Ann Lab Med.
2016 Jul;36(4):358-361. 10.3343/alm.2016.36.4.358.
Preparation of Internal Quality Control Material for Lymphocyte Subset Analysis
- Affiliations
-
- 1Department of Laboratory Medicine, Seoul National University College of Medicine, Seoul, Korea. eysong1@snu.ac.kr
- 2Department of Laboratory Medicine, Seoul National University Boramae Medical Center, Seoul, Korea.
- 3Department of Biostatistics, Seoul National University Boramae Medical Center, Seoul, Korea.
- 4Department of Molecular Medicine and Biopharmaceutical Sciences, Graduate School of Convergence Science and Technology and College of Medicine, Medical Research Center, Seoul National University, Seoul, Korea.
- KMID: 2373556
- DOI: http://doi.org/10.3343/alm.2016.36.4.358
Abstract
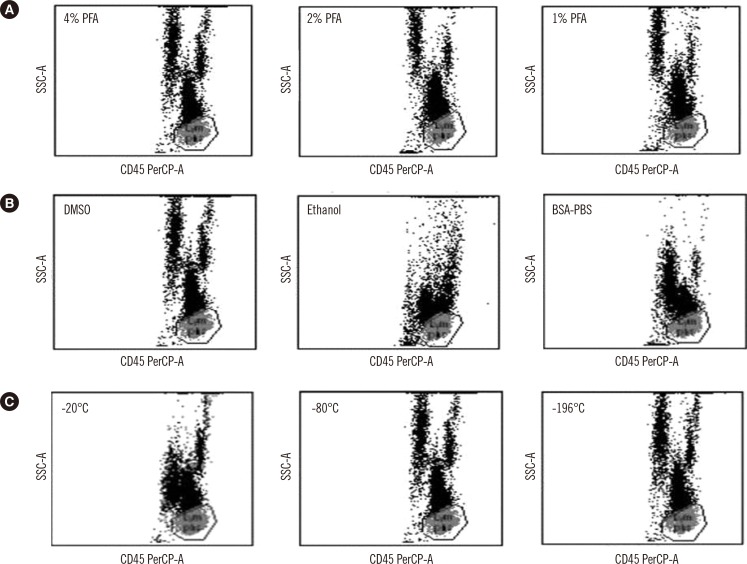
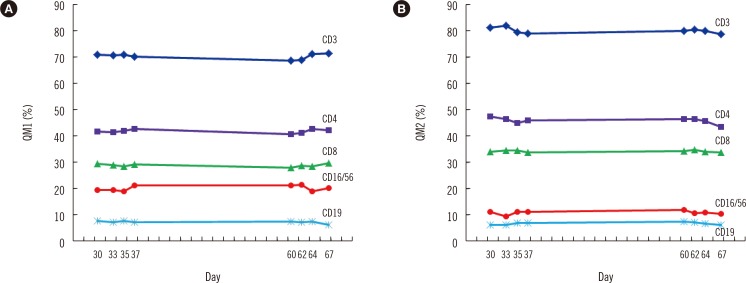
- Lymphocyte subset analysis is widely used in clinical laboratories, and more than two levels of daily QC materials are required for reliable results. Commercially available, expensive QC materials have short shelf lives and may not be suitable in resource-poor settings. We compared different methods for preparing homemade QC material, including fixation with 1%, 2%, or 4% paraformaldehyde (PFA); freezing with 10% dimethylsulfoxide (DMSO), 0.1% bovine serum albumin-phosphate buffered saline, or after ethanolic dehydration; and using cryopreservation temperatures of -20℃, -80℃, or -196℃. We found an optimal experimental condition, which is 'fixation with 4% PFA, freezing with 10% DMSO, and storage at 80℃'. To evaluate long-term stability of QC materials prepared in this optimal condition, two levels of QC materials (QM1 and QM2) were thawed after 30, 33, 35, 37, 60, 62, 64, and 67 days of cryopreservation. Lymphocyte subset was analyzed with BD Multitest IMK kit (BD Biosciences, USA). QM1 and QM2 were stable after 1-2 months of cryopreservation (CV <3% for CD3, CD4, and CD8 and 5-7% for CD16/56 and CD19). We propose this method as an alternative cost-effective protocol for preparing homemade internal QC materials for lymphocyte subset analysis in resource-poor settings.
MeSH Terms
Figure
Reference
-
1. Mandy FF, Nicholson JK, McDougal JS. CDC. Guidelines for performing single-platform absolute CD4+ T-cell determinations with CD45 gating for persons infected with human immunodeficiency virus. Centers for Disease Control and Prevention. MMWR Recomm Rep. 2003; 52:1–13. PMID: 12583540.2. Abdallah KO, Abdallah KO. B cell monitoring of transplant patients treated with anti-CD20. Clin Transpl. 2006; 427–437. PMID: 18365400.3. Wood B, Jevremovic D, Béné MC, Yan M, Jacobs P, Litwin V. ICSH/ICCS Working Group. Validation of cell-based fluorescence assays: practice guidelines from the ICSH and ICCS - part V - assay performance criteria. Cytometry B Clin Cytom. 2013; 84:315–323. PMID: 24022854.
Article4. Westerman LE, Kohatsu L, Ortiz A, McClain B, Kaplan J, Spira T, et al. A quality management systems approach for CD4 testing in resource-poor settings. Am J Clin Pathol. 2010; 134:556–567. PMID: 20855636.
Article5. Fisher Scientific. BD Multi-Check Control Web Catalogue. Accessed on Mar 2016. Available from URL: https://www.fishersci.com/us/en/catalog/search/products?keyword=BD+Multi-Check&nav.6. Zeller R. Fixation, embedding, and sectioning of tissues, embryos, and single cells. Curr Protoc Mol Biol. 2001; Chapter 14:Unit14.1.
Article7. Pinto LA, Trivett MT, Wallace D, Higgins J, Baseler M, Terabe M, et al. Fixation and cryopreservation of whole blood and isolated mononuclear cells: Influence of different procedures on lymphocyte subset analysis by flow cytometry. Cytometry B Clin Cytom. 2005; 63:47–55. PMID: 15624203.
Article8. Barnett D, Granger V, Mayr P, Storie I, Wilson GA, Reilly JT. Evaluation of a novel stable whole blood quality control material for lymphocyte subset analysis: results from the UK NEQAS immune monitoring scheme. Cytometry. 1996; 26:216–222. PMID: 8889394.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Analysis of Circulating B-1 B Lymphocyte Subsets in Patients with Systemic Lupus Erythematosus
- Peripheral Lymphocyte Subsets in Patients with Pulmonary and Extrapulmonary Tuberculosis
- Quantitation of T-lymphocyte Subsets Using the CELL-DYN Sapphire Automated Haematology Analyser
- Flow Cytometric Assays for Lymphocyte Subset Enumeration: CD45 is Inevitable for Lymphocyte Gating and CD16 is Essential for NK Cells
- CD45 is Essential for Lymphocyte Gating in a T-lymphocyte Subset Assay of Bronchoalveolar Lavage Fluid by Flow Cytometry