J Korean Med Sci.
2016 Aug;31(8):1273-1278. 10.3346/jkms.2016.31.8.1273.
Simvastatin Reduces Capsular Fibrosis around Silicone Implants
- Affiliations
-
- 1Department of Plastic and Reconstructive Surgery, College of Medicine, Yeungnam University, Daegu, Korea. kimyon@ynu.ac.kr
- 2RED Plastic Surgery, Changwon, Korea.
- KMID: 2373756
- DOI: http://doi.org/10.3346/jkms.2016.31.8.1273
Abstract
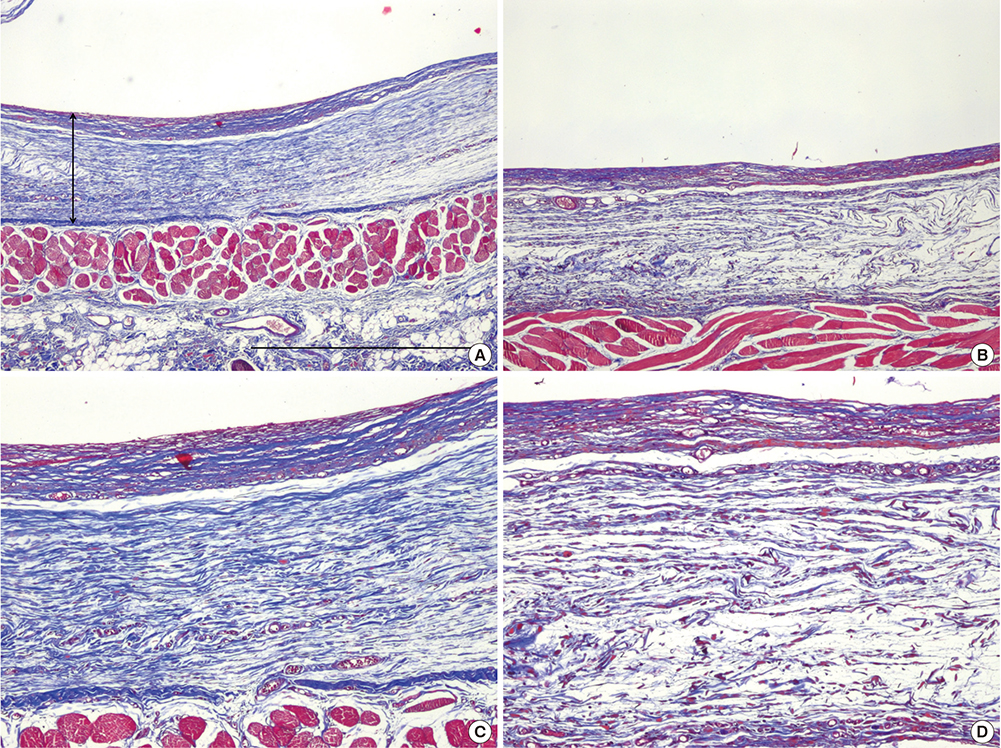
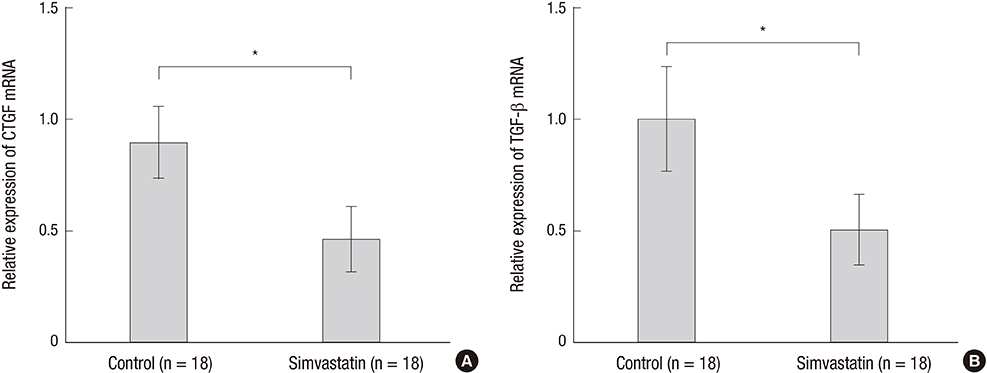
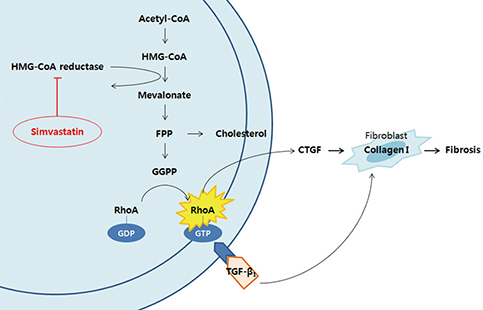
- Capsular fibrosis and contracture occurs in most breast reconstruction patients who undergo radiotherapy, and there is no definitive solution for its prevention. Simvastatin was effective at reducing fibrosis in various models. Peri-implant capsular formation is the result of tissue fibrosis development in irradiated breasts. The purpose of this study was to examine the effect of simvastatin on peri-implant fibrosis in rats. Eighteen male Sprague-Dawley rats were allocated to an experimental group (9 rats, 18 implants) or a control group (9 rats, 18 implants). Two hemispherical silicone implants, 10 mm in diameter, were inserted in subpanniculus pockets in each rat. The next day, 10-Gy of radiation from a clinical accelerator was targeted at the implants. Simvastatin (15 mg/kg/day) was administered by oral gavage in the experimental group, while animals in the control group received water. At 12 weeks post-implantation, peri-implant capsules were harvested and examined histologically and by real-time polymerase chain reaction. The average capsular thickness was 371.2 μm in the simvastatin group and 491.2 μm in the control group. The fibrosis ratio was significantly different, with 32.33% in the simvastatin group and 58.44% in the control group (P < 0.001). Connective tissue growth factor (CTGF) and transforming growth factor (TGF)-β1 gene expression decreased significantly in the simvastatin group compared to the control group (P < 0.001). This study shows that simvastatin reduces radiation-induced capsular fibrosis around silicone implants in rats. This finding offers an alternative therapeutic strategy for reducing capsular fibrosis and contracture after implant-based breast reconstruction.
Keyword
MeSH Terms
-
Administration, Oral
Animals
Breast/*drug effects/metabolism/pathology/radiation effects
*Breast Implants
Connective Tissue Growth Factor/genetics/metabolism
Fibrosis
Gamma Rays
Male
Rats
Rats, Sprague-Dawley
Real-Time Polymerase Chain Reaction
Silicone Gels/*chemistry
Simvastatin/*pharmacology
Transforming Growth Factor beta1/metabolism
Connective Tissue Growth Factor
Silicone Gels
Simvastatin
Transforming Growth Factor beta1
Figure
Reference
-
1. Al-Ghazal SK, Sully L, Fallowfield L, Blamey RW. The psychological impact of immediate rather than delayed breast reconstruction. Eur J Surg Oncol. 2000; 26:17–19.2. Kronowitz SJ, Robb GL. Radiation therapy and breast reconstruction: a critical review of the literature. Plast Reconstr Surg. 2009; 124:395–408.3. Whitfield GA, Horan G, Irwin MS, Malata CM, Wishart GC, Wilson CB. Incidence of severe capsular contracture following implant-based immediate breast reconstruction with or without postoperative chest wall radiotherapy using 40 gray in 15 fractions. Radiother Oncol. 2009; 90:141–147.4. Rosato RM, Dowden RV. Radiation therapy as a cause of capsular contracture. Ann Plast Surg. 1994; 32:342–345.5. Spano A, Palmieri B, Taidelli TP, Nava MB. Reduction of capsular thickness around silicone breast implants by zafirlukast in rats. Eur Surg Res. 2008; 41:8–14.6. Grella E, Grella R, Siniscalco D, Fuccio C, Rossi F, De Novellis V, Maione S, D'Andrea F. Modification of cysteinyl leukotriene receptors expression in capsular contracture: follow-up study and definitive results. Ann Plast Surg. 2009; 63:206–208.7. Kuhn A, Singh S, Smith PD, Ko F, Falcone R, Lyle WG, Maggi SP, Wells KE, Robson MC. Periprosthetic breast capsules contain the fibrogenic cytokines TGF-beta1 and TGF-beta2, suggesting possible new treatment approaches. Ann Plast Surg. 2000; 44:387–391.8. Katzel EB, Koltz PF, Tierney R, Williams JP, Awad HA, O'keefe RJ, Langstein HN. The impact of Smad3 loss of function on TGF-β signaling and radiation-induced capsular contracture. Plast Reconstr Surg. 2011; 127:2263–2269.9. Watts KL, Sampson EM, Schultz GS, Spiteri MA. Simvastatin inhibits growth factor expression and modulates profibrogenic markers in lung fibroblasts. Am J Respir Cell Mol Biol. 2005; 32:290–300.10. Rupérez M, Rodrigues-Díez R, Blanco-Colio LM, Sánchez-López E, Rodríguez-Vita J, Esteban V, Carvajal G, Plaza JJ, Egido J, Ruiz-Ortega M. HMG-CoA reductase inhibitors decrease angiotensin II-induced vascular fibrosis: role of RhoA/ROCK and MAPK pathways. Hypertension. 2007; 50:377–383.11. O'Brien MJ, Keating NM, Elderiny S, Cerda S, Keaveny AP, Afdhal NH, Nunes DP. An assessment of digital image analysis to measure fibrosis in liver biopsy specimens of patients with chronic hepatitis C. Am J Clin Pathol. 2000; 114:712–718.12. Fischer S, Hirche C, Reichenberger MA, Kiefer J, Diehm Y, Mukundan S Jr, Alhefzi M, Bueno EM, Kneser U, Pomahac B. Silicone implants with smooth surfaces induce thinner but denser fibrotic capsules compared to those with textured surfaces in a rodent model. PLoS One. 2015; 10:e0132131.13. Kwak B, Mulhaupt F, Myit S, Mach F. Statins as a newly recognized type of immunomodulator. Nat Med. 2000; 6:1399–1402.14. Weitz-Schmidt G. Statins as anti-inflammatory agents. Trends Pharmacol Sci. 2002; 23:482–487.15. Branton MH, Kopp JB. TGF-beta and fibrosis. Microbes Infect. 1999; 1:1349–1365.16. Rubin P, Johnston CJ, Williams JP, McDonald S, Finkelstein JN. A perpetual cascade of cytokines postirradiation leads to pulmonary fibrosis. Int J Radiat Oncol Biol Phys. 1995; 33:99–109.17. Martin M, Lefaix J, Delanian S. TGF-beta1 and radiation fibrosis: a master switch and a specific therapeutic target? Int J Radiat Oncol Biol Phys. 2000; 47:277–290.18. Katzel EB, Koltz PF, Tierney R, Williams JP, Awad HA, O'Keefe RJ, Langstein HN. A novel animal model for studying silicone gel-related capsular contracture. Plast Reconstr Surg. 2010; 126:1483–1491.19. Mazaheri MK, Schultz GS, Blalock TD, Caffee HH, Chin GA. Role of connective tissue growth factor in breast implant elastomer capsular formation. Ann Plast Surg. 2003; 50:263–268.20. Hayashi N, Kakimuma T, Soma Y, Grotendorst GR, Tamaki K, Harada M, Igarashi A. Connective tissue growth factor is directly related to liver fibrosis. Hepatogastroenterology. 2002; 49:133–135.21. Chen S, Crawford M, Day RM, Briones VR, Leader JE, Jose PA, Lechleider RJ. RhoA modulates smad signaling during transforming growth factor-beta-induced smooth muscle differentiation. J Biol Chem. 2006; 281:1765–1770.22. Law M, Rudnicka AR. Statin safety: a systematic review. Am J Cardiol. 2006; 97:52C–60C.23. Preiss D, Seshasai SR, Welsh P, Murphy SA, Ho JE, Waters DD, DeMicco DA, Barter P, Cannon CP, Sabatine MS, et al. Risk of incident diabetes with intensive-dose compared with moderate-dose statin therapy: a meta-analysis. JAMA. 2011; 305:2556–2564.24. Pedersen TR, Berg K, Cook TJ, Faergeman O, Haghfelt T, Kjekshus J, Miettinen T, Musliner TA, Olsson AG, Pyörälä K, et al. Safety and tolerability of cholesterol lowering with simvastatin during 5 years in the Scandinavian Simvastatin Survival Study. Arch Intern Med. 1996; 156:2085–2092.25. Goldkind L, Laine L. A systematic review of NSAIDs withdrawn from the market due to hepatotoxicity: lessons learned from the bromfenac experience. Pharmacoepidemiol Drug Saf. 2006; 15:213–220.26. Chitturi S, George J. Hepatotoxicity of commonly used drugs: nonsteroidal anti-inflammatory drugs, antihypertensives, antidiabetic agents, anticonvulsants, lipid-lowering agents, psychotropic drugs. Semin Liver Dis. 2002; 22:169–184.27. Ou XM, Feng YL, Wen FQ, Huang XY, Xiao J, Wang K, Wang T. Simvastatin attenuates bleomycin-induced pulmonary fibrosis in mice. Chin Med J (Engl). 2008; 121:1821–1829.28. Hsieh CH, Sun CK, Lu TH, Chen YC, Lin CJ, Wu CJ, Rau CS, Jeng SF. Simvastatin induces heme oxygenase-1 expression but fails to reduce inflammation in the capsule surrounding a silicone shell implant in rats. J Surg Res. 2011; 168:272–280.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Erratum: Correction of Figure: Simvastatin Reduces Capsular Fibrosis around Silicone Implants
- A Clinical Cmparison Silicone and Acrysof IOLs
- A Retrospective Analysis of Ruptured Breast Implants
- Capsular Contracture Rates in Augmentation Mammaplasty: Comparison of Round vs Anatomical Breast Implants
- The effects of curcumin on the fibrous envelope surrounding silicone implants in rats