J Korean Soc Radiol.
2017 Nov;77(5):286-297. 10.3348/jksr.2017.77.5.286.
Identifying CT-Based Risk Factors Associated with Synchronous Liver Metastases in Colorectal Cancer
- Affiliations
-
- 1Department of Radiology, Gachon University Gil Medical Center, Incheon, Korea. sjchoi1118@gmail.com
- KMID: 2394042
- DOI: http://doi.org/10.3348/jksr.2017.77.5.286
Abstract
- PURPOSE
The aim of this study was to determine the radiologic risk factors of colorectal cancer (CRC) with synchronous liver metastases.
MATERIALS AND METHODS
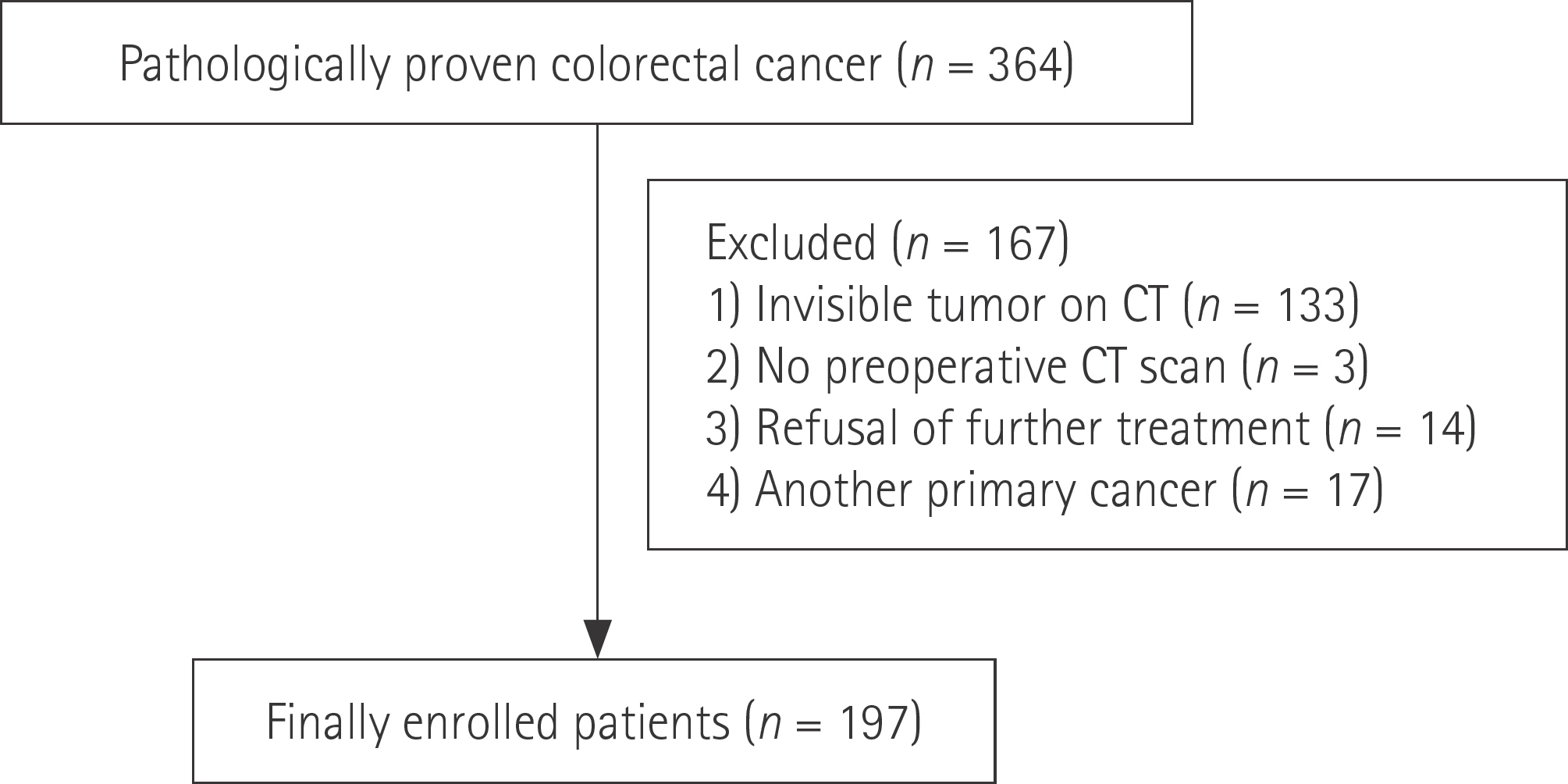
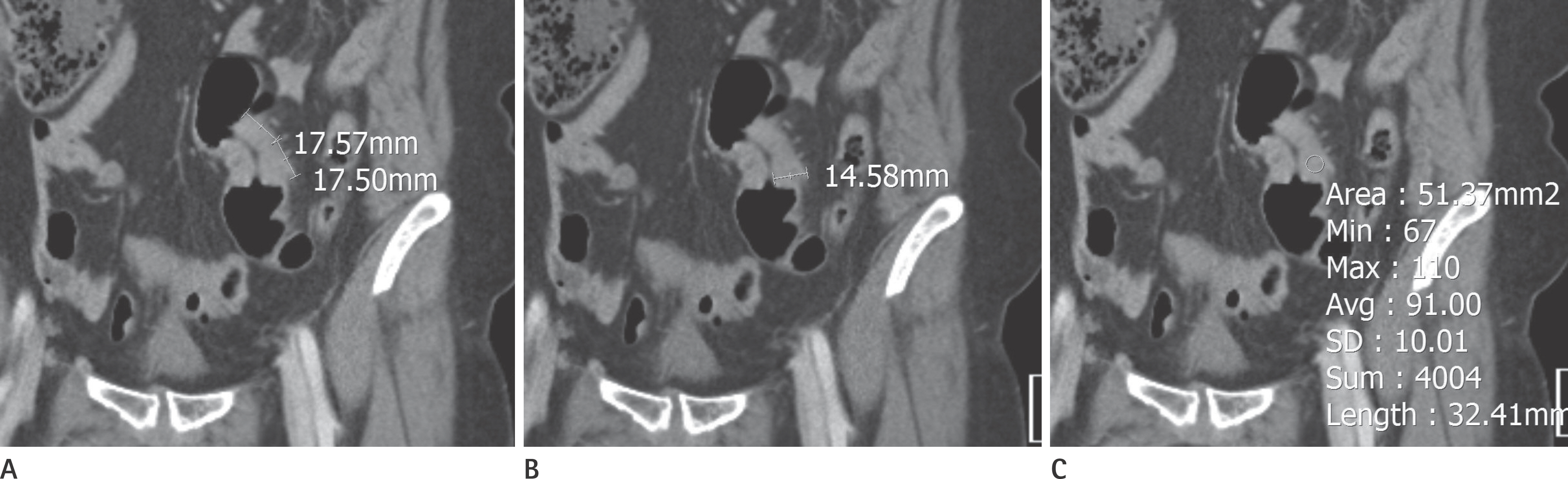
A total of 197 patients with CRC who had a visible tumor on contrast-enhanced abdominopelvic computed tomography and were treated between January 2012 and December 2012 were included. Longitudinal diameter, mural thickness, primary tumor attenuation, and other metastases were evaluated independently. Univariate analysis and multivariate logistic regression analysis were used to identify risk factors associated with the presence of liver metastases.
RESULTS
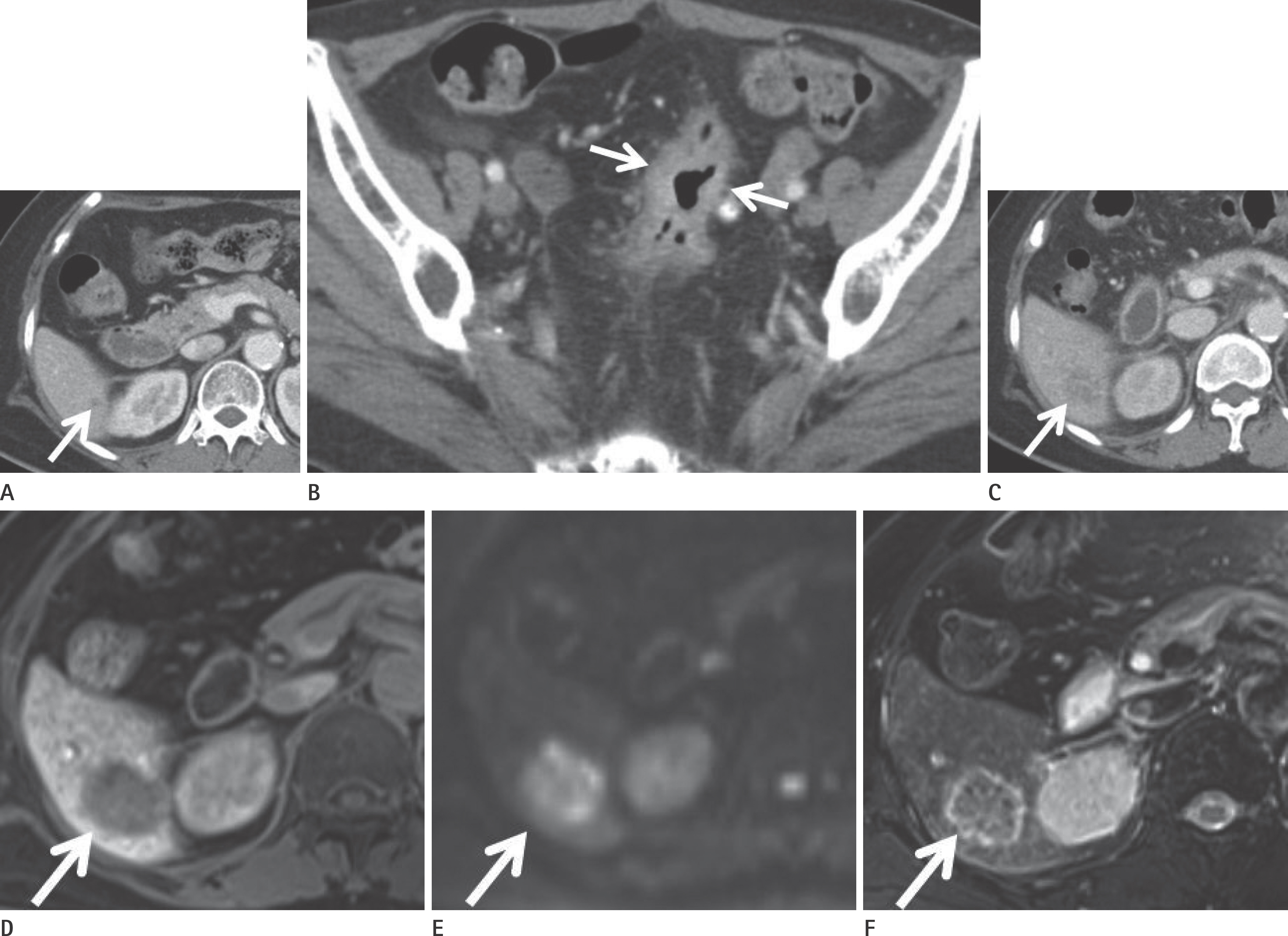
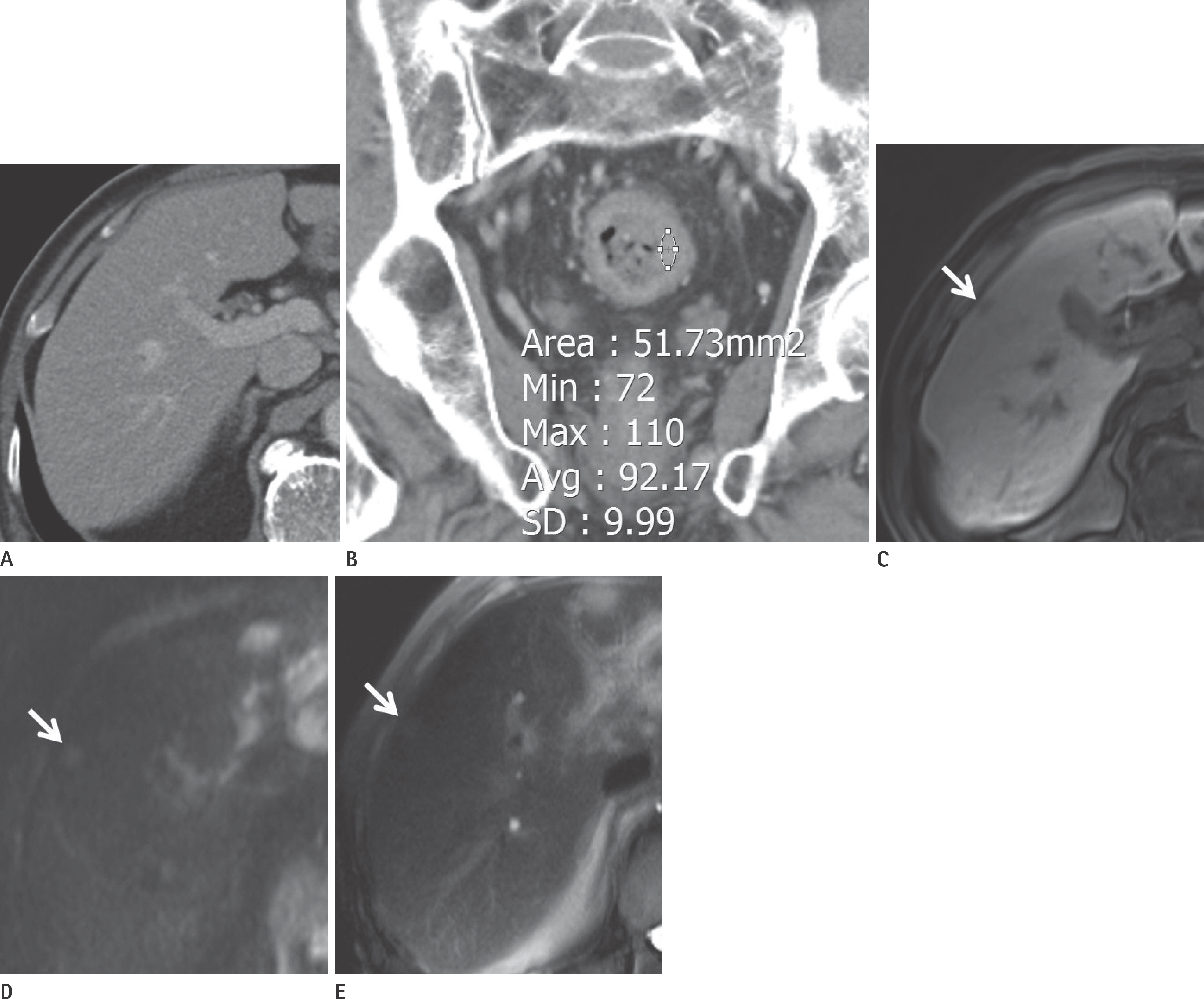
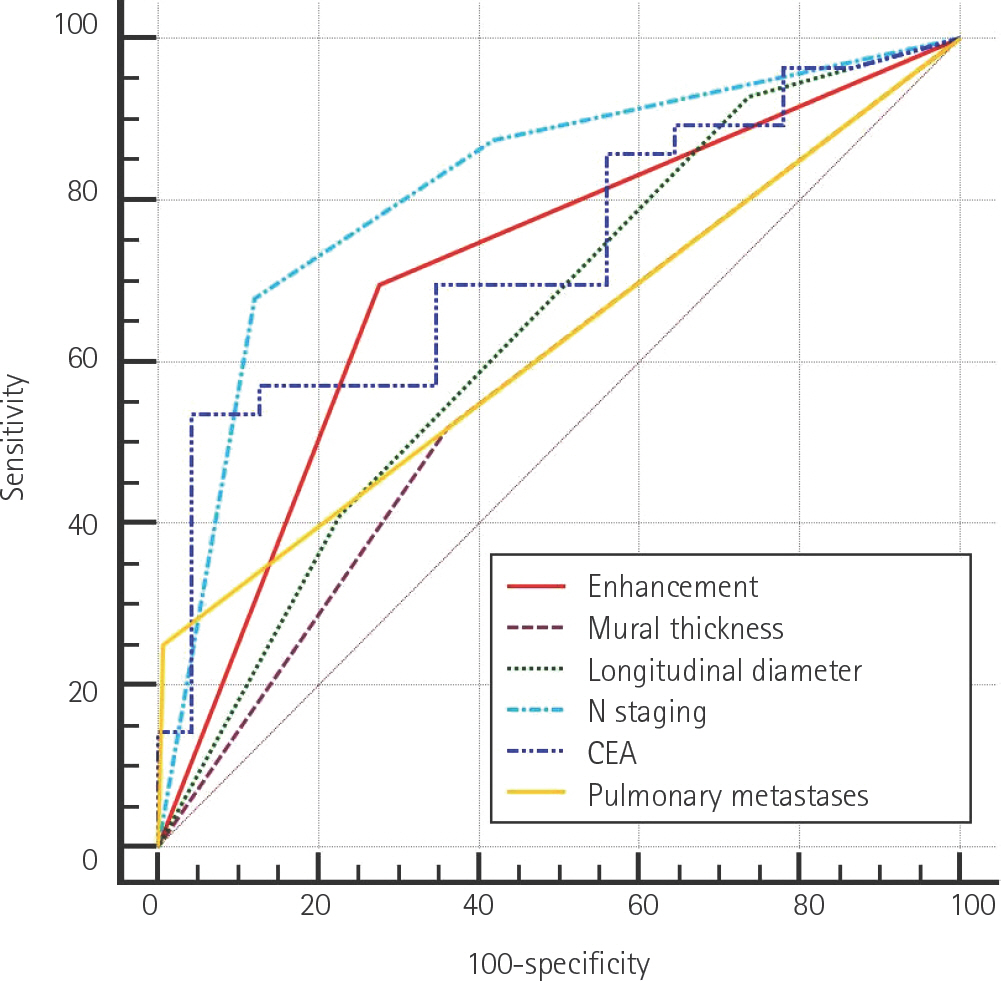
Cases were divided into two groups based on the presence or absence of liver metastases (n = 56 and 141, respectively). Primary tumors with enhancement of ≥ 90 Hounsfield units (HU) were found to have a higher risk of liver metastases than those with enhancement of < 90 HU [odds ratio (OR): 2.619, p = 0.034]. The presence of pulmonary metastases was associated with a higher risk of liver metastases (OR: 14.218, p = 0.025). The presence of lymph node metastases (N2 vs. N0) and carcinoembryonic antigen (CEA) level independently predicted the presence of liver metastases (OR: 8.766, p < 0.001; OR: 1.012, p = 0.048).
CONCLUSION
The identified risk factors of synchronous liver metastases in CRC were tumor mural enhancement, pulmonary metastases, lymph node metastases, and CEA level.
MeSH Terms
Figure
Reference
-
1.Jemal A., Bray F., Center MM., Ferlay J., Ward E., Forman D. Global cancer statistics. CA Cancer J Clin. 2011. 61:69–90.
Article2.Ferlay J., Shin HR., Bray F., Forman D., Mathers C., Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBO-CAN 2008. Int J Cancer. 2010. 127:2893–2917.
Article3.Mella J., Biffin A., Radcliffe AG., Stamatakis JD., Steele RJ. Pop-ulation-based audit of colorectal cancer management in two UK health regions. Colorectal Cancer Working Group, Royal College of Surgeons of England Clinical Epidemiology and Audit Unit. Br J Surg. 1997. 84:1731–1736.4.Paschos KA., Bird N. Current diagnostic and therapeutic ap-proaches for colorectal cancer liver metastasis. Hippokratia. 2008. 12:132–138.5.Geoghegan JG., Scheele J. Treatment of colorectal liver me-tastases. Br J Surg. 1999. 86:158–169.
Article6.Welch JP., Donaldson GA. The clinical correlation of an au-topsy study of recurrent colorectal cancer. Ann Surg. 1979. 189:496–502.7.Schmiegel W., Pox C., Reinacher-Schick A., Adler G., Arnold D., Fleig W, et al. S3 guidelines for colorectal carcinoma: re-sults of an evidence-based consensus conference on Febru-ary 6/7, 2004 and June 8/9, 2007 (for the topics IV, VI and VII). Z Gastroenterol. 2010. 48:65–136.8.Kopetz S., Chang GJ., Overman MJ., Eng C., Sargent DJ., Larson DW, et al. Improved survival in metastatic colorectal cancer is associated with adoption of hepatic resection and im-proved chemotherapy. J Clin Oncol. 2009. 27:3677–3683.
Article9.Minami Y., Kudo M. Radiofrequency ablation of liver metas-tases from colorectal cancer: a literature review. Gut Liver. 2013. 7:1–6.
Article10.Tomlinson JS., Jarnagin WR., DeMatteo RP., Fong Y., Kornprat P., Gonen M, et al. Actual 10-year survival after resection of colorectal liver metastases defines cure. J Clin Oncol. 2007. 25:4575–4580.
Article11.Abdalla EK., Adam R., Bilchik AJ., Jaeck D., Vauthey JN., Mahvi D. Improving resectability of hepatic colorectal metastases: expert consensus statement. Ann Surg Oncol. 2006. 13:1271–1280.
Article12.Rees M., Tekkis PP., Welsh FK., O'Rourke T., John TG. Evaluation of long-term survival after hepatic resection for met-astatic colorectal cancer: a multifactorial model of 929 patients. Ann Surg. 2008. 247:125–135.13.Ye LC., Liu TS., Ren L., Wei Y., Zhu DX., Zai SY, et al. Randomized controlled trial of cetuximab plus chemotherapy for patients with KRAS wild-type unresectable colorectal liver-limited metastases. J Clin Oncol. 2013. 31:1931–1938.
Article14.Cremolini C., Loupakis F., Antoniotti C., Lupi C., Sensi E., Lonar-di S, et al. FOLFOXIRI plus bevacizumab versus FOLFIRI plus bevacizumab as first-line treatment of patients with meta-static colorectal cancer: updated overall survival and molec-ular subgroup analyses of the open-label, phase 3 TRIBE study. Lancet Oncol. 2015. 16:1306–1315.
Article15.Hunter CJ., Garant A., Vuong T., Artho G., Lisbona R., Tekkis P, et al. Adverse features on rectal MRI identify a high-risk group that may benefit from more intensive preoperative staging and treatment. Ann Surg Oncol. 2012. 19:1199–1205.
Article16.Kim YC., Kim JK., Kim MJ., Lee JH., Kim YB., Shin SJ. Feasibility of mesorectal vascular invasion in predicting early distant metastasis in patients with stage T3 rectal cancer based on rectal MRI. Eur Radiol. 2016. 26:297–305.
Article17.Screening for synchronous metastases in colorectal cancer with DW-MRI (SERENADE). Available at:. https://clinicaltri-als.gov/ct2/show/NCT02246634. Published Sep 5, 2014. Ac-cessed Apr 12,. 2017.18.Kim JW., Jeong YY., Chang NK., Heo SH., Shin SS., Lee JH, et al. Perfusion CT in colorectal cancer: comparison of perfusion parameters with tumor grade and microvessel density. Ko-rean J Radiol. 2012. 13(Suppl 1):S89–S97.
Article19.Russell AH., Harris J., Rosenberg PJ., Sause WT., Fisher BJ., Hoff-man JP, et al. Anal sphincter conservation for patients with adenocarcinoma of the distal rectum: long-term results of radiation therapy oncology group protocol 89-02. Int J Ra-diat Oncol Biol Phys. 2000. 46:313–322.
Article20.Mohiuddin M., Marks G., Bannon J. High-dose preoperative radiation and full thickness local excision: a new option for selected T3 distal rectal cancers. Int J Radiat Oncol Biol Phys. 1994. 30:845–849.
Article21.Gertler R., Rosenberg R., Schuster T., Friess H. Defining a high-risk subgroup with colon cancer stages I and II for possible adjuvant therapy. Eur J Cancer. 2009. 45:2992–2999.
Article22.Horton KM., Abrams RA., Fishman EK. Spiral CT of colon can-cer: imaging features and role in management. Radiograph-ics. 2000. 20:419–430.
Article23.Kim JE., Lee JM., Baek JH., Moon SK., Kim SH., Han JK, et al. Dif-ferentiation of poorly differentiated colorectal adenocarci-nomas from well- or moderately differentiated colorectal adenocarcinomas at contrast-enhanced multidetector CT. Abdom Imaging. 2015. 40:1–10.
Article24.Filippone A., Ambrosini R., Fuschi M., Marinelli T., Genovesi D., Bonomo L. Preoperative T and N staging of colorectal can-cer: accuracy of contrast-enhanced multi-detector row CT colonography--initial experience. Radiology. 2004. 231:83–90.
Article25.Sica GT., Ji H., Ros PR. CT and MR imaging of hepatic metas-tases. AJR Am J Roentgenol. 2000. 174:691–698.
Article26.Chung WS., Kim MJ., Chung YE., Kim YE., Park MS., Choi JY, et al. Comparison of gadoxetic acid-enhanced dynamic imaging and diffusion-weighted imaging for the preoperative evaluation of colorectal liver metastases. J Magn Reson Im-aging. 2011. 34:345–353.
Article27.Seo HJ., Kim MJ., Lee JD., Chung WS., Kim YE. Gadoxetate di-sodium-enhanced magnetic resonance imaging versus contrast-enhanced 18F-fluorodeoxyglucose positron emission tomography/computed tomography for the detection of colorectal liver metastases. Invest Radiol. 2011. 46:548–555.
Article28.Adam R., de Gramont A., Figueras J., Kokudo N., Kunstlinger F., Loyer E, et al. Managing synchronous liver metastases from colorectal cancer: a multidisciplinary international consen-sus. Cancer Treat Rev. 2015. 41:729–741.
Article29.Goh V., Glynne-Jones R. Perfusion CT imaging of colorectal cancer. Br J Radiol. 2014. 87:20130811.
Article30.Weiss L., Grundmann E., Torhorst J., Hartveit F., Moberg I., Eder M, et al. Haematogenous metastatic patterns in colonic carcinoma: an analysis of 1541 necropsies. J Pathol. 1986. 150:195–203.31.Viadana E., Bross ID., Pickren JW. The metastatic spread of cancers of the digestive system in man. Oncology. 1978. 35:114–126.
Article32.Weiss L., Voit A., Lane WW. Metastatic patterns in patients with carcinomas of the lower esophagus and upper rectum. Invasion Metastasis. 1984. 4:47–60.33.Mantke R., Schmidt U., Wolff S., Kube R., Lippert H. Incidence of synchronous liver metastases in patients with colorectal cancer in relationship to clinico-pathologic characteristics. Results of a German prospective multicentre observational study. Eur J Surg Oncol. 2012. 38:259–265.
Article34.Kang KA., Jang KM., Kim SH., Kang TW., Cha DI. Risk factor as-sessment to predict the likelihood of a diagnosis of metas-tasis for indeterminate hepatic lesions found at computed tomography in patients with rectal cancer. Clin Radiol. 2017. 72:473–481.
Article35.Jones EC., Chezmar JL., Nelson RC., Bernardino ME. The fre-quency and significance of small (less than or equal to 15 mm) hepatic lesions detected by CT. AJR Am J Roentgenol. 1992. 158:535–539.
Article36.Krakora GA., Coakley FV., Williams G., Yeh BM., Breiman RS., Qayyum A. Small hypoattenuating hepatic lesions at contrast-enhanced CT: prognostic importance in patients with breast cancer. Radiology. 2004. 233:667–673.
Article37.Goshima S., Kanematsu M., Watanabe H., Kondo H., Shiratori Y., Onozuka M, et al. Hepatic hemangioma and metastasis: differentiation with gadoxetate disodium-enhanced 3-T MRI. AJR Am J Roentgenol. 2010. 195:941–946.
Article38.Kim YK., Park G., Kim CS., Yu HC., Han YM. Diagnostic efficacy of gadoxetic acid-enhanced MRI for the detection and char-acterisation of liver metastases: comparison with multide-tector-row CT. Br J Radiol. 2012. 85:539–547.
Article39.Böttcher J., Hansch A., Pfeil A., Schmidt P., Malich A., Schnee-weiss A, et al. Detection and classification of different liver lesions: comparison of Gd-EOB-DTPA-enhanced MRI versus multiphasic spiral CT in a clinical single centre investigation. Eur J Radiol. 2013. 82:1860–1869.
Article