Cancer Res Treat.
2017 Jan;49(1):129-140. 10.4143/crt.2015.466.
Post-bevacizumab Clinical Outcomes and the Impact of Early Discontinuation of Bevacizumab in Patients with Recurrent Malignant Glioma
- Affiliations
-
- 1Department of Internal Medicine, Seoul National University Hospital, Seoul, Korea. sehoon.lee119@gmail.com
- 2Department of Internal Medicine, Seoul National University Bundang Hospital, Seongnam, Korea.
- 3Division of Hematology/Oncology, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- 4Department of Radiology, Seoul National University Hospital, Seoul, Korea.
- 5Department of Neurosurgery, Seoul National University Hospital, Seoul, Korea.
- 6Department of Radiation Oncology, Seoul National University Hospital, Seoul, Korea.
- 7Department of Radiology, Seoul National University Bundang Hospital, Seongnam, Korea.
- 8Department of Neurosurgery, Seoul National University Bundang Hospital, Seongnam, Korea.
- 9Department of Radiation Oncology, Seoul National University Bundang Hospital, Seongnam, Korea.
- KMID: 2367510
- DOI: http://doi.org/10.4143/crt.2015.466
Abstract
- PURPOSE
Bevacizumab±irinotecan is effective for treatment of recurrent malignant gliomas. However, the optimal duration of treatment has not been established.
MATERIALS AND METHODS
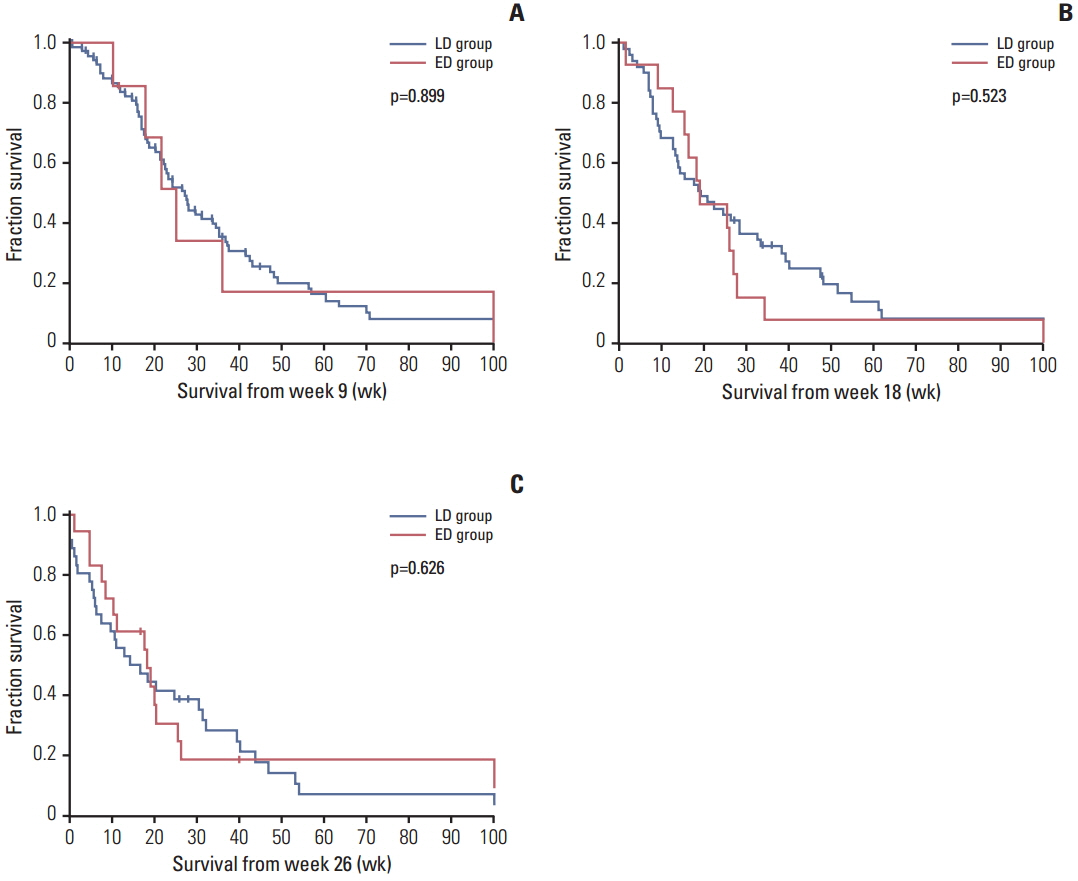
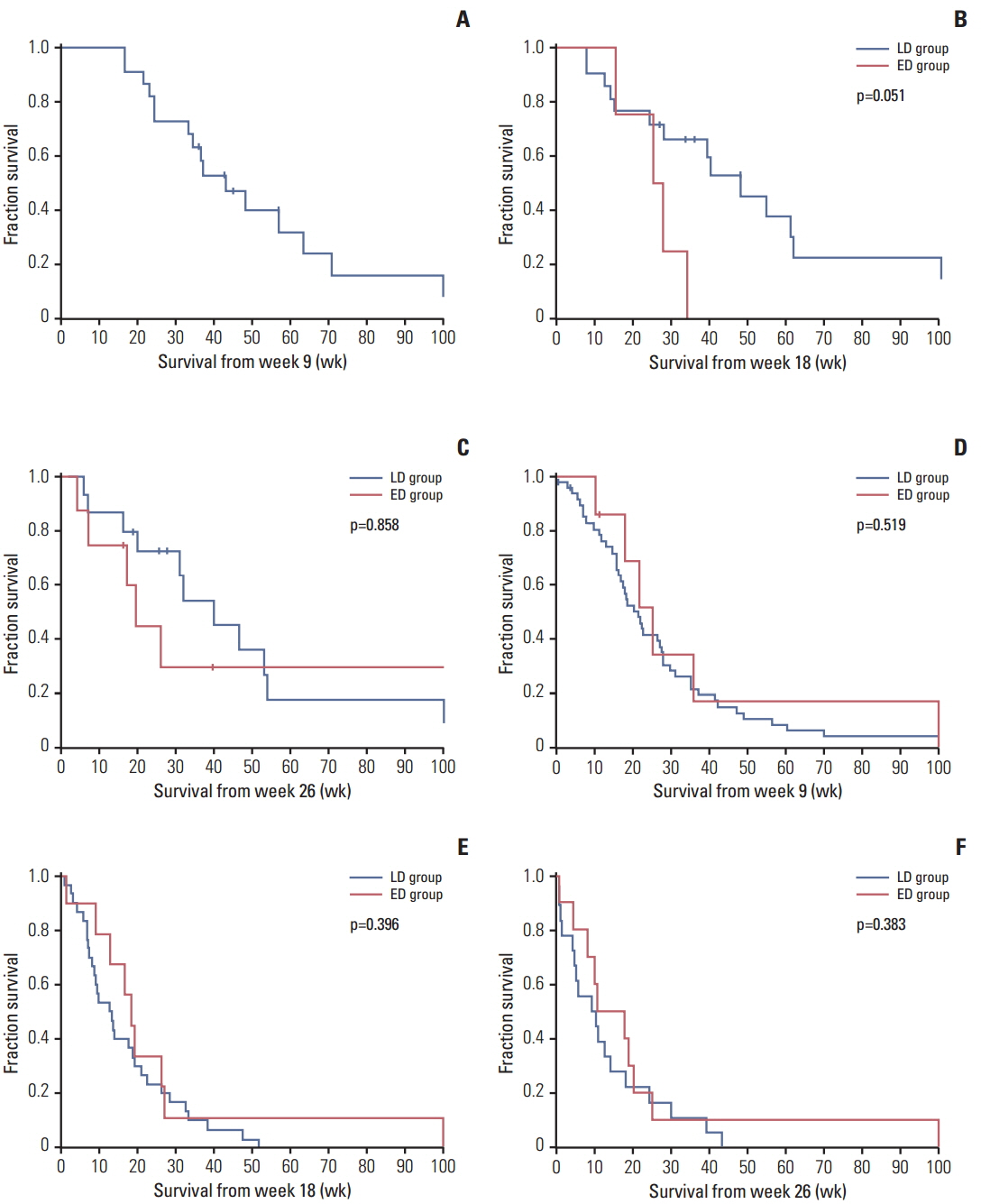
Ninety-four consecutive patients with recurrent malignant glioma who were treated with bevacizumab at our institutions were identified. Patients who continued bevacizumab until tumor progression were enrolled in a late discontinuation (LD) group, while those who stopped bevacizumab before tumor progression were enrolled in an early discontinuation (ED) group. Landmark analyses were performed at weeks 9, 18, and 26 for comparison of patient survival between the two groups.
RESULTS
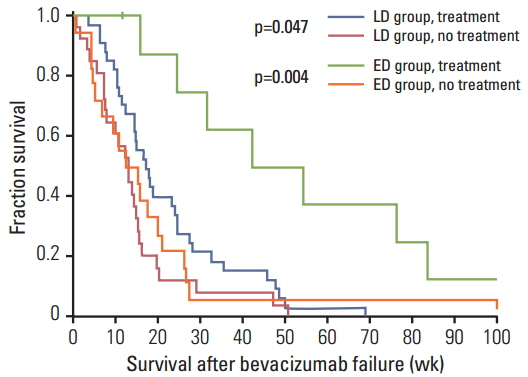
Among 89 assessable patients, 62 (69.7%) and 27 (30.3%) patients were categorized as the LD and ED groups, respectively. According to landmark analysis, survival times from weeks 9, 18, and 26 were not significantly different between the two groups in the overall population. However, the LD group showed a trend toward increased survival compared to the ED group among responders. In the ED group, the median time from discontinuation to disease progression was 11.4 weeks, and none of the patients showed a definite rebound phenomenon. Similar median survival times after disease progression were observed between groups (14.4 weeks vs. 15.7 weeks, p=0.251). Of 83 patients, 38 (45.8%) received further therapy at progression, and those who received further therapy showed longer survival in both the LD and ED groups.
CONCLUSION
In recurrent malignant glioma, duration of bevacizumab was not associated with survival time in the overall population. However, ED of bevacizumab in responding patients might be associated with decreased survival.
Keyword
Figure
Reference
-
References
1. Norden AD, Drappatz J, Wen PY. Antiangiogenic therapies for high-grade glioma. Nat Rev Neurol. 2009; 5:610–20.
Article2. Vredenburgh JJ, Desjardins A, Herndon JE 2nd, Marcello J, Reardon DA, Quinn JA, et al. Bevacizumab plus irinotecan in recurrent glioblastoma multiforme. J Clin Oncol. 2007; 25:4722–9.
Article3. Vredenburgh JJ, Desjardins A, Herndon JE 2nd, Dowell JM, Reardon DA, Quinn JA, et al. Phase II trial of bevacizumab and irinotecan in recurrent malignant glioma. Clin Cancer Res. 2007; 13:1253–9.
Article4. Kreisl TN, Kim L, Moore K, Duic P, Royce C, Stroud I, et al. Phase II trial of single-agent bevacizumab followed by bevacizumab plus irinotecan at tumor progression in recurrent glioblastoma. J Clin Oncol. 2009; 27:740–5.
Article5. Friedman HS, Prados MD, Wen PY, Mikkelsen T, Schiff D, Abrey LE, et al. Bevacizumab alone and in combination with irinotecan in recurrent glioblastoma. J Clin Oncol. 2009; 27:4733–40.
Article6. Mrugala MM. Advances and challenges in the treatment of glioblastoma: a clinician's perspective. Discov Med. 2013; 15:221–30.7. Wick W, Weller M, van den Bent M, Stupp R. Bevacizumab and recurrent malignant gliomas: a European perspective. J Clin Oncol. 2010; 28:e188–9.
Article8. Kesselheim JC, Norden AD, Wen PY, Joffe S. Discontinuing bevacizumab in patients with glioblastoma: an ethical analysis. Oncologist. 2011; 16:1435–9.
Article9. Zuniga RM, Torcuator R, Jain R, Anderson J, Doyle T, Schultz L, et al. Rebound tumour progression after the cessation of bevacizumab therapy in patients with recurrent high-grade glioma. J Neurooncol. 2010; 99:237–42.
Article10. Sherman WJ, Raizer JJ, Grimm SA. Outcome of discontinuing bevacizumab prior to malignant glioma progression. J Neurooncol. 2013; 111:87–9.
Article11. Ranpura V, Hapani S, Wu S. Treatment-related mortality with bevacizumab in cancer patients: a meta-analysis. JAMA. 2011; 305:487–94.12. Brandsma D, Stalpers L, Taal W, Sminia P, van den Bent MJ. Clinical features, mechanisms, and management of pseudoprogression in malignant gliomas. Lancet Oncol. 2008; 9:453–61.
Article13. Wen PY, Macdonald DR, Reardon DA, Cloughesy TF, Sorensen AG, Galanis E, et al. Updated response assessment criteria for high-grade gliomas: response assessment in neurooncology working group. J Clin Oncol. 2010; 28:1963–72.
Article14. Anderson JR, Cain KC, Gelber RD. Analysis of survival by tumor response. J Clin Oncol. 1983; 1:710–9.
Article15. Griffioen AW, Mans LA, de Graaf AM, Nowak-Sliwinska P, de Hoog CL, de Jong TA, et al. Rapid angiogenesis onset after discontinuation of sunitinib treatment of renal cell carcinoma patients. Clin Cancer Res. 2012; 18:3961–71.
Article16. Grothey A, Sugrue MM, Purdie DM, Dong W, Sargent D, Hedrick E, et al. Bevacizumab beyond first progression is associated with prolonged overall survival in metastatic colorectal cancer: results from a large observational cohort study (BRiTE). J Clin Oncol. 2008; 26:5326–34.
Article17. Reardon DA, Herndon JE 2nd, Peters KB, Desjardins A, Coan A, Lou E, et al. Bevacizumab continuation beyond initial bevacizumab progression among recurrent glioblastoma patients. Br J Cancer. 2012; 107:1481–7.
Article18. Jain RK. Normalizing tumor microenvironment to treat cancer: bench to bedside to biomarkers. J Clin Oncol. 2013; 31:2205–18.
Article19. de Groot JF. High-dose antiangiogenic therapy for glioblastoma: less may be more? Clin Cancer Res. 2011; 17:6109–11.20. Paez-Ribes M, Allen E, Hudock J, Takeda T, Okuyama H, Vinals F, et al. Antiangiogenic therapy elicits malignant progression of tumors to increased local invasion and distant metastasis. Cancer Cell. 2009; 15:220–31.
Article21. de Groot JF, Fuller G, Kumar AJ, Piao Y, Eterovic K, Ji Y, et al. Tumor invasion after treatment of glioblastoma with bevacizumab: radiographic and pathologic correlation in humans and mice. Neuro Oncol. 2010; 12:233–42.
Article22. Norden AD, Young GS, Setayesh K, Muzikansky A, Klufas R, Ross GL, et al. Bevacizumab for recurrent malignant gliomas: efficacy, toxicity, and patterns of recurrence. Neurology. 2008; 70:779–87.
Article23. Iwamoto FM, Abrey LE, Beal K, Gutin PH, Rosenblum MK, Reuter VE, et al. Patterns of relapse and prognosis after bevacizumab failure in recurrent glioblastoma. Neurology. 2009; 73:1200–6.
Article24. Prados M, Cloughesy T, Samant M, Fang L, Wen PY, Mikkelsen T, et al. Response as a predictor of survival in patients with recurrent glioblastoma treated with bevacizumab. Neuro Oncol. 2011; 13:143–51.
Article25. Miles D, Harbeck N, Escudier B, Hurwitz H, Saltz L, Van Cutsem E, et al. Disease course patterns after discontinuation of bevacizumab: pooled analysis of randomized phase III trials. J Clin Oncol. 2011; 29:83–8.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Radiological Recurrence Patterns after Bevacizumab Treatment of Recurrent High-Grade Glioma: A Systematic Review and Meta-Analysis
- Bevacizumab in Recurrent Glioma: Patterns of Treatment Failure and Implications
- Recurrent Conjunctival Granuloma Treated with Subconjunctival Bevacizumab
- Clinical Results After Application of Bevacizumab in Recurrent Pterygium
- Letter to the Editor: Commentary on “Bevacizumab Alone Versus Bevacizumab Plus Irinotecan in Patients With Recurrent Glioblastoma”