Infect Chemother.
2013 Mar;45(1):1-10.
Revolutionising Bacteriology to Improve Treatment Outcomes and Antibiotic Stewardship
- Affiliations
-
- 1Norwich Medical School, University of East Anglia, Norwich, Norfolk, NR4 7TJ, United Kingdom. d.livermore@uea.ac.uk
Abstract
- Laboratory investigation of bacterial infections generally takes two days: one to grow the bacteria and another to identify them and to test their susceptibility. Meanwhile the patient is treated empirically, based on likely pathogens and local resistance rates. Many patients are over-treated to prevent under-treatment of a few, compromising antibiotic stewardship. Molecular diagnostics have potential to improve this situation by accelerating precise diagnoses and the early refinement of antibiotic therapy. They include: (i) the use of 'biomarkers' to swiftly distinguish patients with bacterial infection, and (ii) molecular bacteriology to identify pathogens and their resistance genes in clinical specimens, without culture. Biomarker interest centres on procalcitonin, which has given good results particularly for pneumonias, though broader biomarker arrays may prove superior in the future. PCRs already are widely used to diagnose a few infections (e.g. tuberculosis) whilst multiplexes are becoming available for bacteraemia, pneumonia and gastrointestinal infection. These detect likely pathogens, but are not comprehensive, particularly for resistance genes; there is also the challenge of linking pathogens and resistance genes when multiple organisms are present in a sample. Next-generation sequencing offers more comprehensive profiling, but obstacles include sensitivity when the bacterial load is low, as in bacteraemia, and the imperfect correlation of genotype and phenotype. In short, rapid molecular bacteriology presents great potential to improve patient treatments and antibiotic stewardship but faces many technical challenges; moreover it runs counter to the current nostrum of defining resistance in pharmacodynamic terms, rather than by the presence of a mechanism, and the policy of centralising bacteriology services.
MeSH Terms
Figure
Reference
-
1. Croxatto A, Prod'hom G, Greub G. Applications of MALDI-TOF mass spectrometry in clinical diagnostic microbiology. FEMS Microbiol Rev. 2012. 36:380–407.
Article2. Pallen MJ, Loman NJ. Are diagnostic and public health bacteriology ready to become branches of genomic medicine? Genome Med. 2011. 3:53.
Article3. Spanu T, Sanguinetti M, Ciccaglione D, D'Inzeo T, Romano L, Leone F, Fadda G. Use of the VITEK 2 system for rapid identification of clinical isolates of staphylococci from bloodstream infections. J Clin Microbiol. 2003. 41:4259–4263.
Article4. Trenholme GM, Kaplan RL, Karakusis PH, Stine T, Fuhrer J, Landau W, Levin S. Clinical impact of rapid identification and susceptibility testing of bacterial blood culture isolates. J Clin Microbiol. 1989. 27:1342–1345.
Article5. Bruins M, Oord H, Bloembergen P, Wolfhagen M, Casparie A, Degener J, Ruijs G. Lack of effect of shorter turnaround time of microbiological procedures on clinical outcomes: a randomised controlled trial among hospitalised patients in the Netherlands. Eur J Clin Microbiol Infect Dis. 2005. 24:305–313.
Article6. Radford AD, Chapman D, Dixon L, Chantrey J, Darby AC, Hall N. Application of next-generation sequencing technologies in virology. J Gen Virol. 2012. 93:1853–1868.
Article7. MacGowan AP, Bowker KE, Bennett PM, Lovering AM. Surveillance of antimicrobial resistance. Lancet. 1998. 352:1783.
Article8. McNulty CA, Richards J, Livermore DM, Little P, Charlett A, Freeman E, Harvey I, Thomas M. Clinical relevance of laboratory-reported antibiotic resistance in acute uncomplicated urinary tract infection in primary care. J Antimicrob Chemother. 2006. 58:1000–1008.
Article9. Dickinson JD, Kollef MH. Early and adequate antibiotic therapy in the treatment of severe sepsis and septic shock. Curr Infect Dis Rep. 2011. 13:399–405.
Article10. Kumar A, Roberts D, Wood KE, Light B, Parrillo JE, Sharma S, Suppes R, Feinstein D, Zanotti S, Taiberg L, Gurka D, Kumar A, Cheang M. Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Crit Care Med. 2006. 34:1589–1596.
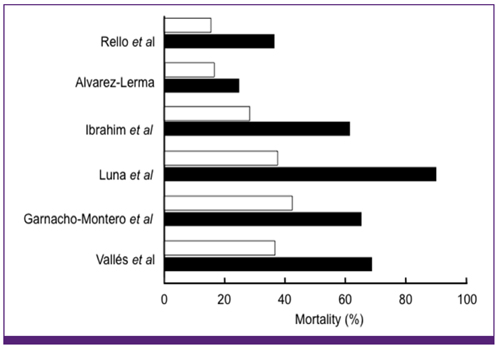
Article11. Rello J, Gallego M, Mariscal D, Soñora R, Valles J. The value of routine microbial investigation in ventilator-associated pneumonia. Am J Respir Crit Care Med. 1997. 156:196–200.
Article12. Alvarez-Lerma F. ICU-Acquired Pneumonia Study Group. Modification of empiric antibiotic treatment in patients with pneumonia acquired in the intensive care unit. Intensive Care Med. 1996. 22:387–394.
Article13. Ibrahim EH, Sherman G, Ward S, Fraser VJ, Kollef MH. The influence of inadequate antimicrobial treatment of bloodstream infections on patient outcomes in the ICU setting. Chest. 2000. 118:146–155.
Article14. Luna CM, Vujacich P, Niederman MS, Vay C, Gherardi C, Matera J, Jolly EC. Impact of BAL data on the therapy and outcome of ventilator-associated pneumonia. Chest. 1997. 111:676–685.
Article15. Garnacho-Montero J, Garcia-Garmendia JL, Barrero-Almodovar A, Jimenez-Jimenez FJ, Perez-Paredes C, Ortiz-Leyba C. Impact of adequate empirical antibiotic therapy on the outcome of patients admitted to the intensive care unit with sepsis. Crit Care Med. 2003. 31:2742–2751.
Article16. Vallés J, Rello J, Ochagavía A, Garnacho J, Alcalá MA. Community-acquired bloodstream infection in critically ill adult patients: impact of shock and inappropriate antibiotic therapy on survival. Chest. 2003. 123:1615–1624.
Article17. Kollef MH. Inadequate antimicrobial treatment: an important determinant of outcome for hospitalized patients. Clin Infect Dis. 2000. 31:Suppl 4. S131–S138.
Article18. Schwaber MJ, Carmeli Y. Mortality and delay in effective therapy associated with extended-spectrum beta-lactamase production in Enterobacteriaceae bacteraemia: a systematic review and meta-analysis. J Antimicrob Chemother. 2007. 60:913–920.
Article19. Peralta G, Sánchez MB, Garrido JC, De Benito I, Cano ME, Martínez-Martínez L, Roiz MP. Impact of antibiotic resistance and of adequate empirical antibiotic treatment in the prognosis of patients with Escherichia coli bacteraemia. J Antimicrob Chemother. 2007. 60:855–863.
Article20. File TM Jr. Recommendations for treatment of hospital-acquired and ventilator-associated pneumonia: review of recent international guidelines. Clin Infect Dis. 2010. 51:Suppl 1. S42–S47.
Article21. Soo Hoo GW, Wen YE, Nguyen TV, Goetz MB. Impact of clinical guidelines in the management of severe hospital-acquired pneumonia. Chest. 2005. 128:2778–2787.
Article22. Theuretzbacher U. Accelerating resistance, inadequate antibacterial drug pipelines and international responses. Int J Antimicrob Agents. 2012. 39:295–299.
Article23. Piddock LJ. The crisis of no new antibiotics--what is the way forward? Lancet Infect Dis. 2012. 12:249–253.
Article24. White AR. BSAC Working Party on The Urgent Need: Regenerating Antibacterial Drug Discovery and Development. Effective antibacterials: at what cost? The economics of antibacterial resistance and its control. J Antimicrob Chemother. 2011. 66:1948–1953.
Article25. Livermore DM. Current epidemiology and growing resistance of gram-negative pathogens. Korean J Intern Med. 2012. 27:128–142.
Article26. Livermore DM, Warner M, Mushtaq S, Doumith M, Zhang J, Woodford N. What remains against carbapenem-resistant Enterobacteriaceae? Evaluation of chloramphenicol, ciprofloxacin, colistin, fosfomycin, minocycline, nitrofurantoin, temocillin and tigecycline. Int J Antimicrob Agents. 2011. 37:415–419.
Article27. Köser CU, Ellington MJ, Cartwright EJ, Gillespie SH, Brown NM, Farrington M, Holden MT, Dougan G, Bentley SD, Parkhill J, Peacock SJ. Routine use of microbial whole genome sequencing in diagnostic and public health microbiology. PLoS Pathog. 2012. 8:e1002824.
Article28. Dunne WM Jr, Westblade LF, Ford B. Next-generation and whole-genome sequencing in the diagnostic clinical microbiology laboratory. Eur J Clin Microbiol Infect Dis. 2012. 31:1719–1726.
Article29. Krüger S, Welte T. Biomarkers in community-acquired pneumonia. Expert Rev Respir Med. 2012. 6:203–214.
Article30. Yo CH, Hsieh PS, Lee SH, Wu JY, Chang SS, Tasi KC, Lee CC. Comparison of the test characteristics of procalcitonin to Creactive protein and leukocytosis for the detection of serious bacterial infections in children presenting with fever without source: a systematic review and meta-analysis. Ann Emerg Med. 2012. 60:591–600.
Article31. Reinhart K, Hartog CS. Biomarkers as a guide for antimicrobial therapy. Int J Antimicrob Agents. 2010. 36:Suppl 2. S17–S21.
Article32. Soni NJ, Samson DJ, Galaydick JL, Vats V, Pitrak DL, Aronson N. Effective health care program: Procalcitonin-guided antibiotic therapy. Publication No. 12(13)-EHC124-EF. 2012. Rockville, MD: Agency for Healthcare Research and Quality.33. Saeed K, Dryden M, Bourne S, Paget C, Proud A. Reduction in antibiotic use through procalcitonin testing in patients in the medical admission unit or intensive care unit with suspicion of infection. J Hosp Infect. 2011. 78:289–292.
Article34. Sakran JV, Michetti CP, Sheridan MJ, Richmond R, Waked T, Aldaghlas T, Rizzo A, Griffen M, Fakhry SM. The utility of procalcitonin in critically ill trauma patients. J Trauma Acute Care Surg. 2012. 73:413–418.
Article35. Yu CW, Juan LI, Wu MH, Shen CJ, Wu JY, Lee CC. Systematic review and meta-analysis of the diagnostic accuracy of procalcitonin, C-reactive protein and white blood cell count for suspected acute appendicitis. Br J Surg. 2013. 100:322–329.
Article36. Almansa R, Socias L, Sanchez-Garcia M, Martín-Loeches I, del Olmo M, Andaluz-Ojeda D, Bobillo F, Rico L, Herrero A, Roig V, San-Jose CA, Rosich S, Barbado J, Disdier C, de Lejarazu RO, Gallegos MC, Fernandez V, Bermejo-Martin JF. Critical COPD respiratory illness is linked to increased transcriptomic activity of neutrophil proteases genes. BMC Res Notes. 2012. 5:401.
Article37. Andaluz-Ojeda D, Bobillo F, Iglesias V, Almansa R, Rico L, Gandía F, Resino S, Tamayo E, de Lejarazu RO, Bermejo-Martin JF. A combined score of pro- and anti-inflammatory interleukins improves mortality prediction in severe sepsis. Cytokine. 2012. 57:332–336.
Article38. Gaydos CA. Nucleic acid amplification tests for gonorrhea and chlamydia: practice and applications. Infect Dis Clin North Am. 2005. 19:367–386. ix
Article39. Lawn SD, Nicol MP. Xpert® MTB/RIF assay: development, evaluation and implementation of a new rapid molecular diagnostic for tuberculosis and rifampicin resistance. Future Microbiol. 2011. 6:1067–1082.
Article40. Wilson ML. Recent advances in the laboratory detection of Mycobacterium tuberculosis complex and drug resistance. Clin Infect Dis. 2011. 52:1350–1355.
Article41. Polisena J, Chen S, Cimon K, McGill S, Forward K, Gardam M. Clinical effectiveness of rapid tests for methicillin resistant Staphylococcus aureus (MRSA) in hospitalized patients: a systematic review. BMC Infect Dis. 2011. 11:336.42. Oh AC, Lee JK, Lee HN, Hong YJ, Chang YH, Hong SI, Kim DH. Clinical utility of the Xpert MRSA assay for early detection of methicillin-resistant Staphylococcus aureus. Mol Med Report. 2012. [Epub ahead of print].
Article43. Cercenado E, Marín M, Burillo A, Martín-Rabadán P, Rivera M, Bouza E. Rapid detection of Staphylococcus aureus in lower respiratory tract secretions from patients with suspected ventilator-associated pneumonia: evaluation of the Cepheid Xpert MRSA/SA SSTI assay. J Clin Microbiol. 2012. 50:4095–4097.
Article44. Spencer DH, Sellenriek P, Burnham CA. Validation and implementation of the GeneXpert MRSA/SA blood culture assay in a pediatric setting. Am J Clin Pathol. 2011. 136:690–694.
Article45. Benzie A, Alexander S, Gill N, Greene L, Thomas S, Ison C. Gonococcal NAATs: what is the current state of play in England and Wales? Int J STD AIDS. 2010. 21:246–248.
Article46. Blanc DS, Basset P, Nahimana-Tessemo I, Jaton K, Greub G, Zanetti G. High proportion of wrongly identified methicillin-resistant Staphylococcus aureus carriers by use of a rapid commercial PCR assay due to presence of staphylococcal cassette chromosome element lacking the mecA gene. J Clin Microbiol. 2011. 49:722–724.
Article47. Seth-Smith HM, Harris SR, Persson K, Marsh P, Barron A, Bignell A, Bjartling C, Clark L, Cutcliffe LT, Lambden PR, Lennard N, Lockey SJ, Quail MA, Salim O, Skilton RJ, Wang Y, Holland MJ, Parkhill J, Thomson NR, Clarke IN. Co-evolution of genomes and plasmids within Chlamydia trachomatis and the emergence in Sweden of a new variant strain. BMC Genomics. 2009. 10:239.
Article48. Poritz MA, Blaschke AJ, Byington CL, Meyers L, Nilsson K, Jones DE, Thatcher SA, Robbins T, Lingenfelter B, Amiott E, Herbener A, Daly J, Dobrowolski SF, Teng DH, Ririe KM. FilmArray, an automated nested multiplex PCR system for multi-pathogen detection: development and application to respiratory tract infection. PLoS One. 2011. 6:e26047.
Article49. Leitner E, Kessler HH, Spindelboeck W, Hoenigl M, Putz-Bankuti C, Stadlbauer-Köllner V, Krause R, Grisold AJ, Feierl G, Stauber RE. Comparison of two molecular assays with conventional blood culture for diagnosis of sepsis. J Microbiol Methods. 2013. 92:253–255.
Article50. Guido M, Quattrocchi M, Zizza A, Pasanisi G, Pavone V, Lobreglio G, Gabutti G, De Donno A. Molecular approaches in the diagnosis of sepsis in neutropenic patients with haematological malignances. J Prev Med Hyg. 2012. 53:104–108.51. Paolucci M, Stanzani M, Melchionda F, Tolomelli G, Castellani G, Landini MP, Varani S, Lewis RE, Sambri V. Routine use of a real-time polymerase chain reaction method for detection of bloodstream infections in neutropaenic patients. Diagn Microbiol Infect Dis. 2013. 75:130–134.
Article52. Lodes U, Bohmeier B, Lippert H, König B, Meyer F. PCR-based rapid sepsis diagnosis eff ectively guides clinical treatment in patients with new onset of SIRS. Langenbecks Arch Surg. 2012. 397:447–455.
Article53. Alvarez J, Mar J, Varela-Ledo E, Garea M, Matinez-Lamas L, Rodriguez J, Regueiro B. Cost analysis of real-time polymerase chain reaction microbiological diagnosis in patients with septic shock. Anaesth Intensive Care. 2012. 40:958–963.
Article54. Dark P, Wilson C, Blackwood B, McAuley DF, Perkins GD, McMullan R, Gates S, Warhurst G. Accuracy of LightCycler(R) SeptiFast for the detection and identification of pathogens in the blood of patients with suspected sepsis: a systematic review protocol. BMJ Open. 2012. 2:e000392.55. Dunne WM Jr, Westblade LF, Ford B. Next-generation and whole-genome sequencing in the diagnostic clinical microbiology laboratory. Eur J Clin Microbiol Infect Dis. 2012. 31:1719–1726.
Article56. Maitra RD, Kim J, Dunbar WB. Recent advances in nanopore sequencing. Electrophoresis. 2012. 33:3418–3428.
Article57. Harris SR, Cartwright EJ, Török ME, Holden MT, Brown NM, Ogilvy-Stuart AL, Ellington MJ, Quail MA, Bentley SD, Parkhill J, Peacock SJ. Whole-genome sequencing for analysis of an outbreak of meticillin-resistant Staphylococcus aureus: a descriptive study. Lancet Infect Dis. 2013. 13:130–136.
Article58. Snitkin ES, Zelazny AM, Thomas PJ, Stock F, Henderson DK, Palmore TN, Segre JA. NISC Comparative Sequencing Program Group. Tracking a hospital outbreak of carbapenem-resistant Klebsiella pneumonia with whole-genome sequencing. Sci Transl Med. 2012. 4:148ra116.59. Babini GS, Yuan M, Hall LM, Livermore DM. Variable susceptibility to piperacillin/tazobactam amongst Klebsiella spp. with extended-spectrum beta-lactamases. J Antimicrob Chemother. 2003. 51:605–612.
Article60. Allen VG, Farrell DJ, Rebbapragada A, Tan J, Tijet N, Perusini SJ, Towns L, Lo S, Low DE, Melano RG. Molecular analysis of antimicrobial resistance mechanisms in Neisseria gonorrhoeae isolates from Ontario, Canada. Antimicrob Agents Chemother. 2011. 55:703–712.
Article61. Tomás M, Doumith M, Warner M, Turton JF, Beceiro A, Bou G, Livermore DM, Woodford N. Efflux pumps, OprD porin, AmpC beta-lactamase, and multiresistance in Pseudomonas aeruginosa isolates from cystic fibrosis patients. Antimicrob Agents Chemother. 2010. 54:2219–2224.
Article62. Mouton JW, Brown DF, Apfalter P, Cantón R, Giske CG, Ivanova M, MacGowan AP, Rodloff A, Soussy CJ, Steinbakk M, Kahlmeter G. The role of pharmacokinetics/pharmacodynamics in setting clinical MIC breakpoints: the EUCAST approach. Clin Microbiol Infect. 2012. 18:E37–E45.
Article63. Guido M, Quattrocchi M, Zizza A, Pasanisi G, Pavone V, Lobreglio G, Gabutti G, De Donno A. Molecular approaches in the diagnosis of sepsis in neutropenic patients with haematological malignances. J Prev Med Hyg. 2012. 53:104–108.64. File TM Jr, Low DE, Eckburg PB, Talbot GH, Friedland HD, Lee J, Llorens L, Critchley IA, Thye DA. FOCUS 1 investigators. FOCUS 1: a randomized, double-blinded, multicentre, Phase III trial of the efficacy and safety of ceftaroline fosamil versus ceftriaxone in community-acquired pneumonia. J Antimicrob Chemother. 2011. 66:Suppl 3. iii19–iii32.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Antibiotic Stewardship: A Key Strategy to Combat Antibiotic Resistance
- Clinical Utility of Procalcitonin on Antibiotic Stewardship: A Narrative Review
- Current Status of Antibiotic Stewardship and the Role of Biomarkers in Antibiotic Stewardship Programs
- Optimal antimicrobial therapy and antimicrobial stewardship in sepsis and septic shock
- Participation in and perceptions of antibiotic stewardship behaviors among nurses at a children’s hospital in South Korea: a descriptive study