Ann Lab Med.
2016 Jan;36(1):55-59. 10.3343/alm.2016.36.1.55.
Establishing Reference Intervals for Sex Hormones on the Analytical Platforms Advia Centaur and Immulite 2000XP
- Affiliations
-
- 1UKM Kinderwunschzentrum, Department of Gynecology and Obstetrics, University Hospital Munster, Munster, Germany.
- 2Institute for Transfusion Medicine and Transplantation Immunology, University Hospital Munster, Munster, Germany.
- 3Center for Laboratory Medicine, University Hospital Munster, Munster, Germany. nofer@uni-muenster.de
- KMID: 2373498
- DOI: http://doi.org/10.3343/alm.2016.36.1.55
Abstract
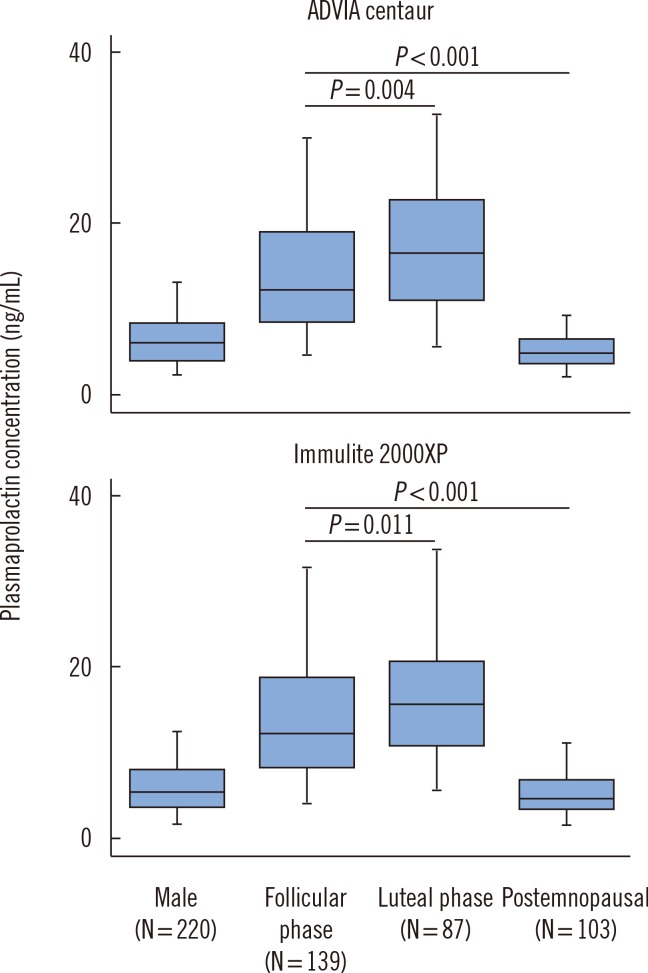
- Reliable reference intervals for sex hormones are indispensable in evaluations of the hypothalamo-pituitary-gonadal axis. This study established reference intervals for estradiol, progesterone, luteinizing hormone, follicle-stimulating hormone, and prolactin with the immunoassay platforms Advia Centaur and Immulite 2000XP (Siemens Healthcare, Germany). We recruited healthy men (n=220), women in the follicular (n=139) or luteal (n=87) phases of the menstrual cycle, and postmenopausal women (n=103). Data was analyzed according to CLSI EP28-A3c guidelines. Although reference intervals established with both platforms showed good agreement with ranges quoted by the assay manufacturer, two discrepancies were noted. First, intervals for prolactin in women were influenced by hormonal status, and the partition analysis supported their separation into subgroups based on menstrual cycle. Second, the upper limit for estradiol in the follicular phase was nearly a half of that provided by the manufacturer. This discrepancy was attributed to the stringent definition of the follicular phase (consistently set at days 3-5 after menstruation onset). Our findings suggest that reference values for prolactin should both be gender specific and account for menstrual cycle phase. The results also emphasize that clear-cut selection criteria are required when assembling populations for establishing endocrine reference intervals.
Keyword
MeSH Terms
Figure
Reference
-
1. Casanueva FF, Molitch ME, Schlechte JA, Abs R, Bonert V, Bronstein MD, et al. Guidelines of the Pituitary Society for the diagnosis and management of prolactinomas. Clin Endocrinol (Oxf). 2006; 65:265–273. PMID: 16886971.2. Mulders AG, Laven JS, Eijkemans MJ, Hughes EG, Fauser BC. Patient predictors for outcome of gonadotrophin ovulation induction in women with normogonadotrophic anovulatory infertility: a meta-analysis. Hum Reprod Update. 2003; 9:429–449. PMID: 14640376.3. Bellem A, Meiyappan S, Romans S, Einstein G. Measuring estrogens and progestagens in humans: an overview of methods. Gend Med. 2011; 8:283–299. PMID: 21907634.4. Stanczyk FZ, Jurow J, Hsing AW. Limitations of direct immunoassays for measuring circulating estradiol levels in postmenopausal women and men in epidemiologic studies. Cancer Epidemiol Biomarkers Prev. 2010; 19:903–906. PMID: 20332268.5. Horowitz GL, Altaie S, et al. Defining, establishing and verifying reference intervals in the clinical laboratory; Approved guideline-3rd ed. CLSI document C28-A3E. Wayne, PA: Clinical Laboratory Standards Institute;2008.6. Dixon WJ. Processing data for outliers. Biometrics. 1953; 9:74–89.7. Passing H, Bablok W. A new biometrical procedure for testing the equality of measurements from two different analytical methods. Application of linear regression procedures for method comparison studies in Clinical Chemistry, Part I. J Clin Chem Clin Biochem. 1983; 21:709–720. PMID: 6655447.8. Lahti A, Petersen PH, Boyd JC, Rustad P, Laake P, Solberg HE. Partitioning of nongaussian-distributed biochemical reference data into subgroups. Clin Chem. 2004; 50:891–900. PMID: 15010425.9. Sikaris K, McLachlan RI, Kazlauskas R, de Kretser D, Holden CA, Handelsman DJ. Reproductive hormone reference intervals for healthy fertile young men: evaluation of automated platform assays. J Clin Endocrinol Metab. 2005; 90:5928–5936. PMID: 16118337.10. Radicioni A, Lenzi A, Spaziani M, Anzuini A, Ruga G, Papi G, et al. A multicenter evaluation of immunoassays for follicle-stimulating hormone, luteinizing hormone and testosterone: concordance, imprecision and reference values. J Endocrinol Invest. 2013; 36:739–744. PMID: 24196213.11. Tanner MJ, Hadlow NC, Wardrop R. Variation of female prolactin levels with menopausal status and phase of menstrual cycle. Aust N Z J Obstet Gynaecol. 2011; 51:321–324. PMID: 21806583.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Comparisons of Bayer ADVIA Centaur BNP Assay with Biosite Triage BNP Assay
- Evaluation of Plasma Homocysteine Assay by ADVIA Centaur Analyzer
- Performance Evaluation of ADVIA Centaur XPT
- Evaluation of Centaur Syphilis, Immulite Syphilis, and Mediace TPLA for Detecting Treponemal Antibodies
- Evaluation of Automated Chemiluminescent Immunoanalyzer Immulite 2000