond. Int J Biochem Cell Biol. 2003; 35:1601–1607. PMID: 12962699.5. Pearce FL, Befus AD, Gauldie J, Bienenstock J. Mucosal mast cells. II. Effects of anti-allergic compounds on histamine secretion by isolated intestinal mast cells. J Immunol. 1982; 128:2481–2486. PMID: 6176639.6. Murrant T, Bihari D. Anaphylaxis and anaphylactoid reactions. Int J Clin Pract. 2000; 54:322–328. PMID: 10954960.7. Kawakami T, Ando T, Kimura M, Wilson BS, Kawakami Y. Mast cells in atopic dermatitis. Curr Opin Immunol. 2009; 21:666–678. PMID: 19828304.8. Choi YH, Chai OH, Han EH, Choi SY, Kim HT, Song CH. Lipoic acid suppresses compound 48/80-induced anaphylaxis-like reaction. Anat Cell Biol. 2010; 43:317–324. PMID: 21267406.9. Choi YH, Yan GH, Chai OH, Song CH. Inhibitory effects of curcumin on passive cutaneous anaphylactoid response and compound 48/80-induced mast cell activation. Anat Cell Biol. 2010; 43:36–43. PMID: 21190003.10. Nishikawa H, Kitani S. Tea catechins have dual effect on mast cell degranulation induced by compound 48/80. Int Immunopharmacol. 2008; 8:1207–1215. PMID: 18602066.11. Galli SJ, Tsai M, Piliponsky AM. The development of allergic inflammation. Nature. 2008; 454:445–454. PMID: 18650915.12. Levy D, Burstein R, Kainz V, Jakubowski M, Strassman AM. Mast cell degranulation activates a pain pathway underlying migraine headache. Pain. 2007; 130:166–176. PMID: 17459586.13. Oliveira SM, Drewes CC, Silva CR, Trevisan G, Boschen SL, Moreira CG, de Almeida Cabrini D, Da Cunha C, Ferreira J. Involvement of mast cells in a mouse model of postoperative pain. Eur J Pharmacol. 2011; 672:88–95. PMID: 22004612.14. Drummond PD. The effect of cutaneous mast cell degranulation on sensitivity to heat. Inflamm Res. 2004; 53:309–315. PMID: 15241566.15. Rudick CN, Bryce PJ, Guichelaar LA, Berry RE, Klumpp DJ. Mast cell-derived histamine mediates cystitis pain. PLoS One. 2008; 3:e2096. PMID: 18461160.16. Barbara G, Stanghellini V, De Giorgio R, Cremon C, Cottrell GS, Santini D, Pasquinelli G, Morselli-Labate AM, Grady EF, Bunnett NW, Collins SM, Corinaldesi R. Activated mast cells in proximity to colonic nerves correlate with abdominal pain in irritable bowel syndrome. Gastroenterology. 2004; 126:693–702. PMID: 14988823.17. Anaf V, Chapron C, El Nakadi I, De Moor V, Simonart T, Noël JC. Pain, mast cells, and nerves in peritoneal, ovarian, and deep infiltrating endometriosis. Fertil Steril. 2006; 86:1336–1343. PMID: 17007852.18. Theoharides TC, Kalogeromitros D. The critical role of mast cells in allergy and inflammation. Ann N Y Acad Sci. 2006; 1088:78–99. PMID: 17192558.19. Miyatake A, Fujita M, Nagasaka Y, Fujita K, Tamari M, Watanabe D, Nakano N, Hidari KI, Suzuki Y. The new role of disodium cromoglycate in the treatment of adults with bronchial asthma. Allergol Int. 2007; 56:231–239. PMID: 17519581.20. Beach JE, Blair AM, Clarke AJ, Bonfield CT. Cromolyn sodium toxicity studies in primates. Toxicol Appl Pharmacol. 1981; 57:367–400. PMID: 6784269.21. Hossen MA, Inoue T, Shinmei Y, Minami K, Fujii Y, Kamei C. Caffeic acid inhibits compound 48/80-induced allergic symptoms in mice. Biol Pharm Bull. 2006; 29:64–66. PMID: 16394511.22. Han SY, Bae JY, Park SH, Kim YH, Park JH, Kang YH. Resveratrol inhibits IgE-mediated basophilic mast cell degranulation and passive cutaneous anaphylaxis in mice. J Nutr. 2013; 143:632–639. PMID: 23514766.23. Li GZ, Chai OH, Song CH. Inhibitory effects of epigallocatechin gallate on compound 48/80-induced mast cell activation and passive cutaneous anaphylaxis. Exp Mol Med. 2005; 37:290–296. PMID: 16155406.24. Tokura T, Nakano N, Ito T, Matsuda H, Nagasako-Akazome Y, Kanda T, Ikeda M, Okumura K, Ogawa H, Nishiyama C. Inhibitory effect of polyphenol-enriched apple extracts on mast cell degranulation in vitro targeting the binding between IgE and FcεRI. Biosci Biotechnol Biochem. 2005; 69:1974–1977. PMID: 16244451.25. Kanda T, Akiyama H, Yanagida A, Tanabe M, Goda Y, Toyoda M, Teshima R, Saito Y. Inhibitory effects of apple polyphenol on induced histamine release from RBL-2H3 cells and rat mast cells. Biosci Biotechnol Biochem. 1998; 62:1284–1289. PMID: 9720210.26. Matsuo N, Yamada K, Shoji K, Mori M, Sugano M. Effect of tea polyphenols on histamine release from rat basophilic leukemia (RBL-2H3) cells: the structure-inhibitory activity relationship. Allergy. 1997; 52:58–64. PMID: 9062630.27. Ravindran PN, Nirmal Babu K, Shylaja M. Cinnamon and cassia: the genus Cinnamomum. Boca Raton, FL: CRC Press;2004.28. Cao H, Anderson RA. Cinnamon polyphenol extract regulates tristetraprolin and related gene expression in mouse adipocytes. J Agric Food Chem. 2011; 59:2739–2744. PMID: 21329350.29. Joshi SS, Kuszynski CA, Bagchi M, Bagchi D. Chemopreventive effects of grape seed proanthocyanidin extract on Chang liver cells. Toxicology. 2000; 155:83–90. PMID: 11154800.30. Vetal S, Bodhankar SL, Mohan V, Thakurdesai PA. Anti-inflammatory and anti-arthritic activity of type-A procyanidine polyphenols from bark of Cinnamomum zeylanicum in rats. Food Sci Hum Wellness. 2013; 2:59–67.31. Kandhare AD, Bodhankar SL, Singh V, Mohan V, Thakurdesai PA. Anti-asthmatic effects of type-A procyanidine polyphenols from cinnamon bark in ovalbumin-induced airway hyperresponsiveness in laboratory animals. Biomed Aging Pathol. 2013; 3:23–30.32. Aswar UM, Kandhare AD, Mohan V, Thakurdesai PA. Anti-allergic effect of intranasal administration of type-A procyanidin polyphenols based standardized extract of cinnamon bark in ovalbumin sensitized BALB/c mice. Phytother Res. 2015; 29:423–433. PMID: 25504814.33. Kandhare A, Aswar U, Mohan V, Bodhankar SL, Thakudesai PA. Effect of Type-A procyanidine polyphenols from bark of Cinnamomum Zeylanicum on allergic rhinitis model in balb-c mice [GU-6]. In : 45th Annual Conference of Indian Pharmacological Society (IPSCON-2012) and International Conference on “Navigating Pharmacology towards Safe and Effective Therapy”; 2013 Jan 5-7; Nagpur, India. Hyderabad: Indian Pharmacological Society;2013.34. Anderson RA, Broadhurst CL, Polansky MM, Schmidt WF, Khan A, Flanagan VP, Schoene NW, Graves DJ. Isolation and characterization of polyphenol type-A polymers from cinnamon with insulin-like biological activity. J Agric Food Chem. 2004; 52:65–70. PMID: 14709014.35. Lazarus SA, Adamson GE, Hammerstone JF, Schmitz HH. High-performance liquid Chromatography/Mass spectrometry analysis of proanthocyanidins in foods and beverages. J Agric Food Chem. 1999; 47:3693–3701. PMID: 10552707.36. Cochrane DE, Douglas WW. Calcium-induced extrusion of secretory granules (exocytosis) in mast cells exposed to 48-80 or the ionophores A-23187 and X-537A. Proc Natl Acad Sci U S A. 1974; 71:408–412. PMID: 4205591.37. Hachisuka H, Nomura H, Sakamoto F, Mori O, Okubo K, Sasai Y. Effect of antianaphylactic agents on substance-P induced histamine release from rat peritoneal mast cells. Arch Dermatol Res. 1988; 280:158–162. PMID: 2454082.38. Mascotti K, McCullough J, Burger SR. HPC viability measurement: trypan blue versus acridine orange and propidium iodide. Transfusion. 2000; 40:693–696. PMID: 10864990.39. Yoshimura T, Hamaguchi E, Usami E, Nakashima K, Kawaguchi M, Suzuki N, Okamoto Y, Nakao T, Yamazaki F. Increased in vitro release of interferon-gamma from ampicillin-stimulated peripheral blood mononuclear cells in Stevens-Johnson syndrome. Biol Pharm Bull. 2004; 27:929–931. PMID: 15187450.40. Gao Y, Hou R, Fei Q, Fang L, Han Y, Cai R, Peng C, Qi Y. The Three-Herb Formula Shuang-Huang-Lian stabilizes mast cells through activation of mitochondrial calcium uniporter. Sci Rep. 2017; 7:38736. PMID: 28045016.41. Schroeder JT. Basophils: emerging roles in the pathogenesis of allergic disease. Immunol Rev. 2011; 242:144–160. PMID: 21682743.42. Aung G, Niyonsaba F, Ushio H, Kajiwara N, Saito H, Ikeda S, Ogawa H, Okumura K. Catestatin, a neuroendocrine antimicrobial peptide, induces human mast cell migration, degranulation and production of cytokines and chemokines. Immunology. 2011; 132:527–539. PMID: 21214543.43. Jutel M, Akdis CA. Immunological mechanisms of allergen-specific immunotherapy. Allergy. 2011; 66:725–732. PMID: 21466562.44. Sada K, Miah SM, Maeno K, Kyo S, Qu X, Yamamura H. Regulation of FcεRI-mediated degranulation by an adaptor protein 3BP2 in rat basophilic leukemia RBL-2H3 cells. Blood. 2002; 100:2138–2144. PMID: 12200378.45. Theoharides TC, Kops SK, Bondy PK, Askenase PW. Differential release of serotonin without comparable histamine under diverse conditions in the rat mast cell. Biochem Pharmacol. 1985; 34:1389–1398. PMID: 2581583.46. Tasaka K, Mio M, Okamoto M. Intracellular calcium release induced by histamine releasers and its inhibition by some antiallergic drugs. Ann Allergy. 1986; 56:464–469. PMID: 2424349.47. Gleich GJ. The late phase of the immunoglobulin E-mediated reaction: a link between anaphylaxis and common allergic disease? J Allergy Clin Immunol. 1982; 70:160–169. PMID: 6125535.48. Lewis RA, Robin JL, Austen KF. Pharmacologic regulation of mediator generation and release from the murine bone marrow derived mast cell. Int Arch Allergy Appl Immunol. 1985; 77:121–125. PMID: 2409010.49. Galli SJ. New insights into “the riddle of the mast cells”: microenvironmental regulation of mast cell development and phenotypic heterogeneity. Lab Invest. 1990; 62:5–33. PMID: 2404155.50. Charlesworth EN, Hood AF, Soter NA, Kagey-Sobotka A, Norman PS, Lichtenstein LM. Cutaneous late-phase response to allergen: mediator release and inflammatory cell infiltration. J Clin Invest. 1989; 83:1519–1526. PMID: 2468688.51. Kalinen M. Hypotheses on the contribution of late-phase allergic responses to the understanding and treatment of allergic diseases. J Allergy Clin Immunol. 1984; 73:311–315. PMID: 6366029.52. Jutel M, Klunker S, Akdis M, Malolepszy J, Thomet OA, Zak-Nejmark T, Blaser K, Akdis CA. Histamine upregulates Th1 and downregulates Th2 responses due to different patterns of surface histamine 1 and 2 receptor expression. Int Arch Allergy Immunol. 2001; 124:190–192. PMID: 11306965.53. Jutel M, Watanabe T, Klunker S, Akdis M, Thomet OA, Malolepszy J, Zak-Nejmark T, Koga R, Kobayashi T, Blaser K, Akdis CA. Histamine regulates T-cell and antibody responses by differential expression of H1 and H2 receptors. Nature. 2001; 413:420–425. PMID: 11574888.54. Ling P, Ngo K, Nguyen S, Thurmond RL, Edwards JP, Karlsson L, Fung-Leung WP. Histamine H4 receptor mediates eosinophil chemotaxis with cell shape change and adhesion molecule upregulation. Br J Pharmacol. 2004; 142:161–171. PMID: 15131002.55. László V, Rothe G, Hegyesi H, Szeberényi JB, Orsó E, Schmitz G, Falus A. Increased histidine decarboxylase expression during in vitro monocyte maturation; a possible role of endogenously synthesised histamine in monocyte/macrophage differentiation. Inflamm Res. 2001; 50:428–434. PMID: 11556524.56. Jutel M, Blaser K, Akdis CA. Histamine in allergic inflammation and immune modulation. Int Arch Allergy Immunol. 2005; 137:82–92. PMID: 15832054.57. Wendeler M, Sandhoff K. Hexosaminidase assays. Glycoconj J. 2009; 26:945–952. PMID: 18473163.58. Amin K. The role of mast cells in allergic inflammation. Respir Med. 2012; 106:9–14. PMID: 22112783.59. Pradalier A. Late-phase reaction in asthma: basic mechanisms. Int Arch Allergy Immunol. 1993; 101:322–325. PMID: 8324396.60. Wershil BK, Wang ZS, Gordon JR, Galli SJ. Recruitment of neutrophils during IgE-dependent cutaneous late phase reactions in the mouse is mast cell-dependent: partial inhibition of the reaction with antiserum against tumor necrosis factor-alpha. J Clin Invest. 1991; 87:446–453. PMID: 1991831.61. Matsuda H, Kawakita K, Kiso Y, Nakano T, Kitamura Y. Substance P induces granulocyte infiltration through degranulation of mast cells. J Immunol. 1989; 142:927–931. PMID: 2464033.62. Silvestri M, Bontempelli M, Giacomelli M, Malerba M, Rossi GA, Di Stefano A, Rossi A, Ricciardolo FL. High serum levels of tumour necrosis factor-alpha and interleukin-8 in severe asthma: markers of systemic inflammation? Clin Exp Allergy. 2006; 36:1373–1381. PMID: 17083347.63. Deo SS, Mistry KJ, Kakade AM, Niphadkar PV. Role played by Th2 type cytokines in IgE mediated allergy and asthma. Lung India. 2010; 27:66–71. PMID: 20616938.64. Vo TH, Le NH, Patel MS, Phan LT, Tran Minh NN. Acute allergic reactions in Vietnamese children after drinking a new milk product. Foodborne Pathog Dis. 2012; 9:156–159. PMID: 22315953.65. MacGlashan DW Jr, Bochner BS, Adelman DC, Jardieu PM, Togias A, McKenzie-White J, Sterbinsky SA, Hamilton RG, Lichtenstein LM. Down-regulation of Fc(epsilon)RI expression on human basophils during in vivo treatment of atopic patients with anti-IgE antibody. J Immunol. 1997; 158:1438–1445. PMID: 9013989.66. Saini SS, MacGlashan DW Jr, Sterbinsky SA, Togias A, Adelman DC, Lichtenstein LM, Bochner BS. Down-regulation of human basophil IgE and FCεRIα surface densities and mediator release by anti-IgE-infusions is reversible in vitro and in vivo. J Immunol. 1999; 162:5624–5630. PMID: 10228046.67. Nakano N, Nishiyama C, Tokura T, Nagasako-Akazome Y, Ohtake Y, Okumura K, Ogawa H. Procyanidin C1 from apple extracts inhibits Fc epsilon RI-mediated mast cell activation. Int Arch Allergy Immunol. 2008; 147:213–221. PMID: 18594151.68. Oliver JM, Burg DL, Wilson BS, McLaughlin JL, Geahlen RL. Inhibition of mast cell FceR1-mediated signaling and effector function by the Syk-selective inhibitor, piceatannol. J Biol Chem. 1994; 269:29697–29703. PMID: 7961959.69. Miura K, Lavens-Phillips S, MacGlashan DW Jr. Piceatannol is an effective inhibitor of IgE-mediated secretion from human basophils but is neither selective for this receptor nor acts on syk kinase at concentrations where mediator release inhibition occurs. Clin Exp Allergy. 2001; 31:1732–1739. PMID: 11696049.70. Qin HD, Shi YQ, Liu ZH, Li ZG, Wang HS, Wang H, Liu ZP. Effect of chlorogenic acid on mast cell-dependent anaphylactic reaction. Int Immunopharmacol. 2010; 10:1135–1141. PMID: 20620227.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Inhibitory Effect of Polysaccharide Fraction from Cortex Mori on Compound 48/80-Induced Mast Cell Activation
- Inhibitory Effect of Corni fructus on Compound 48/80-induced Mast Cell Activation and Vascular Permeability
- Inhibitory Effect of Arctium lappa Linne on Compound 48/80-induced Mast Cell Activation and Vascular Permeability
- Inhibitory Effect of Rubus Coreanus on Compound 48/80- or Anti-DNP IgE-Induced Mast Cell Activation
- Inhibitory effects of epigallocatechin gallate on compound 48/80-inducedmast cell activation and passive cutaneous anaphylaxis