J Rheum Dis.
2017 Aug;24(4):192-202. 10.4078/jrd.2017.24.4.192.
Metabolomics Approach to Explore the Effects of Rebamipide on Inflammatory Arthritis Using Ultra Performance Liquid Chromatography/Quadrupole Time-of-Flight Mass Spectrometry
- Affiliations
-
- 1Division of Rheumatology, Department of Internal Medicine, College of Medicine, The Catholic University of Korea, Seoul, Korea. min6403@catholic.ac.kr
- 2Department of Medical Records and Health Information Management, College of Nursing and Health, Kongju National University, Gongju, Korea.
- 3Molecular Recognition Research Center, Korea Institute of Science and Technology, Seoul, Korea. jbhluck@kist.re.kr
- KMID: 2389060
- DOI: http://doi.org/10.4078/jrd.2017.24.4.192
Abstract
OBJECTIVE
Rebampide is a gastroprotective agent used to treat gastritis. It possesses anti-inflammatory and anti-arthritis effects, but the mechanisms of these effects are not well understood. The objective of this study was to explore mechanisms underlying the therapeutic effects of rebamipide in inflammatory arthritis.
METHODS
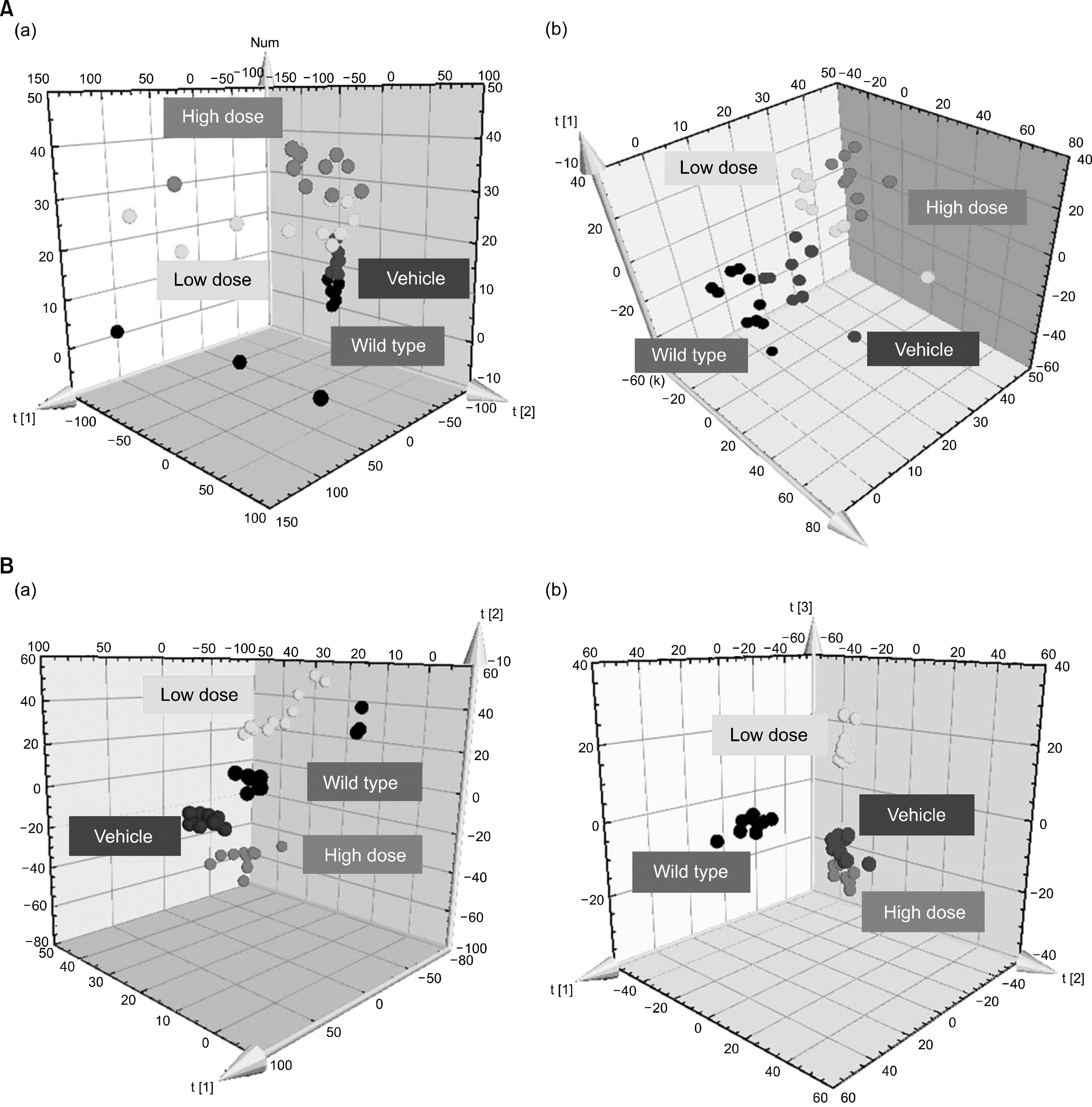
Collagen-induced arthritis (CIA) was induced in DBA/1J mice. DBA/1J mice were immunized with chicken type II collagen, then treated intraperitoneally with rebamipide (10 mg/kg or 30 mg/kg) or vehicle (10% carboxymethylcellulose solution) alone. Seven weeks later, plasma samples were collected. Plasma metabolic profiles were analyzed using ultra performance liquid chromatography/quadrupole time-of-flight mass spectrometry-based metabolomics study and metabolite biomarkers were identified through multivariate data analysis.
RESULTS
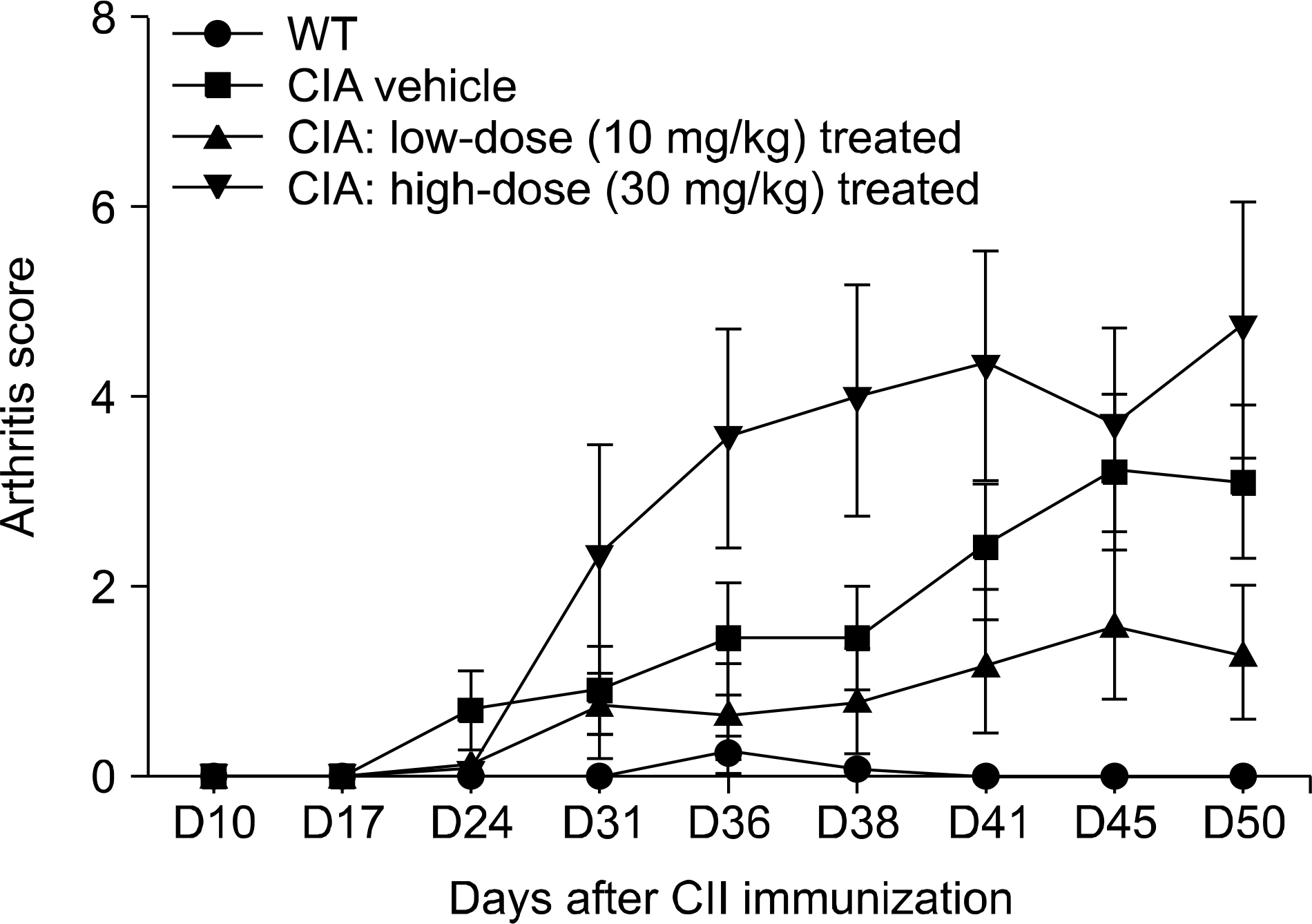
Low dose rebamipide treatment reduced the clinical arthritis score compared with vehicle treatment, whereas high dose rebamipide in CIA aggravated arthritis severity. Based on multivariate analysis, 17 metabolites were identified. The plasma levels of metabolites associated with fatty acids and phospholipid metabolism were significantly lower with rebamipide treatment than with vehicle. The levels of 15-deoxy-Δ¹²,¹â´ prostaglandin J2 and thromboxane B3 decreased only in high dose-treated groups. Certain peptide molecules, including enterostatin (VPDPR) enterostatin and bradykinin dramatically increased in rebamipide-treated groups at both doses. Additionally, corticosterone increased in the low dose-treated group and decreased in the high dose-treated group.
CONCLUSION
Metabolomics analysis revealed the anti-inflammatory effects of rebamipide and suggested the potential of the drug repositioning in metabolism- and lipid-associated diseases.
Keyword
MeSH Terms
-
Animals
Arthritis*
Arthritis, Experimental
Biomarkers
Bradykinin
Carboxymethylcellulose Sodium
Chickens
Collagen Type II
Corticosterone
Drug Repositioning
Fatty Acids
Gastritis
Mass Spectrometry*
Metabolism
Metabolome
Metabolomics*
Mice
Multivariate Analysis
Plasma
Statistics as Topic
Therapeutic Uses
Biomarkers
Bradykinin
Carboxymethylcellulose Sodium
Collagen Type II
Corticosterone
Fatty Acids
Therapeutic Uses
Figure
Reference
-
1. Kobayashi T, Zinchuk VS, Garcia del Saz E, Jiang F, Yamasaki Y, Kataoka S, et al. Suppressive effect of rebamipide, an antiulcer agent, against activation of human neutrophils exposed to formyl-methionyl-leucyl-pheny-lalanine. Histol Histopathol. 2000; 15:1067–76.2. Remans PH, Wijbrandts CA, Sanders ME, Toes RE, Breedveld FC, Tak PP, et al. CTLA-4IG suppresses reactive oxygen species by preventing synovial adherent cell-induced inactivation of Rap1, a Ras family GTPASE mediator of oxidative stress in rheumatoid arthritis T cells. Arthritis Rheum. 2006; 54:3135–43.
Article3. Kurimoto C, Kawano S, Tsuji G, Hatachi S, Jikimoto T, Sugiyama D, et al. Thioredoxin may exert a protective effect against tissue damage caused by oxidative stress in salivary glands of patients with Sjögren's syndrome. J Rheumatol. 2007; 34:2035–43.4. Najim RA, Sharquie KE, Abu-Raghif AR. Oxidative stress in patients with Behcet's disease: I correlation with severity and clinical parameters. J Dermatol. 2007; 34:308–14.
Article5. Scott JL, Gabrielides C, Davidson RK, Swingler TE, Clark IM, Wallis GA, et al. Superoxide dismutase downregulation in osteoarthritis progression and end-stage disease. Ann Rheum Dis. 2010; 69:1502–10.
Article6. Kohashi M, Ishimaru N, Arakaki R, Hayashi Y. Effective treatment with oral administration of rebamipide in a mouse model of Sjögren's syndrome. Arthritis Rheum. 2008; 58:389–400.
Article7. Arimoto A, Kitagawa K, Mita N, Takahashi Y, Shibuya E, Sasaki H. Effect of rebamipide ophthalmic suspension on signs and symptoms of keratoconjunctivitis sicca in Sjögren syndrome patients with or without punctal occlusions. Cornea. 2014; 33:806–11.
Article8. Moon SJ, Park JS, Woo YJ, Lim MA, Kim SM, Lee SY, et al. Rebamipide suppresses collagen-induced arthritis through reciprocal regulation of th17/treg cell differentiation and heme oxygenase 1 induction. Arthritis Rheumatol. 2014; 66:874–85.
Article9. Moon SJ, Woo YJ, Jeong JH, Park MK, Oh HJ, Park JS, et al. Rebamipide attenuates pain severity and cartilage degeneration in a rat model of osteoarthritis by downregulating oxidative damage and catabolic activity in chondrocytes. Osteoarthritis Cartilage. 2012; 20:1426–38.
Article10. Visconti R, Grieco D. New insights on oxidative stress in cancer. Curr Opin Drug Discov Devel. 2009; 12:240–5.11. Maritim AC, Sanders RA, Watkins JB 3rd. Diabetes, oxidative stress, and antioxidants: a review. J Biochem Mol Toxicol. 2003; 17:24–38.
Article12. Rauscher FM, Sanders RA, Watkins JB 3rd. Effects of coen-zyme Q10 treatment on antioxidant pathways in normal and streptozotocin-induced diabetic rats. J Biochem Mol Toxicol. 2001; 15:41–6.
Article13. Surh YJ, Kundu JK, Na HK, Lee JS. Redox-sensitive transcription factors as prime targets for chemoprevention with anti-inflammatory and antioxidative phytochemicals. J Nutr. 2005; 135(12 Suppl):2993S–3001S.
Article14. Cortés-Jofré M, Rueda JR, Corsini-Muñoz G, Fonseca-Cortés C, Caraballoso M, Bonfill Cosp X. Drugs for preventing lung cancer in healthy people. Cochrane Database Syst Rev. 2012; 10:CD002141.
Article15. de Groot RH, Ouwehand C, Jolles J. Eating the right amount of fish: inverted U-shape association between fish consumption and cognitive performance and academic achievement in Dutch adolescents. Prostaglandins Leukot Essent Fatty Acids. 2012; 86:113–7.
Article16. Wang X, Zhang A, Sun H. Future perspectives of Chinese medical formulae: chinmedomics as an effector. OMICS. 2012; 16:414–21.
Article17. Rosloniec EF, Cremer M, Kang AH, Myers LK, Brand DD. Collagen-induced arthritis. Curr Protoc Immunol. 2010; Chapter 15: Unit 15.5.1-25.
Article18. Sangster T, Major H, Plumb R, Wilson AJ, Wilson ID. A pragmatic and readily implemented quality control strategy for HPLC-MS and GC-MS-based metabonomic analysis. Analyst. 2006; 131:1075–8.
Article19. Gika HG, Theodoridis GA, Wilson ID. Liquid chromatography and ultra-performance liquid chromatography-mass spectrometry fingerprinting of human urine: sample stability under different handling and storage conditions for metabonomics studies. J Chromatogr A. 2008; 1189; 314–22.20. Lai L, Michopoulos F, Gika H, Theodoridis G, Wilkinson RW, Odedra R, et al. Methodological considerations in the development of HPLC-MS methods for the analysis of rodent plasma for metabonomic studies. Mol Biosyst. 2010; 6:108–20.
Article21. Xing J, Chen X, Sun Y, Luan Y, Zhong D. Interaction of bai-calin and baicalein with antibiotics in the gastrointestinal tract. J Pharm Pharmacol. 2005; 57:743–50.
Article22. Wu X, Cao H, Zhao L, Song J, She Y, Feng Y. Metabolomic analysis of glycerophospholipid signatures of inflammation treated with non-steroidal anti-inflammatory drugs-induced-RAW264.7 cells using (1)H NMR and U-HPLC/ Q-TOF-MS. J Chromatogr B Analyt Technol Biomed Life Sci. 2016; 1028; 199–215.23. Barnes PJ, Adcock IM. How do corticosteroids work in asth-ma? Ann Intern Med. 2003; 139:359–70.
Article24. Rhen T, Cidlowski JA. Antiinflammatory action of glucocorticoids–new mechanisms for old drugs. N Engl J Med. 2005; 353:1711–23.25. Straus DS, Glass CK. Cyclopentenone prostaglandins: new insights on biological activities and cellular targets. Med Res Rev. 2001; 21:185–210.
Article26. Bell-Parikh LC, Ide T, Lawson JA, McNamara P, Reilly M, FitzGerald GA. Biosynthesis of 15-deoxy-delta12,14-PGJ2 and the ligation of PPARgamma. J Clin Invest. 2003; 112:945–55.27. Chawla A, Boisvert WA, Lee CH, Laffitte BA, Barak Y, Joseph SB, et al. A PPAR gamma-LXR-ABCA1 pathway in macrophages is involved in cholesterol efflux and atherogenesis. Mol Cell. 2001; 7:161–71.28. Ren D, Collingwood TN, Rebar EJ, Wolffe AP, Camp HS. PPARgamma knockdown by engineered transcription factors: exogenous PPARgamma2 but not PPARgamma1 re-activates adipogenesis. Genes Dev. 2002; 16:27–32.29. Shi MA, Shi GP. Different roles of mast cells in obesity and diabetes: lessons from experimental animals and humans. Front Immunol. 2012; 3:7.
Article30. Jessop DS, Harbuz MS. A defect in cortisol production in rheumatoid arthritis: why are we still looking? Rheumatology (Oxford). 2005; 44:1097–100.
Article31. Straub RH, Paimela L, Peltomaa R, Schölmerich J, Leirisalo-Repo M. Inadequately low serum levels of steroid hormones in relation to interleukin-6 and tumor necrosis factor in untreated patients with early rheumatoid arthritis and reactive arthritis. Arthritis Rheum. 2002; 46:654–62.
Article32. Straub RH, Buttgereit F, Cutolo M. Alterations of the hypo-thalamic-pituitary-adrenal axis in systemic immune diseases – a role for misguided energy regulation. Clin Exp Rheumatol. 2011; 29(5 Suppl 68):S23–31.33. Tsigos C, Papanicolaou DA, Defensor R, Mitsiadis CS, Kyrou I, Chrousos GP. Dose effects of recombinant human interleukin-6 on pituitary hormone secretion and energy expenditure. Neuroendocrinology. 1997; 66:54–62.34. del Rey A, Wolff C, Wildmann J, Randolf A, Hahnel A, Besedovsky HO, Straub RH. Disrupted brain-immune system-joint communication during experimental arthritis. Arthritis Rheum. 2008; 58:3090–9.
Article35. Spurney RF, Fan PY, Ruiz P, Sanfilippo F, Pisetsky DS, Coffman TM. Thromboxane receptor blockade reduces renal injury in murine lupus nephritis. Kidney Int. 1992; 41:973–82.
Article36. Perico N, Rossini M, Imberti O, Malanchini B, Cornejo RP, Gaspari F, et al. Thromboxane receptor blockade attenuates chronic cyclosporine nephrotoxicity and improves survival in rats with renal isograft. J Am Soc Nephrol. 1992; 2:1398–404.
Article37. Smith SR, Creech EA, Schaffer AV, Martin LL, Rakhit A, Douglas FL, et al. Effects of thromboxane synthase inhibition with CGS 13080 in human cyclosporine nephrotoxicity. Kidney Int. 1992; 41:199–205.
Article38. Lin L, Park M, York DA. Enterostatin inhibition of dietary fat intake is modulated through the melanocortin system. Peptides. 2007; 28:643–9.
Article39. Lin L, Park M, Hulver M, York DA. Different metabolic responses to central and peripheral injection of enterostatin. Am J Physiol Regul Integr Comp Physiol. 2006; 290:R909–15.
Article40. Ookuma M, York DA. Inhibition of insulin release by enterostatin. Int J Obes Relat Metab Disord. 1998; 22:800–5.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Simultaneous Screening of 177 Drugs of Abuse in Urine Using Ultra-performance Liquid Chromatography with Tandem Mass Spectrometry in Drug-intoxicated Patients
- Determination of donepezil in human plasma using ultra performance liquid chromatography-tandem mass spectrometry
- Utility of Integrated Analysis of Pharmacogenomics and Pharmacometabolomics in Early Phase Clinical Trial: A Case Study of a New Molecular Entity
- Bioanalytical methods for the detection of duloxetine and thioctic acid in plasma using ultra performance liquid chromatography with tandem mass spectrometry (UPLC-MS/MS)
- Metabolism and excretion of novel pulmonary-targeting docetaxel liposome in rabbits