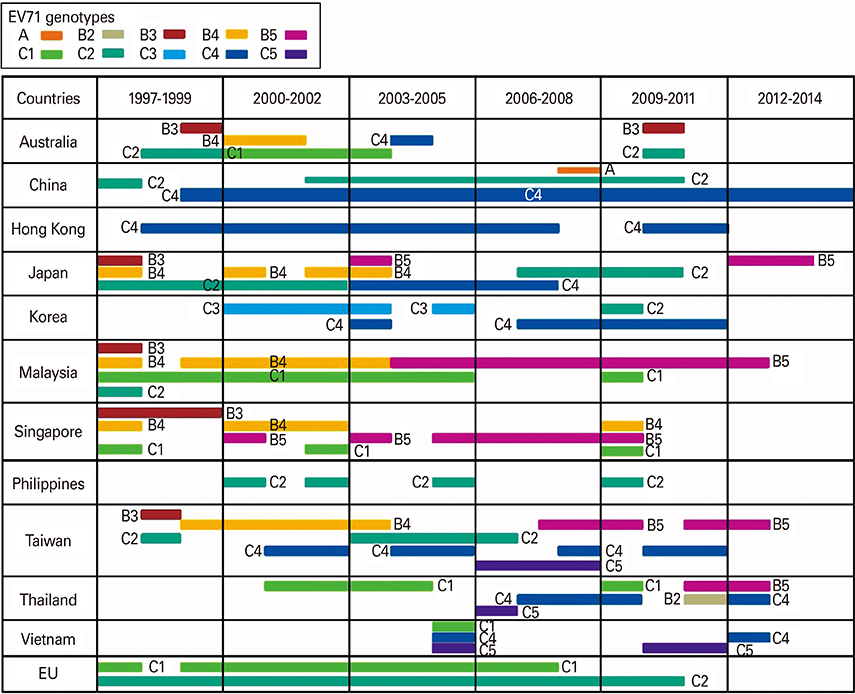
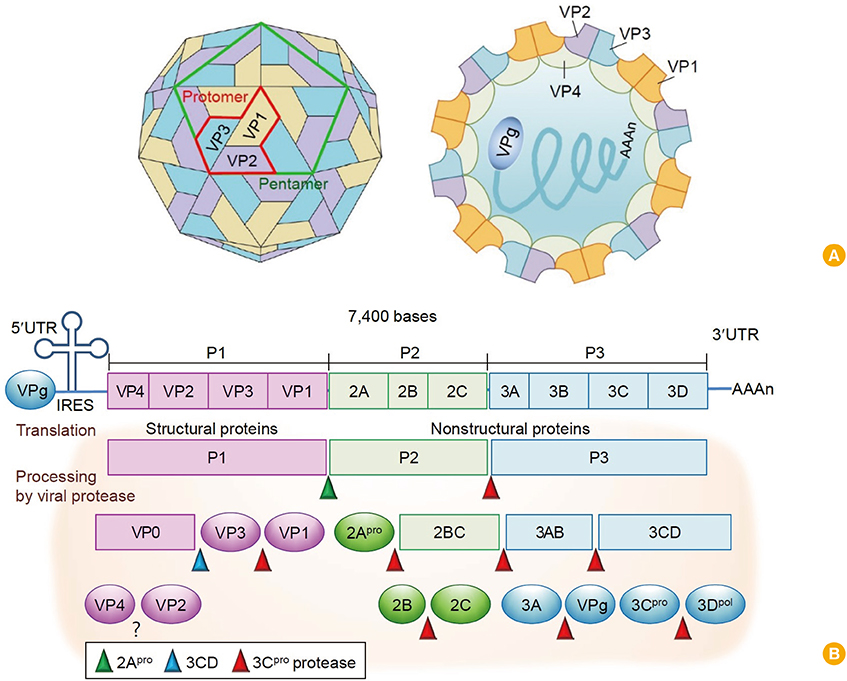
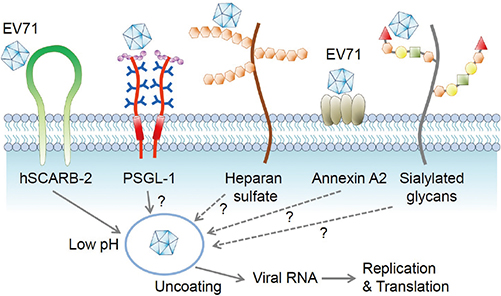
Clin Exp Vaccine Res.
2017 Jan;6(1):4-14. 10.7774/cevr.2017.6.1.4.
Enterovirus 71 infection and vaccines
- Affiliations
-
- 1Laboratory of Microbiology, College of Pharmacy, Ajou University, Suwon, Korea. sychang@ajou.ac.kr
- 2Research Institute of Pharmaceutical Science and Technology (RIPST), Ajou University, Suwon, Korea.
- KMID: 2366886
- DOI: http://doi.org/10.7774/cevr.2017.6.1.4
Abstract
- Hand, foot and mouth disease (HFMD) is a highly contagious viral infection affecting young children during the spring to fall seasons. Recently, serious outbreaks of HFMD were reported frequently in the Asia-Pacific region, including China and Korea. The symptoms of HFMD are usually mild, comprising fever, loss of appetite, and a rash with blisters, which do not need specific treatment. However, there are uncommon neurological or cardiac complications such as meningitis and acute flaccid paralysis that can be fatal. HFMD is most commonly caused by infection with coxsackievirus A16, and secondly by enterovirus 71 (EV71). Many other strains of coxsackievirus and enterovirus can also cause HFMD. Importantly, HFMD caused by EV71 tends to be associated with fatal complications. Therefore, there is an urgent need to protect against EV71 infection. Development of vaccines against EV71 would be the most effective approach to prevent EV71 outbreaks. Here, we summarize EV71 infection and development of vaccines, focusing on current scientific and clinical progress.
Keyword
MeSH Terms
Figure
Reference
-
1. Goksugur N, Goksugur S. Images in clinical medicine. Hand, foot, and mouth disease. N Engl J Med. 2010; 362:e49.2. Ooi MH, Wong SC, Lewthwaite P, Cardosa MJ, Solomon T. Clinical features, diagnosis, and management of enterovirus 71. Lancet Neurol. 2010; 9:1097–1105.
Article3. Omana-Cepeda C, Martinez-Valverde A, del Mar Sabater-Recolons M, Jane-Salas E, Mari-Roig A, Lopez-Lopez J. A literature review and case report of hand, foot and mouth disease in an immunocompetent adult. BMC Res Notes. 2016; 9:165.
Article4. Zou XN, Zhang XZ, Wang B, Qiu YT. Etiologic and epidemiologic analysis of hand, foot, and mouth disease in Guangzhou city: a review of 4,753 cases. Braz J Infect Dis. 2012; 16:457–465.
Article5. Solomon T, Lewthwaite P, Perera D, Cardosa MJ, McMinn P, Ooi MH. Virology, epidemiology, pathogenesis, and control of enterovirus 71. Lancet Infect Dis. 2010; 10:778–790.
Article6. Kaminska K, Martinetti G, Lucchini R, Kaya G, Mainetti C. Coxsackievirus A6 and hand, foot and mouth disease: three case reports of familial child-to-immunocompetent adult transmission and a literature review. Case Rep Dermatol. 2013; 5:203–209.
Article7. Chumakov M, Voroshilova M, Shindarov L, et al. Enterovirus 71 isolated from cases of epidemic poliomyelitis-like disease in Bulgaria. Arch Virol. 1979; 60:329–340.
Article8. Kim BI, Ki H, Park S, Cho E, Chun BC. Effect of climatic factors on hand, foot, and mouth disease in South Korea, 2010-2013. PLoS One. 2016; 11:e0157500.
Article9. Zhou F, Kong F, Wang B, McPhie K, Gilbert GL, Dwyer DE. Molecular characterization of enterovirus 71 and coxsackievirus A16 using the 5’ untranslated region and VP1 region. J Med Microbiol. 2011; 60(Pt 3):349–358.
Article10. Duff MF. Hand-foot-and-mouth syndrome in humans: coxackie A10 infections in New Zealand. Br Med J. 1968; 2:661–664.
Article11. Chong P, Liu CC, Chow YH, Chou AH, Klein M. Review of enterovirus 71 vaccines. Clin Infect Dis. 2015; 60:797–803.
Article12. Wang SM, Ho TS, Lin HC, Lei HY, Wang JR, Liu CC. Reemerging of enterovirus 71 in Taiwan: the age impact on disease severity. Eur J Clin Microbiol Infect Dis. 2012; 31:1219–1224.
Article13. Cho HK, Lee NY, Lee H, et al. Enterovirus 71-associated hand, foot and mouth diseases with neurologic symptoms, a university hospital experience in Korea, 2009. Korean J Pediatr. 2010; 53:639–643.
Article14. Chen B, Sumi A, Toyoda S, et al. Time series analysis of reported cases of hand, foot, and mouth disease from 2010 to 2013 in Wuhan, China. BMC Infect Dis. 2015; 15:495.
Article15. Huang SW, Hsu YW, Smith DJ, et al. Reemergence of enterovirus 71 in 2008 in taiwan: dynamics of genetic and antigenic evolution from 1998 to 2008. J Clin Microbiol. 2009; 47:3653–3662.
Article16. Donato C, Hoi le T, Hoa NT, et al. Genetic characterization of Enterovirus 71 strains circulating in Vietnam in 2012. Virology. 2016; 495:1–9.
Article17. Kim SJ, Kim JH, Kang JH, et al. Risk factors for neurologic complications of hand, foot and mouth disease in the Republic of Korea, 2009. J Korean Med Sci. 2013; 28:120–127.
Article18. Yu H, Chen W, Chang H, et al. Genetic analysis of the VP1 region of enterovirus 71 reveals the emergence of genotype A in central China in 2008. Virus Genes. 2010; 41:1–4.
Article19. Wang X, Peng W, Ren J, et al. A sensor-adaptor mechanism for enterovirus uncoating from structures of EV71. Nat Struct Mol Biol. 2012; 19:424–429.
Article20. Fields BN, Knipe DM, Howley PM. Fields virology. Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins;2007.21. Huang SW, Cheng HL, Hsieh HY, et al. Mutations in the non-structural protein region contribute to intra-genotypic evolution of enterovirus 71. J Biomed Sci. 2014; 21:33.
Article22. Ma HC, Liu Y, Wang C, et al. An interaction between glutathione and the capsid is required for the morphogenesis of C-cluster enteroviruses. PLoS Pathog. 2014; 10:e1004052.
Article23. Rossmann MG, Arnold E, Erickson JW, et al. Structure of a human common cold virus and functional relationship to other picornaviruses. Nature. 1985; 317:145–153.
Article24. Tee KK, Lam TT, Chan YF, et al. Evolutionary genetics of human enterovirus 71: origin, population dynamics, natural selection, and seasonal periodicity of the VP1 gene. J Virol. 2010; 84:3339–3350.
Article25. Lin JY, Li ML, Shih SR. Far upstream element binding protein 2 interacts with enterovirus 71 internal ribosomal entry site and negatively regulates viral translation. Nucleic Acids Res. 2009; 37:47–59.
Article26. Lin JY, Shih SR, Pan M, et al. hnRNP A1 interacts with the 5’ untranslated regions of enterovirus 71 and Sindbis virus RNA and is required for viral replication. J Virol. 2009; 83:6106–6114.
Article27. Yamayoshi S, Yamashita Y, Li J, et al. Scavenger receptor B2 is a cellular receptor for enterovirus 71. Nat Med. 2009; 15:798–801.
Article28. Nishimura Y, Shimojima M, Tano Y, Miyamura T, Wakita T, Shimizu H. Human P-selectin glycoprotein ligand-1 is a functional receptor for enterovirus 71. Nat Med. 2009; 15:794–797.
Article29. Yang SL, Chou YT, Wu CN, Ho MS. Annexin II binds to capsid protein VP1 of enterovirus 71 and enhances viral infectivity. J Virol. 2011; 85:11809–11820.
Article30. Tan CW, Poh CL, Sam IC, Chan YF. Enterovirus 71 uses cell surface heparan sulfate glycosaminoglycan as an attachment receptor. J Virol. 2013; 87:611–620.
Article31. Yang B, Chuang H, Yang KD. Sialylated glycans as receptor and inhibitor of enterovirus 71 infection to DLD-1 intestinal cells. Virol J. 2009; 6:141.
Article32. Ren XX, Ma L, Liu QW, et al. The molecule of DC-SIGN captures enterovirus 71 and confers dendritic cell-mediated viral trans-infection. Virol J. 2014; 11:47.
Article33. Yamayoshi S, Fujii K, Koike S. Receptors for enterovirus 71. Emerg Microbes Infect. 2014; 3:e53.
Article34. Yamayoshi S, Iizuka S, Yamashita T, et al. Human SCARB2-dependent infection by coxsackievirus A7, A14, and A16 and enterovirus 71. J Virol. 2012; 86:5686–5696.
Article35. Patel KP, Bergelson JM. Receptors identified for hand, foot and mouth virus. Nat Med. 2009; 15:728–729.
Article36. Nishimura Y, Lee H, Hafenstein S, et al. Enterovirus 71 binding to PSGL-1 on leukocytes: VP1-145 acts as a molecular switch to control receptor interaction. PLoS Pathog. 2013; 9:e1003511.
Article37. Lin YW, Wang SW, Tung YY, Chen SH. Enterovirus 71 infection of human dendritic cells. Exp Biol Med (Maywood). 2009; 234:1166–1173.
Article38. Chen P, Song Z, Qi Y, et al. Molecular determinants of enterovirus 71 viral entry: cleft around GLN-172 on VP1 protein interacts with variable region on scavenge receptor B 2. J Biol Chem. 2012; 287:6406–6420.39. Yi L, Lu J, Kung HF, He ML. The virology and developments toward control of human enterovirus 71. Crit Rev Microbiol. 2011; 37:313–327.
Article40. Liu L, Zhao H, Zhang Y, et al. Neonatal rhesus monkey is a potential animal model for studying pathogenesis of EV71 infection. Virology. 2011; 412:91–100.
Article41. Khong WX, Yan B, Yeo H, et al. A non-mouse-adapted enterovirus 71 (EV71) strain exhibits neurotropism, causing neurological manifestations in a novel mouse model of EV71 infection. J Virol. 2012; 86:2121–2131.
Article42. Kim YI, Song JH, Kwon BE, et al. Pros and cons of VP1-specific maternal IgG for the protection of Enterovirus 71 infection. Vaccine. 2015; 33:6604–6610.
Article43. van den Broek MF, Muller U, Huang S, Aguet M, Zinkernagel RM. Antiviral defense in mice lacking both alpha/beta and gamma interferon receptors. J Virol. 1995; 69:4792–4796.
Article44. Liu ML, Lee YP, Wang YF, et al. Type I interferons protect mice against enterovirus 71 infection. J Gen Virol. 2005; 86(Pt 12):3263–3269.
Article45. Fujii K, Nagata N, Sato Y, et al. Transgenic mouse model for the study of enterovirus 71 neuropathogenesis. Proc Natl Acad Sci U S A. 2013; 110:14753–14758.
Article46. Lin YW, Yu SL, Shao HY, et al. Human SCARB2 transgenic mice as an infectious animal model for enterovirus 71. PLoS One. 2013; 8:e57591.
Article47. Chang HW, Lin YW, Ho HM, et al. Protective efficacy of VP1-specific neutralizing antibody associated with a reduction of viral load and pro-inflammatory cytokines in human SCARB2-transgenic mice. PLoS One. 2013; 8:e69858.
Article48. Wu CN, Lin YC, Fann C, Liao NS, Shih SR, Ho MS. Protection against lethal enterovirus 71 infection in newborn mice by passive immunization with subunit VP1 vaccines and inactivated virus. Vaccine. 2001; 20:895–904.
Article49. Wang M, Jiang S, Wang Y. Recombinant VP1 protein expressed in Pichia pastoris induces protective immune responses against EV71 in mice. Biochem Biophys Res Commun. 2013; 430:387–393.
Article50. Premanand B, Kiener TK, Meng T, et al. Induction of protective immune responses against EV71 in mice by baculovirus encoding a novel expression cassette for capsid protein VP1. Antiviral Res. 2012; 95:311–315.
Article51. Foo DG, Alonso S, Phoon MC, Ramachandran NP, Chow VT, Poh CL. Identification of neutralizing linear epitopes from the VP1 capsid protein of enterovirus 71 using synthetic peptides. Virus Res. 2007; 125:61–68.
Article52. Chung YC, Ho MS, Wu JC, et al. Immunization with virus-like particles of enterovirus 71 elicits potent immune responses and protects mice against lethal challenge. Vaccine. 2008; 26:1855–1862.
Article53. Sun S, Gao F, Mao Q, et al. Immunogenicity and protective efficacy of an EV71 virus-like particle vaccine against lethal challenge in newborn mice. Hum Vaccin Immunother. 2015; 11:2406–2413.
Article54. Zhao H, Li HY, Han JF, et al. Novel recombinant chimeric virus-like particle is immunogenic and protective against both enterovirus 71 and coxsackievirus A16 in mice. Sci Rep. 2015; 5:7878.
Article55. Ye X, Ku Z, Liu Q, et al. Chimeric virus-like particle vaccines displaying conserved enterovirus 71 epitopes elicit protective neutralizing antibodies in mice through divergent mechanisms. J Virol. 2014; 88:72–81.
Article56. Tung WS, Bakar SA, Sekawi Z, Rosli R. DNA vaccine constructs against enterovirus 71 elicit immune response in mice. Genet Vaccines Ther. 2007; 5:6.
Article57. Chiu CH, Chu C, He CC, Lin TY. Protection of neonatal mice from lethal enterovirus 71 infection by maternal immunization with attenuated Salmonella enterica serovar Typhimurium expressing VP1 of enterovirus 71. Microbes Infect. 2006; 8:1671–1678.
Article58. Tsou YL, Lin YW, Shao HY, et al. Recombinant adeno-vaccine expressing enterovirus 71-like particles against hand, foot, and mouth disease. PLoS Negl Trop Dis. 2015; 9:e0003692.
Article59. Chen HF, Chang MH, Chiang BL, Jeng ST. Oral immunization of mice using transgenic tomato fruit expressing VP1 protein from enterovirus 71. Vaccine. 2006; 24:2944–2951.
Article60. Arita M, Nagata N, Iwata N, et al. An attenuated strain of enterovirus 71 belonging to genotype a showed a broad spectrum of antigenicity with attenuated neurovirulence in cynomolgus monkeys. J Virol. 2007; 81:9386–9395.
Article61. Meng T, Kwang J. Attenuation of human enterovirus 71 high-replication-fidelity variants in AG129 mice. J Virol. 2014; 88:5803–5815.
Article62. Ong KC, Devi S, Cardosa MJ, Wong KT. Formaldehyde-inactivated whole-virus vaccine protects a murine model of enterovirus 71 encephalomyelitis against disease. J Virol. 2010; 84:661–665.
Article63. Liu L, Zhang Y, Wang J, et al. Study of the integrated immune response induced by an inactivated EV71 vaccine. PLoS One. 2013; 8:e54451.
Article64. Chou AH, Liu CC, Chang JY, et al. Formalin-inactivated EV71 vaccine candidate induced cross-neutralizing antibody against subgenotypes B1, B4, B5 and C4A in adult volunteers. PLoS One. 2013; 8:e79783.
Article65. Zhu FC, Meng FY, Li JX, et al. Efficacy, safety, and immunology of an inactivated alum-adjuvant enterovirus 71 vaccine in children in China: a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2013; 381:2024–2032.
Article66. Zhu F, Xu W, Xia J, et al. Efficacy, safety, and immunogenicity of an enterovirus 71 vaccine in China. N Engl J Med. 2014; 370:818–828.
Article67. Li R, Liu L, Mo Z, et al. An inactivated enterovirus 71 vaccine in healthy children. N Engl J Med. 2014; 370:829–837.
Article68. Kuo RL, Shih SR. Strategies to develop antivirals against enterovirus 71. Virol J. 2013; 10:28.
Article69. Zhang G, Zhou F, Gu B, et al. In vitro and in vivo evaluation of ribavirin and pleconaril antiviral activity against enterovirus 71 infection. Arch Virol. 2012; 157:669–679.
Article70. Shih SR, Tsai MC, Tseng SN, et al. Mutation in enterovirus 71 capsid protein VP1 confers resistance to the inhibitory effects of pyridyl imidazolidinone. Antimicrob Agents Chemother. 2004; 48:3523–3529.
Article71. Weng TY, Chen LC, Shyu HW, et al. Lactoferrin inhibits enterovirus 71 infection by binding to VP1 protein and host cells. Antiviral Res. 2005; 67:31–37.
Article72. Matthews DA, Dragovich PS, Webber SE, et al. Structure-assisted design of mechanism-based irreversible inhibitors of human rhinovirus 3C protease with potent antiviral activity against multiple rhinovirus serotypes. Proc Natl Acad Sci U S A. 1999; 96:11000–11007.
Article73. Lu G, Qi J, Chen Z, et al. Enterovirus 71 and coxsackievirus A16 3C proteases: binding to rupintrivir and their substrates and anti-hand, foot, and mouth disease virus drug design. J Virol. 2011; 85:10319–10331.
Article74. Kishimoto C, Crumpacker CS, Abelmann WH. Ribavirin treatment of murine coxsackievirus B3 myocarditis with analyses of lymphocyte subsets. J Am Coll Cardiol. 1988; 12:1334–1341.
Article75. Chen TC, Chang HY, Lin PF, et al. Novel antiviral agent DTriP-22 targets RNA-dependent RNA polymerase of enterovirus 71. Antimicrob Agents Chemother. 2009; 53:2740–2747.
Article76. Arita M, Takebe Y, Wakita T, Shimizu H. A bifunctional anti-enterovirus compound that inhibits replication and the early stage of enterovirus 71 infection. J Gen Virol. 2010; 91:2734–2744.
Article77. Tsai FJ, Lin CW, Lai CC, et al. Kaempferol inhibits enterovirus 71 replication and internal ribosome entry site (IRES) activity through FUBP and HNRP proteins. Food Chem. 2011; 128:312–322.
Article78. Tan EL, Tan TM, Tak Kwong Chow V, Poh CL. Inhibition of enterovirus 71 in virus-infected mice by RNA interference. Mol Ther. 2007; 15:1931–1938.
Article79. Ng Q, He F, Kwang J. Recent progress towards novel EV71 anti-therapeutics and vaccines. Viruses. 2015; 7:6441–6457.
Article80. Lim XF, Jia Q, Khong WX, et al. Characterization of an isotype-dependent monoclonal antibody against linear neutralizing epitope effective for prophylaxis of enterovirus 71 infection. PLoS One. 2012; 7:e29751.
Article81. Kiener TK, Jia Q, Meng T, Chow VT, Kwang J. A novel universal neutralizing monoclonal antibody against enterovirus 71 that targets the highly conserved “knob” region of VP3 protein. PLoS Negl Trop Dis. 2014; 8:e2895.
Article82. Klein MH. EV71 vaccines: a first step towards multivalent hand, foot and mouth disease vaccines. Expert Rev Vaccines. 2015; 14:337–340.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Paralytic Disease Related to Enterovirus 71 Infection: A report of 4 cases
- A case of Enterovirus 71 Infection Presented with Acute Flaccid Paralysis in Jeju Island
- Enterovirus 71 infection and neurological complications
- Experimental animal models for development of human enterovirus vaccine
- Neurological Manifestations and Experience with IV Immunoglobulin in Children with Enterovirus 71 Infections