Korean J Physiol Pharmacol.
2016 Jul;20(4):379-385. 10.4196/kjpp.2016.20.4.379.
Effects of analgesics and antidepressants on TREK-2 and TRESK currents
- Affiliations
-
- 1Department of Neurosurgery, Gyeongsang National University Hospital, College of Medicine and Institute of Health Sciences, Gyeongsang National University, Jinju 52727, Korea.
- 2Department of Physiology, College of Medicine and Institute of Health Sciences, Gyeongsang National University, Jinju 52727, Korea. dawon@gnu.ac.kr
- KMID: 2371055
- DOI: http://doi.org/10.4196/kjpp.2016.20.4.379
Abstract
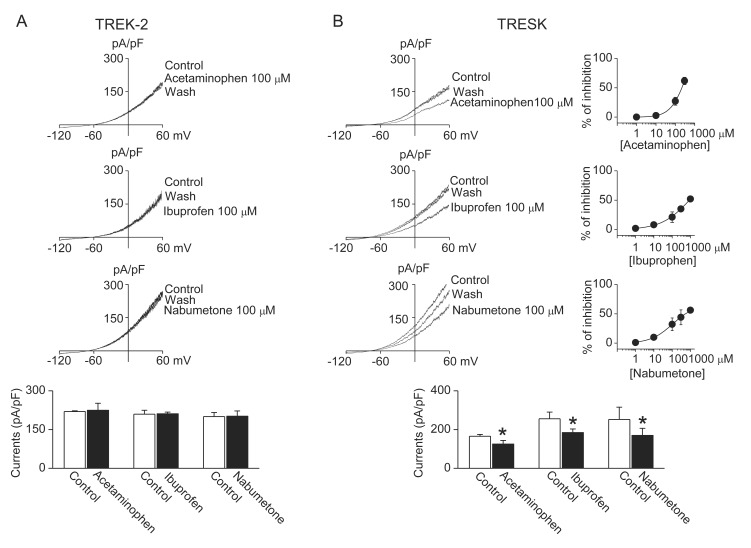
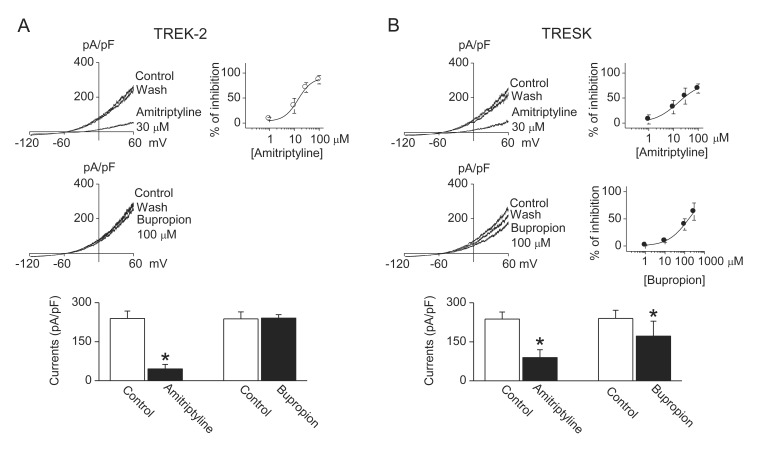
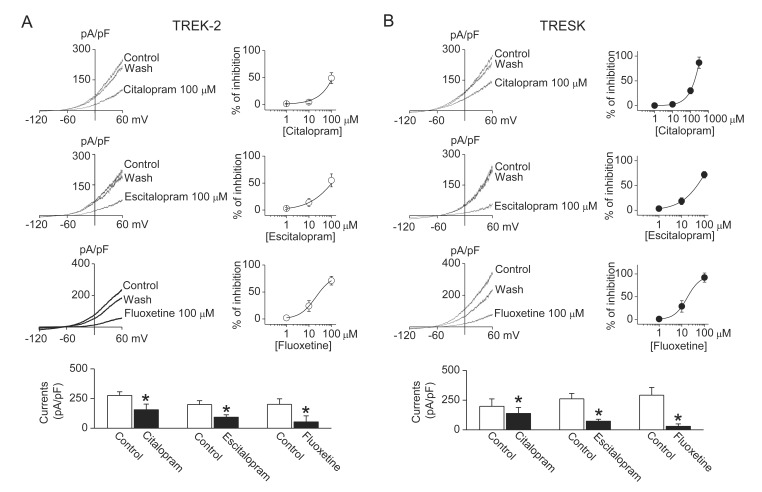
- TWIK-related K+ channel-2 (TREK-2) and TWIK-related spinal cord K+ (TRESK) channel are members of two-pore domain K+ channel family. They are well expressed and help to set the resting membrane potential in sensory neurons. Modulation of TREK-2 and TRESK channels are involved in the pathogenesis of pain, and specifi c activators of TREK-2 and TRESK may be benefi cial for the treatment of pain symptoms. However, the effect of commonly used analgesics on TREK-2 and TRESK channels are not known. Here, we investigated the effect of analgesics on TREK-2 and TRESK channels. The effects of analgesics were examined in HEK cells transfected with TREK-2 or TRESK. Amitriptyline, citalopram, escitalopram, and fluoxetine significantly inhibited TREK-2 and TRESK currents in HEK cells (p<0.05, n=10). Acetaminophen, ibuprofen, nabumetone, and bupropion inhibited TRESK, but had no effect on TREK-2. These results show that all analgesics tested in this study inhibit TRESK activity. Further study is needed to identify the mechanisms by which the analgesics modulate TREK-2 and TRESK differently.
Keyword
MeSH Terms
-
Acetaminophen
Amitriptyline
Analgesics*
Antidepressive Agents*
Bupropion
Citalopram
Fluoxetine
Humans
Ibuprofen
Membrane Potentials
Potassium Channels, Tandem Pore Domain
Sensory Receptor Cells
Spinal Cord
Acetaminophen
Amitriptyline
Analgesics
Antidepressive Agents
Bupropion
Citalopram
Fluoxetine
Ibuprofen
Potassium Channels, Tandem Pore Domain
Figure
Reference
-
1. Global Burden of Disease Study 2013 Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990-2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet. 2015; 386:743–800. PMID: 26063472.2. Hall-Flavin DK, Winner JG, Allen JD, Jordan JJ, Nesheim RS, Snyder KA, Drews MS, Eisterhold LL, Biernacka JM, Mrazek DA. Using a pharmacogenomic algorithm to guide the treatment of depression. Transl Psychiatry. 2012; 2:e172. PMID: 23047243.
Article4. Cohen GL. Migraine prophylactic drugs work via ion channels. Med Hypotheses. 2005; 65:114–122. PMID: 15893128.
Article5. Tsantoulas C, McMahon SB. Opening paths to novel analgesics: the role of potassium channels in chronic pain. Trends Neurosci. 2014; 37:146–158. PMID: 24461875.
Article6. Kang D, Kim D. TREK-2 (K2P10.1) and TRESK (K2P18.1) are major background K+ channels in dorsal root ganglion neurons. Am J Physiol Cell Physiol. 2006; 291:C138–C146. PMID: 16495368.7. Lafrenière RG, Cader MZ, Poulin JF, Andres-Enguix I, Simoneau M, Gupta N, Boisvert K, Lafrenière F, McLaughlan S, Dubé MP, Marcinkiewicz MM, Ramagopalan S, Ansorge O, Brais B, Sequeiros J, Pereira-Monteiro JM, Griffiths LR, Tucker SJ, Ebers G, Rouleau GA. A dominant-negative mutation in the TRESK potassium channel is linked to familial migraine with aura. Nat Med. 2010; 16:1157–1160. PMID: 20871611.
Article8. Pereira V, Busserolles J, Christin M, Devilliers M, Poupon L, Legha W, Alloui A, Aissouni Y, Bourinet E, Lesage F, Eschalier A, Lazdunski M, Noël J. Role of the TREK2 potassium channel in cold and warm thermosensation and in pain perception. Pain. 2014; 155:2534–2544. PMID: 25239074.
Article9. Acosta C, Djouhri L, Watkins R, Berry C, Bromage K, Lawson SN. TREK2 expressed selectively in IB4-binding C-fiber nociceptors hyperpolarizes their membrane potentials and limits spontaneous pain. J Neurosci. 2014; 34:1494–1509. PMID: 24453337.
Article10. Kollert S, Dombert B, Döring F, Wischmeyer E. Activation of TRESK channels by the inflammatory mediator lysophosphatidic acid balances nociceptive signalling. Sci Rep. 2015; 5:12548. PMID: 26224542.
Article11. Liu P, Xiao Z, Ren F, Guo Z, Chen Z, Zhao H, Cao YQ. Functional analysis of a migraine-associated TRESK K+ channel mutation. J Neurosci. 2013; 33:12810–12824. PMID: 23904616.12. Mathie A, Veale EL. Two-pore domain potassium channels: potential therapeutic targets for the treatment of pain. Pflugers Arch. 2015; 467:931–943. PMID: 25420526.
Article13. Wolkerstorfer A, Handler N, Buschmann H. New approaches to treating pain. Bioorg Med Chem Lett. 2016; 26:1103–1119. PMID: 26774577.
Article14. Mika J, Zychowska M, Makuch W, Rojewska E, Przewlocka B. Neuronal and immunological basis of action of antidepressants in chronic pain - clinical and experimental studies. Pharmacol Rep. 2013; 65:1611–1621. PMID: 24553009.
Article15. Khouzam HR. Psychopharmacology of chronic pain: a focus on antidepressants and atypical antipsychotics. Postgrad Med. 2016; 128:323–330. PMID: 26821680.
Article16. Jesse CR, Wilhelm EA, Nogueira CW. Depression-like behavior and mechanical allodynia are reduced by bis selenide treatment in mice with chronic constriction injury: a comparison with fluoxetine, amitriptyline, and bupropion. Psychopharmacology (Berl). 2010; 212:513–522. PMID: 20689938.
Article17. Moore R, Derry S, Aldington D, Cole P, Wiffen PJ. Amitriptyline for neuropathic pain in adults. Cochrane Database Syst Rev. 2015; 7:CD008242. PMID: 26146793.
Article18. Gilron I, Baron R, Jensen T. Neuropathic pain: principles of diagnosis and treatment. Mayo Clin Proc. 2015; 90:532–545. PMID: 25841257.
Article19. Saarto T, Wiffen PJ. Antidepressants for treating neuropathic pain. Cochrane Database Syst Rev. 2007; CD005454. PMID: 17943857.20. Nielsen JC, Bjerring P, Arendt-Nielsen L, Petterson KJ. Analgesic efficacy of immediate and sustained release paracetamol and plasma concentration of paracetamol. Double blind, placebo-controlled evaluation using painful laser stimulation. Eur J Clin Pharmacol. 1992; 42:261–264. PMID: 1577043.
Article21. Mehlisch DR, Sykes J. Ibuprofen blood plasma levels and onset of analgesia. Int J Clin Pract Suppl. 2013; (178):3–8. PMID: 23163542.
Article22. Baeck SK, Lim MA, Park SY, Lee JS, Lee HS, Koo KS. Blood concentrations of amitriptyline and its metabolite in rats after acute oral administration of amitriptyline. J Anal Toxicol. 2000; 24:271–274. PMID: 10872574.
Article23. Laib AK, Brünen S, Pfeifer P, Vincent P, Hiemke C. Serum concentrations of hydroxybupropion for dose optimization of depressed patients treated with bupropion. Ther Drug Monit. 2014; 36:473–479. PMID: 24452068.
Article24. Paulzen M, Gründer G, Veselinovic T, Wolf B, Hiemke C, Lammertz SE. Duloxetine enters the brain - But why is it not found in the cerebrospinal fluid. J Affect Disord. 2016; 189:159–163. PMID: 26437230.
Article25. Reis M, Chermá MD, Carlsson B, Bengtsson F. Therapeutic drug monitoring of escitalopram in an outpatient setting. Ther Drug Monit. 2007; 29:758–766. PMID: 18043473.
Article26. Amsterdam JD, Fawcett J, Quitkin FM, Reimherr FW, Rosenbaum JF, Michelson D, Hornig-Rohan M, Beasley CM. Fluoxetine and norfluoxetine plasma concentrations in major depression: a multicenter study. Am J Psychiatry. 1997; 154:963–969. PMID: 9210747.27. Karson CN, Newton JE, Livingston R, Jolly JB, Cooper TB, Sprigg J, Komoroski RA. Human brain fluoxetine concentrations. J Neuropsychiatry Clin Neurosci. 1993; 5:322–329. PMID: 8369643.28. Brueggemann LI, Mani BK, Mackie AR, Cribbs LL, Byron KL. Novel actions of nonsteroidal anti-inflammatory drugs on vascular ion channels: accounting for cardiovascular side effects and identifying new therapeutic applications. Mol Cell Pharmacol. 2010; 2:15–19. PMID: 20689646.29. Martin DK, Schyvens CG, Wyse KR, Bursill JA, Owe-Young RA, Macdonald PS, Campbell TJ. Role of non-steroidal antiinflammatory drugs (NSAIDs) in modulating vascular smooth muscle cells by activating large-conductance potassium ion channels. Patch Clamp Technique. 2012; 14:283–300.30. Freeman LC, Narvaez DF, McCoy A, von Stein FB, Young S, Silver K, Ganta S, Koch D, Hunter R, Gilmour RF, Lillich JD. Depolarization and decreased surface expression of K+ channels contribute to NSAID-inhibition of intestinal restitution. Biochem Pharmacol. 2007; 74:74–85. PMID: 17499219.31. Heldestad A, Jonsborg S, Henriksson R, Grankvist K. Acetaminophen protection against estramustine-induced cytotoxicity on cultured fibroblasts. Pharmacol Toxicol. 1998; 82:128–131. PMID: 9553990.
Article32. Heurteaux C, Lucas G, Guy N, El Yacoubi M, Thümmler S, Peng XD, Noble F, Blondeau N, Widmann C, Borsotto M, Gobbi G, Vaugeois JM, Debonnel G, Lazdunski M. Deletion of the background potassium channel TREK-1 results in a depression-resistant phenotype. Nat Neurosci. 2006; 9:1134–1141. PMID: 16906152.
Article33. Gotter AL, Santarelli VP, Doran SM, Tannenbaum PL, Kraus RL, Rosahl TW, Meziane H, Montial M, Reiss DR, Wessner K, McCampbell A, Stevens J, Brunner JI, Fox SV, Uebele VN, Bayliss DA, Winrow CJ, Renger JJ. TASK-3 as a potential antidepressant target. Brain Res. 2011; 1416:69–79. PMID: 21885038.
Article34. Borsotto M, Veyssiere J, Moha Ou, Devader C, Mazella J, Heurteaux C. Targeting two-pore domain K+ channels TREK-1 and TASK-3 for the treatment of depression: a new therapeutic concept. Br J Pharmacol. 2015; 172:771–784. PMID: 25263033.35. Kang D, Kim GT, Kim EJ, La JH, Lee JS, Lee ES, Park JY, Hong SG, Han J. Lamotrigine inhibits TRESK regulated by G-protein coupled receptor agonists. Biochem Biophys Res Commun. 2008; 367:609–615. PMID: 18190784.
Article36. Kim CW, Choe C, Kim EJ, Lee JI, Yoon SY, Cho YW, Han S, Tak HM, Han J, Kang D. Dual effects of fluoxetine on mouse early embryonic development. Toxicol Appl Pharmacol. 2012; 265:61–72. PMID: 23022515.
Article37. Semenchuk MR, Davis B. Efficacy of sustained-release bupropion in neuropathic pain: an open-label study. Clin J Pain. 2000; 16:6–11. PMID: 10741812.
Article38. Vázquez-Gómez E, Arias HR, Feuerbach D, Miranda-Morales M, Mihailescu S, Targowska-Duda KM, Jozwiak K, García-Colunga J. Bupropion-induced inhibition of α7 nicotinic acetylcholine receptors expressed in heterologous cells and neurons from dorsalraphe nucleus and hippocampus. Eur J Pharmacol. 2014; 740:103–111. PMID: 25016090.39. Yang Y, Shimomura K, Sakuma K, Maejima Y, Iwasaki Y, Galvanovskis J, Dezaki K, Nakata M, Yada T. Bupropion can close KATP channel and induce insulin secretion. J Pediatr Endocrinol Metab. 2013; 26:343–346. PMID: 23348218.
Article40. Kang D, Han J, Kim D. Mechanism of inhibition of TREK-2 (K2P10.1) by the Gq-coupled M3 muscarinic receptor. Am J Physiol Cell Physiol. 2006; 291:C649–C656. PMID: 16672694.
Article41. Czirják G, Tóth ZE, Enyedi P. The two-pore domain K+ channel, TRESK, is activated by the cytoplasmic calcium signal through calcineurin. J Biol Chem. 2004; 279:18550–18558. PMID: 14981085.42. Veale EL, Mathie A. Aristolochic acid, a plant extract used in the treatment of pain and linked to Balkan endemic nephropathy, is a regulator of K2P channels. Br J Pharmacol. 2016; 173:1639–1652. PMID: 26914156.
Article43. Bang H, Kim Y, Kim D. TREK-2, a new member of the mechanosensitive tandem-pore K+ channel family. J Biol Chem. 2000; 275:17412–17419. PMID: 10747911.44. Kang D, Mariash E, Kim D. Functional expression of TRESK-2, a new member of the tandem-pore K+ channel family. J Biol Chem. 2004; 279:28063–28070. PMID: 15123670.45. Rahm AK, Gierten J, Kisselbach J, Staudacher I, Staudacher K, Schweizer PA, Becker R, Katus HA, Thomas D. PKC-dependent activation of human K2P 18.1 K+ channels. Br J Pharmacol. 2012; 166:764–773. PMID: 22168364.46. Enyedi P, Czirják G. Properties, regulation, pharmacology, andfunctions of the K2p channel, TRESK. Pflugers Arch. 2015; 467:945–945. PMID: 25366493.47. Callejo G, Giblin JP, Gasull X. Modulation of TRESK background K+ channel by membrane stretch. PLoS One. 2013; 8:e64471. PMID: 23691227.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Volatile General Anesthetics and Tandem Pore Domain K+ Channel
- Enhanced Expression of TREK-1 Is Related with Chronic Constriction Injury of Neuropathic Pain Mouse Model in Dorsal Root Ganglion
- Fluoxetine inhibits L-type Ca2+ and transient outward K+ currents in rat ventricular myocytes
- TWIK-Related Spinal Cord K+ Channel Expression Is Increased in the Spinal Dorsal Horn after Spinal Nerve Ligation
- Current Pharmacotherapy of Depression