J Periodontal Implant Sci.
2017 Aug;47(4):194-210. 10.5051/jpis.2017.47.4.194.
Alveolar socket preservation with demineralised bovine bone mineral and a collagen matrix
- Affiliations
-
- 1Implant Center for Edentulism and Jawbone Atrophies, Maxillofacial Surgery and Odontostomatology Unit, IRCCS CÃ Granda Ospedale Maggiore Policlinico Foundation, University of Milan, Milan, Italy. pierpaolo.poli@unimi.it
- 2Section of Implant Dentistry and Oral Rehabilitation, Department of Biomedical, Surgical and Dental Sciences, Dental Clinic, IRCCS Galeazzi Orthopedic Institute, University of Milan, Milan, Italy.
- 3Dental School, Vita-Salute University and Department of Dentistry, IRCCS San Raffaele Hospital, Milan, Italy.
- 4Medical Center, University of Freiburg Institute for Clinical Chemistry and Laboratory Medicine, Freiburg, Germany.
- KMID: 2388107
- DOI: http://doi.org/10.5051/jpis.2017.47.4.194
Abstract
- PURPOSE
The aim of the present study was to evaluate the healing of post-extraction sockets following alveolar ridge preservation clinically, radiologically, and histologically.
METHODS
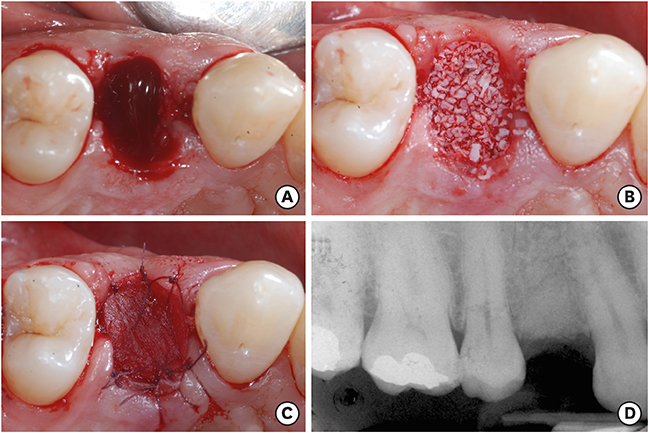
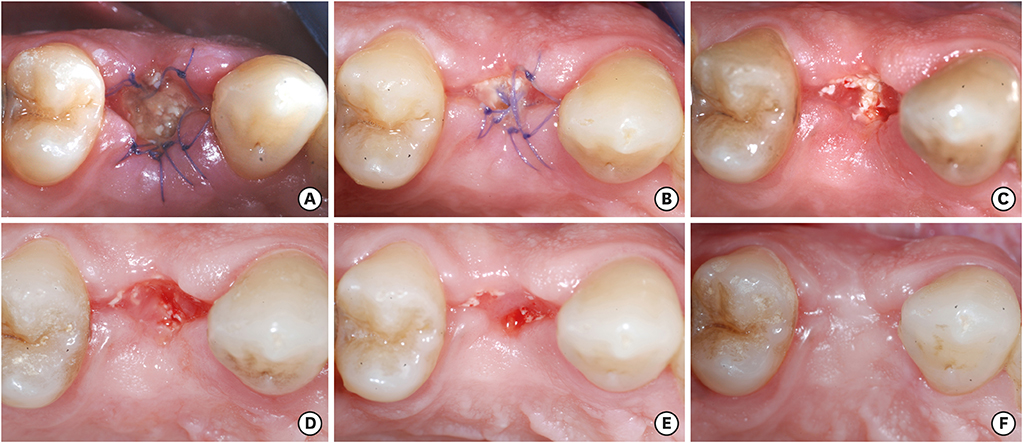
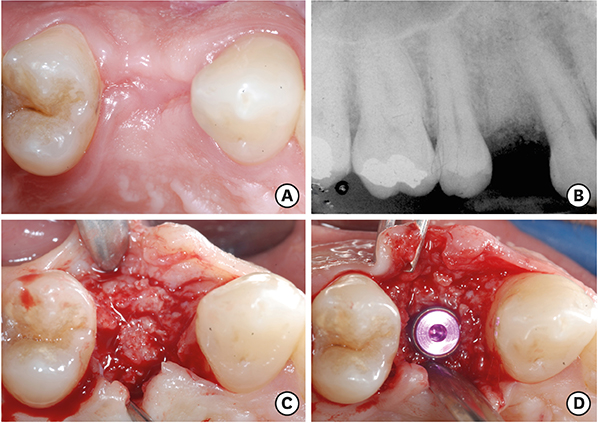
Overall, 7 extraction sockets in 7 patients were grafted with demineralised bovine bone mineral and covered with a porcine-derived non-crosslinked collagen matrix (CM). Soft tissue healing was clinically evaluated on the basis of a specific healing index. Horizontal and vertical ridge dimensional changes were assessed clinically and radiographically at baseline and 6 months after implant placement. For histological and histomorphometric analysis, bone biopsies were harvested from the augmented sites during implant surgery 6 months after the socket preservation procedure.
RESULTS
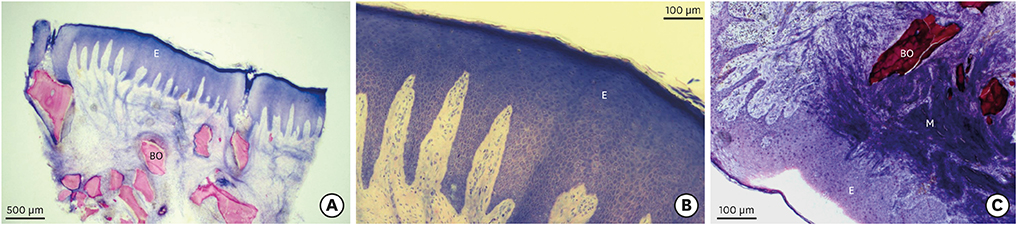
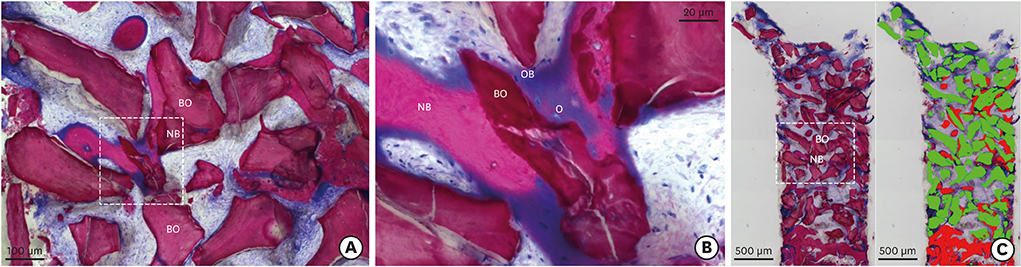
Clinically, healing proceeded uneventfully in all the sockets. A trend towards reduced horizontal and vertical socket dimensions was observed from baseline to the final examination. The mean width and height of resorption were 1.21 mm (P=0.005) and 0.46 mm (P=0.004), respectively. Histologically, residual xenograft particles (31.97%±3.52%) were surrounded by either newly formed bone (16.02%±7.06%) or connective tissue (50.67%±8.42%) without fibrous encapsulation. The CM underwent a physiological substitution process in favour of well-vascularised collagen-rich connective tissue.
CONCLUSIONS
Socket preservation using demineralised bovine bone mineral in combination with CM provided stable dimensional changes of the alveolar ridge associated with good re-epithelialisation of the soft tissues during a 6-month healing period.
Keyword
MeSH Terms
Figure
Reference
-
1. Tan WL, Wong TL, Wong MC, Lang NP. A systematic review of post-extractional alveolar hard and soft tissue dimensional changes in humans. Clin Oral Implants Res. 2012; 23:Suppl 5. 1–21.
Article2. Araújo MG, Silva CO, Misawa M, Sukekava F. Alveolar socket healing: what can we learn? Periodontol 2000. 2015; 68:122–134.
Article3. Botticelli D, Berglundh T, Lindhe J. Hard-tissue alterations following immediate implant placement in extraction sites. J Clin Periodontol. 2004; 31:820–828.
Article4. Laney WR. Glossary of oral and maxillofacial implants. Chicago: Quintessence;2007.5. Jambhekar S, Kernen F, Bidra AS. Clinical and histologic outcomes of socket grafting after flapless tooth extraction: a systematic review of randomized controlled clinical trials. J Prosthet Dent. 2015; 113:371–382.
Article6. Araújo MG, Liljenberg B, Lindhe J. Dynamics of Bio-Oss Collagen incorporation in fresh extraction wounds: an experimental study in the dog. Clin Oral Implants Res. 2010; 21:55–64.
Article7. Sbordone C, Toti P, Martuscelli R, Guidetti F, Ramaglia L, Sbordone L. Retrospective volume analysis of bone remodeling after tooth extraction with and without deproteinized bovine bone mineral insertion. Clin Oral Implants Res. 2016; 27:1152–1159.
Article8. Gross JS. Bone grafting materials for dental applications: a practical guide. Compend Contin Educ Dent. 1997; 18:1013–1018.9. Lindhe J, Cecchinato D, Donati M, Tomasi C, Liljenberg B. Ridge preservation with the use of deproteinized bovine bone mineral. Clin Oral Implants Res. 2014; 25:786–790.
Article10. Cardaropoli D, Tamagnone L, Roffredo A, Gaveglio L. Relationship between the buccal bone plate thickness and the healing of postextraction sockets with/without ridge preservation. Int J Periodontics Restorative Dent. 2014; 34:211–217.
Article11. Becker W, Clokie C, Sennerby L, Urist MR, Becker BE. Histologic findings after implantation and evaluation of different grafting materials and titanium micro screws into extraction sockets: case reports. J Periodontol. 1998; 69:414–421.
Article12. Carmagnola D, Adriaens P, Berglundh T. Healing of human extraction sockets filled with Bio-Oss. Clin Oral Implants Res. 2003; 14:137–143.13. Sisti A, Canullo L, Mottola MP, Covani U, Barone A, Botticelli D. Clinical evaluation of a ridge augmentation procedure for the severely resorbed alveolar socket: multicenter randomized controlled trial, preliminary results. Clin Oral Implants Res. 2012; 23:526–535.
Article14. Avila-Ortiz G, Elangovan S, Kramer KW, Blanchette D, Dawson DV. Effect of alveolar ridge preservation after tooth extraction: a systematic review and meta-analysis. J Dent Res. 2014; 93:950–958.
Article15. Ghanaati S, Schlee M, Webber MJ, et al. Evaluation of the tissue reaction to a new bilayered collagen matrix in vivo and its translation to the clinic. Biomed Mater. 2011; 6:015010.16. Herford AS, Akin L, Cicciu M, Maiorana C, Boyne PJ. Use of a porcine collagen matrix as an alternative to autogenous tissue for grafting oral soft tissue defects. J Oral Maxillofac Surg. 2010; 68:1463–1470.
Article17. Sanz M, Lorenzo R, Aranda JJ, Martin C, Orsini M. Clinical evaluation of a new collagen matrix (Mucograft prototype) to enhance the width of keratinized tissue in patients with fixed prosthetic restorations: a randomized prospective clinical trial. J Clin Periodontol. 2009; 36:868–876.
Article18. Thoma DS, Sancho-Puchades M, Ettlin DA, Hämmerle CH, Jung RE. Impact of a collagen matrix on early healing, aesthetics and patient morbidity in oral mucosal wounds - a randomized study in humans. J Clin Periodontol. 2012; 39:157–165.
Article19. Jung RE, Hürzeler MB, Thoma DS, Khraisat A, Hämmerle CH. Local tolerance and efficiency of two prototype collagen matrices to increase the width of keratinized tissue. J Clin Periodontol. 2011; 38:173–179.
Article20. Parashis AO, Hawley CE, Stark PC, Ganguly R, Hanley JB, Steffensen B. Prospective clinical and radiographic study of alveolar ridge preservation combining freeze-dried bone allograft with two xenogeneic collagen matrices. J Periodontol. 2016; 87:416–425.
Article21. Landsberg CJ. Implementing socket seal surgery as a socket preservation technique for pontic site development: surgical steps revisited--a report of two cases. J Periodontol. 2008; 79:945–954.
Article22. Landry R, Turnbell R, Howley T. Effectiveness of benzydamyne HCl in the treatment of periodontal post-surgical patients. Res Clin Forums. 1988; 10:105–118.23. Donath K, Breuner G. A method for the study of undecalcified bones and teeth with attached soft tissues. The Säge-Schliff (sawing and grinding) technique. J Oral Pathol. 1982; 11:318–326.
Article24. Morjaria KR, Wilson R, Palmer RM. Bone healing after tooth extraction with or without an intervention: a systematic review of randomized controlled trials. Clin Implant Dent Relat Res. 2014; 16:1–20.25. Cardaropoli D, Tamagnone L, Roffredo A, Gaveglio L, Cardaropoli G. Socket preservation using bovine bone mineral and collagen membrane: a randomized controlled clinical trial with histologic analysis. Int J Periodontics Restorative Dent. 2012; 32:421–430.26. Darby I, Chen ST, Buser D. Ridge preservation techniques for implant therapy. Int J Oral Maxillofac Implants. 2009; 24:Suppl. 260–271.27. Willenbacher M, Al-Nawas B, Berres M, Kämmerer PW, Schiegnitz E. The effects of alveolar ridge preservation: a meta-analysis. Clin Implant Dent Relat Res. 2016; 18:1248–1268.28. Festa VM, Addabbo F, Laino L, Femiano F, Rullo R. Porcine-derived xenograft combined with a soft cortical membrane versus extraction alone for implant site development: a clinical study in humans. Clin Implant Dent Relat Res. 2013; 15:707–713.
Article29. Nevins M, Camelo M, De Paoli S, et al. A study of the fate of the buccal wall of extraction sockets of teeth with prominent roots. Int J Periodontics Restorative Dent. 2006; 26:19–29.
Article30. Chappuis V, Engel O, Reyes M, Shahim K, Nolte LP, Buser D. Ridge alterations post-extraction in the esthetic zone: a 3D analysis with CBCT. J Dent Res. 2013; 92:195S–201S.31. Meloni SM, Tallarico M, Lolli FM, Deledda A, Pisano M, Jovanovic SA. Postextraction socket preservation using epithelial connective tissue graft vs porcine collagen matrix. 1-year results of a randomised controlled trial. Eur J Oral Implantology. 2015; 8:39–48.32. Hallman M, Lundgren S, Sennerby L. Histologic analysis of clinical biopsies taken 6 months and 3 years after maxillary sinus floor augmentation with 80% bovine hydroxyapatite and 20% autogenous bone mixed with fibrin glue. Clin Implant Dent Relat Res. 2001; 3:87–96.
Article33. Sartori S, Silvestri M, Forni F, Icaro Cornaglia A, Tesei P, Cattaneo V. Ten-year follow-up in a maxillary sinus augmentation using anorganic bovine bone (Bio-Oss). A case report with histomorphometric evaluation. Clin Oral Implants Res. 2003; 14:369–372.
Article34. De Risi V, Clementini M, Vittorini G, Mannocci A, De Sanctis M. Alveolar ridge preservation techniques: a systematic review and meta-analysis of histological and histomorphometrical data. Clin Oral Implants Res. 2015; 26:50–68.
Article35. Lindgren C, Mordenfeld A, Johansson CB, Hallman M. A 3-year clinical follow-up of implants placed in two different biomaterials used for sinus augmentation. Int J Oral Maxillofac Implants. 2012; 27:1151–1162.36. Rocchietta I, Schupbach P, Ghezzi C, Maschera E, Simion M. Soft tissue integration of a porcine collagen membrane: an experimental study in pigs. Int J Periodontics Restorative Dent. 2012; 32:e34–e40.37. Jung RE, Philipp A, Annen BM, et al. Radiographic evaluation of different techniques for ridge preservation after tooth extraction: a randomized controlled clinical trial. J Clin Periodontol. 2013; 40:90–98.
Article38. Perelman-Karmon M, Kozlovsky A, Liloy R, Artzi Z. Socket site preservation using bovine bone mineral with and without a bioresorbable collagen membrane. Int J Periodontics Restorative Dent. 2012; 32:459–465.39. Roccuzzo M, Gaudioso L, Bunino M, Dalmasso P. Long-term stability of soft tissues following alveolar ridge preservation: 10-year results of a prospective study around nonsubmerged implants. Int J Periodontics Restorative Dent. 2014; 34:795–804.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The influence of membrane exposure on post-extraction dimensional change following ridge preservation technique
- Alveolar ridge preservation following extraction of multiple anterior teeth with and without adjunctive enamel matrix derivative: A case report
- A comparison between anorganic bone and collagen-preserving bone xenografts for alveolar ridge preservation: systematic review and future perspectives
- The study on ridge preservation for implant site development
- Alveolar Ridge Preservation in the Severely Damaged Sockets of the Anterior Maxilla Followed by Delayed Implant Placement