J Bacteriol Virol.
2017 Jun;47(2):87-95. 10.4167/jbv.2017.47.2.87.
Replication of Recombinant Flock House Virus RNA Encapsidated by Turnip Yellow Mosaic Virus Coat Proteins in Nicotiana benthamiana
- Affiliations
-
- 1School of Life Sciences, Chungbuk National University, Cheongju, Korea. tjcho@chungbuk.ac.kr
- KMID: 2384411
- DOI: http://doi.org/10.4167/jbv.2017.47.2.87
Abstract
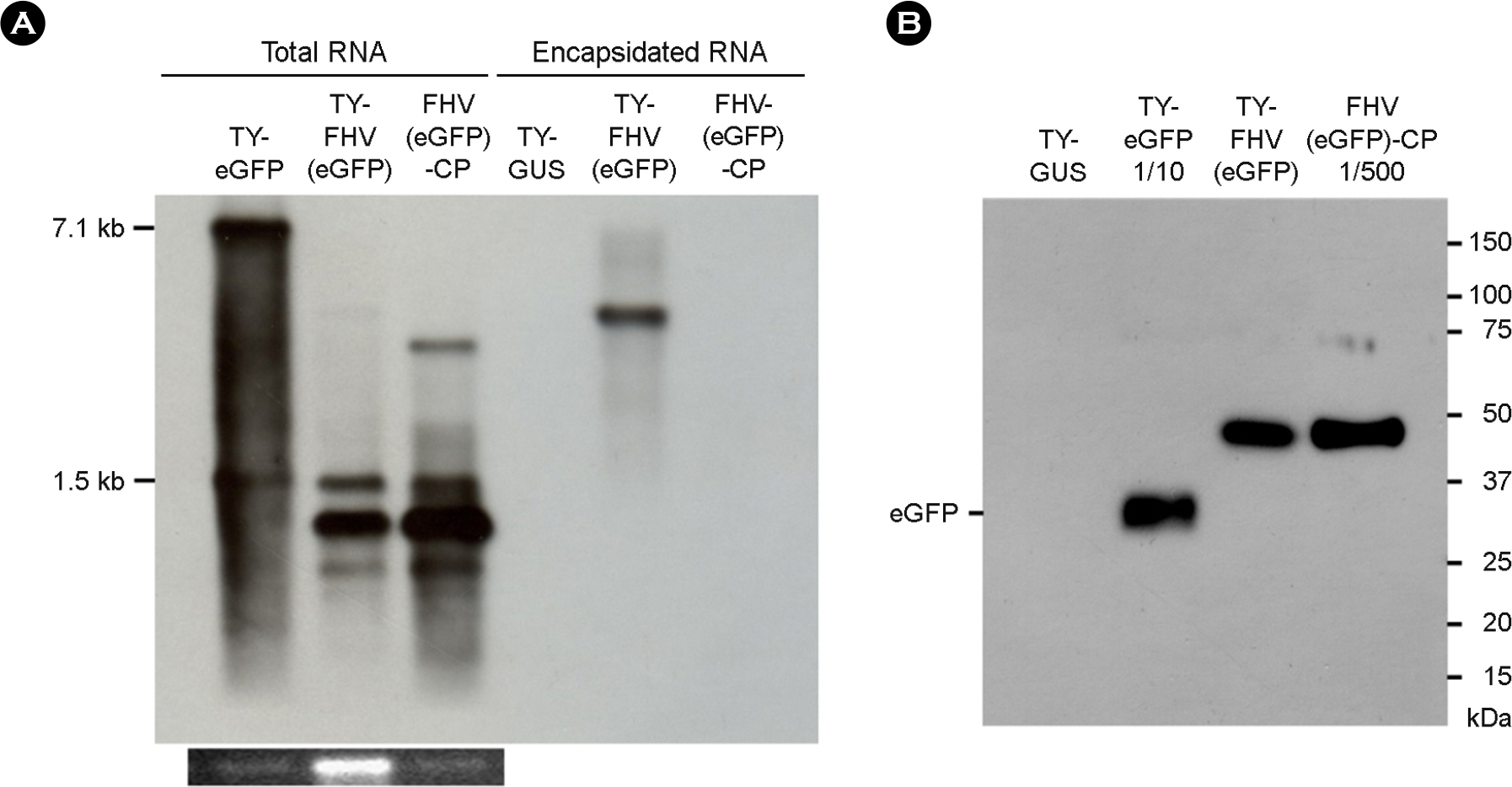
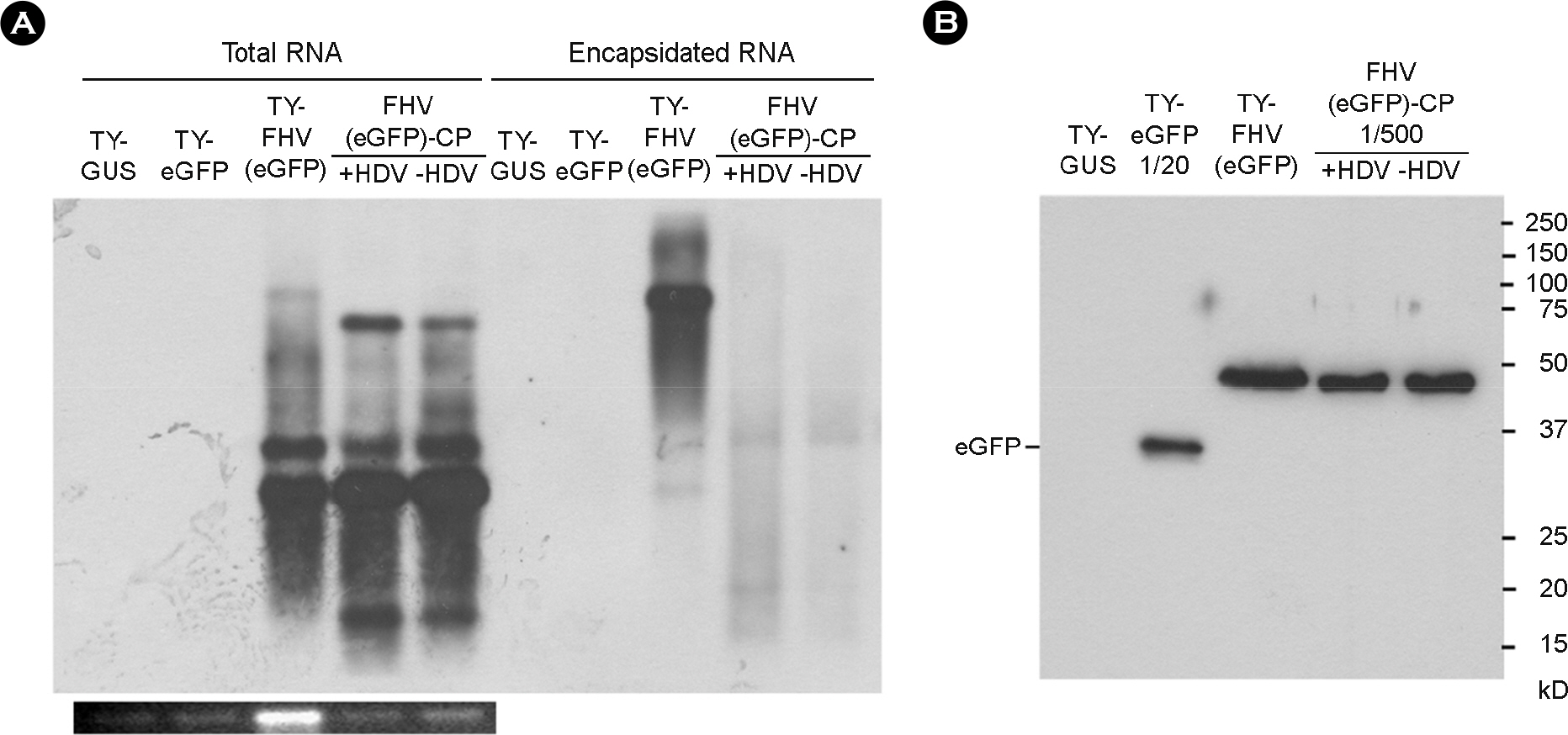
- It was previously observed that recombinant flock house virus (FHV) RNA1 was efficiently packaged into turnip yellow mosaic virus (TYMV), provided that the TYMV coat protein (CP) sequence was present at the 3"²-end. FHV RNA encapsidated by TYMV CPs also had a four-nucleotide extension at the 5"²-end. Since even a short extension at the 5"²- and 3"²-ends of FHV RNA1 inhibits replication, we examined whether the recombinant FHV RNA is indeed capable of replication. To this end, we introduced constructs expressing recombinant FHV RNAs into the plant Nicotiana benthamiana. Northern blot analysis of inoculated leaves suggested abundant production of recombinant FHV RNA1 and its subgenomic RNA. This demonstrated that recombinant FHV RNA with terminal extensions at both ends was competent for replication. We also showed that the recombinant FHV RNA can express the reporter gene encoding enhanced green fluorescent protein.
MeSH Terms
Figure
Reference
-
1). Geall AJ, Mandl CW, Ulmer JB. RNA: The new revolu-tion in nucleic acid vaccines. Semin Immunol. 2013; 25:152–9.
Article2). Sahin U, Karikó K, Tüereci Ö. mRNA-based thera- peutics – developing a new class of drugs. Nat Rev Drug Discov. 2014; 13:759–80.3). Smith ML, Corbo T, Bernales J, Lindbo JA, Pogue GP, Palmer KE, et al. Assembly of trans-encapsidated recombinant viral vectors engineered from Tobacco mosaic virus and Semliki Forest virus and their evaluation as immunogens. Virology. 2007; 358:321–33.
Article4). Azizgolshani O, Garmann RF, Cadena-Nava R, Knobler CM, Gelbart WM. Reconstituted plant viral capsids can release genes to mammalian cells. Virology. 2013; 441:12–7.
Article5). Shin HI, Kim IC, Cho TJ. Replication and encapsidation of recombinant Turnip yellow mosaic virus RNA. BMB Rep. 2008; 41:739–44.
Article6). Dreher TW. Turnip yellow mosaic virus: transfer RNA mimicry, chloroplasts and a C-rich genome. Mol Plant Pathol. 2004; 5:367–75.
Article7). Kim HB, Kim DY, Cho TJ. Replication and packaging of Turnip yellow mosaic virus RNA containing Flock house virus RNA1 sequence. BMB Rep. 2014; 47:330–5.
Article8). Ball LA. Requirements for the self-directed replication of Flock house virus RNA1. J Virol. 1995; 69:720–7.9). Price BD, Ruekert RR, Ahlquist P. Complete replication of an animal virus and maintenance of expression vectors derived from it in Saccharomyces cerevisiae. Proc Natl Acad Sci U S A. 1996; 93:9465–70.10). Selling BH, Allison RF, Kaesberg P. Genomic RNA of an insect virus directs synthesis of infectious virions in plants. Proc Natl Acad Sci U S A. 1990; 87:434–8.
Article11). Johnson KL, Ball LA. Induction and maintenance of autonomous Flock house virus RNA1 replication. J Virol. 1999; 73:7933–42.
Article12). Li H, Li WX, Ding SW. Induction and suppression of RNA silencing by an animal virus. Science. 2002; 296:1319–21.
Article13). Shi BJ, Ding SW, Symons RH. Plasmid vector for cloning infectious cDNAs from plant RNA viruses: high infectivity of cDNA clones of tomato aspermy cucumovirus. J Gen Virol. 1997; 78:1181–5.
Article14). Xiang C, Han P, Lutziger I, Wang K, Oliver DJ. A mini binary vector series for plant transformation. Plant Mol Biol. 1999; 40:711–7.15). Cho TJ, Dreher TW. Encapsidation of genomic but not subgenomic Turnip yellow mosaic virus RNA by coat protein provided in trans. Virology. 2006; 356:126–35.
Article16). Perrotta AT, Shih I, Been MD. Imidazole rescue of a cytosine mutation in a self-cleaving ribozyme. Science. 1999; 286:123–6.
Article17). Albariño CG, Price BD, Eckerle LD, Ball LA. Characterization and template properties of RNA dimers generated during Flock house virus RNA replication. Virology. 2001; 289:269–82.
Article18). Annamalai P, Rofail F, DeMason DA, Rao AL. Replication-coupled packaging mechanism in positive-strand RNA viruses: Synchronized coexpression of functional multigenome RNA components of an animal and a plant virus in Nicotiana benthamiana cells by agroinfiltraion. J Virol. 2008; 82:1484–95.19). Ball LA, Li Y. Cis-Acting requirements for the replication of Flock house virus RNA2. J Virol. 1993; 67:3544–51.20). Eckerle LD, Albariño CG, Ball LA. Flock house virus subgenomic RNA3 is replicated and its replication correlates with transactivation of RNA2. Virology. 2003; 317:95–108.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Genome Size Constraint in Replication and Packaging of Turnip Yellow Mosaic Virus
- A Sequence in Coat Protein Open Reading Frame Is Required for Turnip Yellow Mosaic Virus Replication
- Characterization of a Replication Element in the Coat Protein ORF of Turnip Yellow Mosaic Virus
- N-terminal Extension of Coat Protein of Turnip Yellow Mosaic Virus has Variable Effects on Replication, RNA Packaging, and Virion Assembly Depending on the Inserted Sequence
- Cysteine-Added Mutants of Turnip Yellow Mosaic Virus