J Pathol Transl Med.
2017 May;51(3):292-305. 10.4132/jptm.2017.02.17.
An Experimental Infarct Targeting the Internal Capsule: Histopathological and Ultrastructural Changes
- Affiliations
-
- 1Department of Pathology, Chonnam National University Medical School and Research Institute of Medical Sciences, Gwangju, Korea. mclee@jnu.ac.kr
- 2Department of Forensic Medicine, Chonnam National University Medical School and Research Institute of Medical Sciences, Gwangju, Korea.
- 3Department of Medical System Engineering and School of Mechatronics, Gwangju Institute of Science and Technology, Gwangju, Korea.
- KMID: 2392596
- DOI: http://doi.org/10.4132/jptm.2017.02.17
Abstract
- BACKGROUND
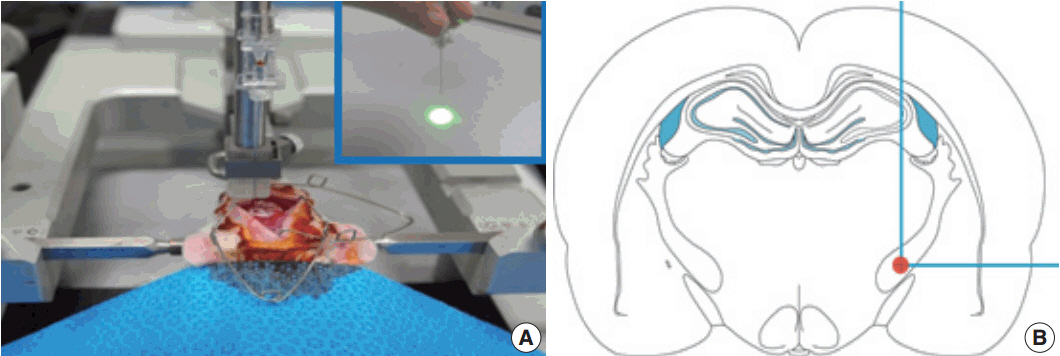
Stroke involving the cerebral white matter (WM) has increased in prevalence, but most experimental studies have focused on ischemic injury of the gray matter. This study was performed to investigate the WM in a unique rat model of photothrombotic infarct targeting the posterior limb of internal capsule (PLIC), focusing on the identification of the most vulnerable structure in WM by ischemic injury, subsequent glial reaction to the injury, and the fundamental histopathologic feature causing different neurologic outcomes.
METHODS
Light microscopy with immunohistochemical stains and electron microscopic examinations of the lesion were performed between 3 hours and 21 days post-ischemic injury.
RESULTS
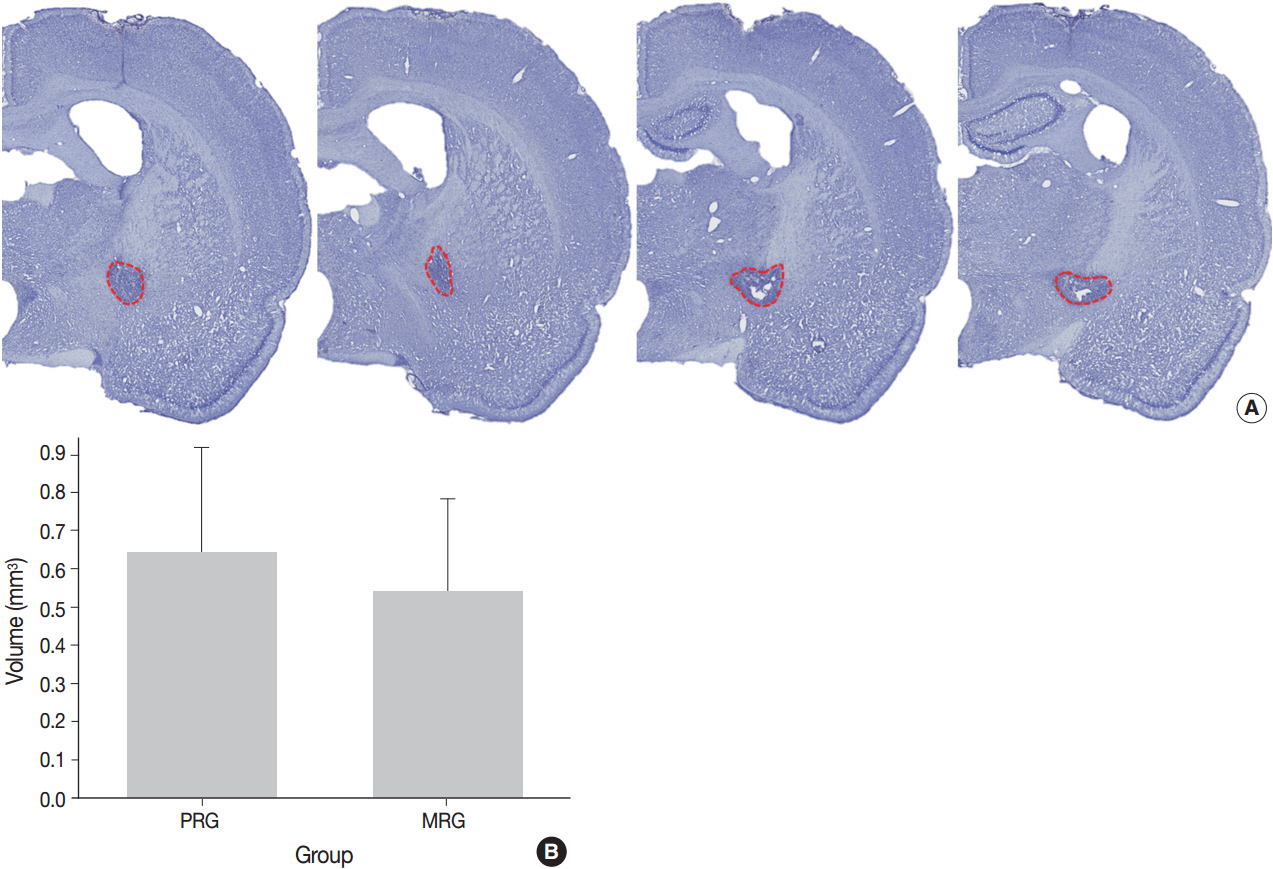
Initial pathological change develops in myelinated axon, concomitantly with reactive change of astrocytes. The first pathology to present is nodular loosening to separate the myelin sheath with axonal wrinkling. Subsequent pathologies include rupture of the myelin sheath with extrusion of axonal organelles, progressive necrosis, oligodendrocyte degeneration and death, and reactive gliosis. Increase of glial fibrillary acidic protein (GFAP) immunoreactivity is an early event in the ischemic lesion. WM pathologies result in motor dysfunction. Motor function recovery after the infarct was correlated to the extent of PLIC injury proper rather than the infarct volume.
CONCLUSIONS
Pathologic changes indicate that the cerebral WM, independent of cortical neurons, is highly vulnerable to the effects of focal ischemia, among which myelin sheath is first damaged. Early increase of GFAP immunoreactivity indicates that astrocyte response initially begins with myelinated axonal injury, and supports the biologic role related to WM injury or plasticity. The reaction of astrocytes in the experimental model might be important for the study of pathogenesis and treatment of the WM stroke.
Keyword
MeSH Terms
-
Astrocytes
Axons
Coloring Agents
Extremities
Glial Fibrillary Acidic Protein
Gliosis
Gray Matter
Internal Capsule*
Ischemia
Microscopy
Models, Animal
Models, Theoretical
Myelin Sheath
Necrosis
Neurons
Oligodendroglia
Organelles
Pathology
Plastics
Prevalence
Recovery of Function
Rupture
Stroke
White Matter
Coloring Agents
Glial Fibrillary Acidic Protein
Plastics
Figure
Reference
-
1. Schneider AT, Kissela B, Woo D, et al. Ischemic stroke subtypes: a population-based study of incidence rates among blacks and whites. Stroke. 2004; 35:1552–6.2. Hachinski VC, Potter P, Merskey H. Leuko-araiosis. Arch Neurol. 1987; 44:21–3.
Article3. Matsusue E, Sugihara S, Fujii S, Ohama E, Kinoshita T, Ogawa T. White matter changes in elderly people: MR-pathologic correlations. Magn Reson Med Sci. 2006; 5:99–104.
Article4. Fu JH, Lu CZ, Hong Z, Dong Q, Luo Y, Wong KS. Extent of white matter lesions is related to acute subcortical infarcts and predicts further stroke risk in patients with first ever ischaemic stroke. J Neurol Neurosurg Psychiatry. 2005; 76:793–6.
Article5. Kim GM, Park KY, Avery R, et al. Extensive leukoaraiosis is associated with high early risk of recurrence after ischemic stroke. Stroke. 2014; 45:479–85.
Article6. Bailey EL, McCulloch J, Sudlow C, Wardlaw JM. Potential animal models of lacunar stroke: a systematic review. Stroke. 2009; 40:e451–8.7. He SQ, Dum RP, Strick PL. Topographic organization of corticospinal projections from the frontal lobe: motor areas on the lateral surface of the hemisphere. J Neurosci. 1993; 13:952–80.
Article8. Lee MC, Jin CY, Kim HS, et al. Stem cell dynamics in an experimental model of stroke. Chonnam Med J. 2011; 47:90–8.
Article9. Song S, Park JT, Na JY, et al. Early expressions of hypoxia-inducible factor 1alpha and vascular endothelial growth factor increase the neuronal plasticity of activated endogenous neural stem cells after focal cerebral ischemia. Neural Regen Res. 2014; 9:912–8.
Article10. Jang JW, Lee JK, Lee MC, Piao MS, Kim SH, Kim HS. Melatonin reduced the elevated matrix metalloproteinase-9 level in a rat photothrombotic stroke model. J Neurol Sci. 2012; 323:221–7.
Article11. Liebigt S, Schlegel N, Oberland J, Witte OW, Redecker C, Keiner S. Effects of rehabilitative training and anti-inflammatory treatment on functional recovery and cellular reorganization following stroke. Exp Neurol. 2012; 233:776–82.
Article12. Bush EC, Allman JM. The scaling of white matter to gray matter in cerebellum and neocortex. Brain Behav Evol. 2003; 61:1–5.
Article13. Kudo T, Takeda M, Tanimukai S, Nishimura T. Neuropathologic changes in the gerbil brain after chronic hypoperfusion. Stroke. 1993; 24:259–64.
Article14. He Z, Yamawaki T, Yang S, Day AL, Simpkins JW, Naritomi H. Experimental model of small deep infarcts involving the hypothalamus in rats: changes in body temperature and postural reflex. Stroke. 1999; 30:2743–51.15. Hughes PM, Anthony DC, Ruddin M, et al. Focal lesions in the rat central nervous system induced by endothelin-1. J Neuropathol Exp Neurol. 2003; 62:1276–86.
Article16. Frost SB, Barbay S, Mumert ML, Stowe AM, Nudo RJ. An animal model of capsular infarct: endothelin-1 injections in the rat. Behav Brain Res. 2006; 169:206–11.
Article17. Whitehead SN, Hachinski VC, Cechetto DF. Interaction between a rat model of cerebral ischemia and beta-amyloid toxicity: inflammatory responses. Stroke. 2005; 36:107–12.18. Souza-Rodrigues RD, Costa AM, Lima RR, Dos Santos CD, Picanco-Diniz CW, Gomes-Leal W. Inflammatory response and white matter damage after microinjections of endothelin-1 into the rat striatum. Brain Res. 2008; 1200:78–88.
Article19. Tanaka Y, Imai H, Konno K, et al. Experimental model of lacunar infarction in the gyrencephalic brain of the miniature pig: neurological assessment and histological, immunohistochemical, and physiological evaluation of dynamic corticospinal tract deformation. Stroke. 2008; 39:205–12.20. Shibata M, Ohtani R, Ihara M, Tomimoto H. White matter lesions and glial activation in a novel mouse model of chronic cerebral hypoperfusion. Stroke. 2004; 35:2598–603.
Article21. Kim HS, Kim D, Kim RG, et al. A rat model of photothrombotic capsular infarct with a marked motor deficit: a behavioral, histologic, and microPET study. J Cereb Blood Flow Metab. 2014; 34:683–9.
Article22. Kim HS, Park MS, Lee JK, Kim HJ, Park JT, Lee MC. Time point expression of apoptosis regulatory proteins in a photochemically-induced focal cerebral ischemic rat brain. Chonnam Med J. 2011; 47:144–9.
Article23. Swanson RA, Morton MT, Tsao-Wu G, Savalos RA, Davidson C, Sharp FR. A semiautomated method for measuring brain infarct volume. J Cereb Blood Flow Metab. 1990; 10:290–3.
Article24. Lee KH, Kang KJ, Moon KS, et al. Prognostic significance of neuronal marker expression in glioblastomas. Childs Nerv Syst. 2012; 28:1879–86.
Article25. Kim JM, Lee TH, Lee MC, et al. Endoneurial microangiopathy of sural nerve in experimental vacor-induced diabetes. Ultrastruct Pathol. 2002; 26:393–401.
Article26. Donovan V, Kim C, Anugerah AK, et al. Repeated mild traumatic brain injury results in long-term white-matter disruption. J Cereb Blood Flow Metab. 2014; 34:715–23.
Article27. Bashkatov AN, Genina EA, Tuchin VV. Tissue optical properties. In : Boas DA, Pitris C, Ramanujam N, editors. Handbook of biomedical optics. Boca Raton: CRC Press;2011. p. 67–100.28. Silver J, Miller JH. Regeneration beyond the glial scar. Nat Rev Neurosci. 2004; 5:146–56.
Article29. Franke H, Verkhratsky A, Burnstock G, Illes P. Pathophysiology of astroglial purinergic signalling. Purinergic Signal. 2012; 8:629–57.
Article30. Sato Y, Chin Y, Kato T, et al. White matter activated glial cells produce BDNF in a stroke model of monkeys. Neurosci Res. 2009; 65:71–8.
Article31. Carrera E, Tononi G. Diaschisis: past, present, future. Brain. 2014; 137(Pt 9):2408–22.
Article32. Fulmer CG, VonDran MW, Stillman AA, Huang Y, Hempstead BL, Dreyfus CF. Astrocyte-derived BDNF supports myelin protein synthesis after cuprizone-induced demyelination. J Neurosci. 2014; 34:8186–96.
Article33. Miyamoto N, Maki T, Shindo A, et al. Astrocytes promote oligodendrogenesis after white matter damage via brain-derived neurotrophic factor. J Neurosci. 2015; 35:14002–8.
Article34. Ramos-Cejudo J, Gutiérrez-Fernández M, Otero-Ortega L, et al. Brain-derived neurotrophic factor administration mediated oligodendrocyte differentiation and myelin formation in subcortical ischemic stroke. Stroke. 2015; 46:221–8.
Article35. Sozmen EG, Kolekar A, Havton LA, Carmichael ST. A white matter stroke model in the mouse: axonal damage, progenitor responses and MRI correlates. J Neurosci Methods. 2009; 180:261–72.
Article36. He Z, Yang SH, Naritomi H, et al. Definition of the anterior choroidal artery territory in rats using intraluminal occluding technique. J Neurol Sci. 2000; 182:16–28.
Article37. Karadottir R, Cavelier P, Bergersen LH, Attwell D. NMDA receptors are expressed in oligodendrocytes and activated in ischaemia. Nature. 2005; 438:1162–6.
Article38. Bakiri Y, Hamilton NB, Káradóttir R, Attwell D. Testing NMDA receptor block as a therapeutic strategy for reducing ischaemic damage to CNS white matter. Glia. 2008; 56:233–40.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Precision Capsular Infarct Modeling to Produce Hand Motor Deficits in Cynomolgus Macaques
- Effects of the Rheomacrodex and Alteration of Blood Pressure after Experimental Occlusion of the Middle Cerebral Artery
- The Open Artery Hypothesis
- The Usefulness of Diffusion Tensor MRI for the Prediction of Clinical Outcome in Patients with Acute Subcortical Infarction
- Three-axis Modification of Coordinates Enables Accurate Stereotactic Targeting in Non-human Primate Brains of Different Sizes