Investig Magn Reson Imaging.
2016 Dec;20(4):241-249. 10.13104/imri.2016.20.4.241.
Added Value of Diffusion Weighted Imaging for Detecting Pancreatic Abnormality in Patients with Clinically Suspected Acute Pancreatitis
- Affiliations
-
- 1Department of Radiology, Inje University College of Medicine, Haeundae Paik Hospital, Busan, Korea. radiresi@gmail.com
- 2Department of Radiology, Myongji Hospital, Goyang-si, Korea.
- KMID: 2366406
- DOI: http://doi.org/10.13104/imri.2016.20.4.241
Abstract
- PURPOSE
To evaluate the added value of diffusion weighted imaging (DWI) to computed tomography (CT) for detecting pancreatic abnormality in patients with clinically suspected acute pancreatitis (AP).
MATERIALS AND METHODS
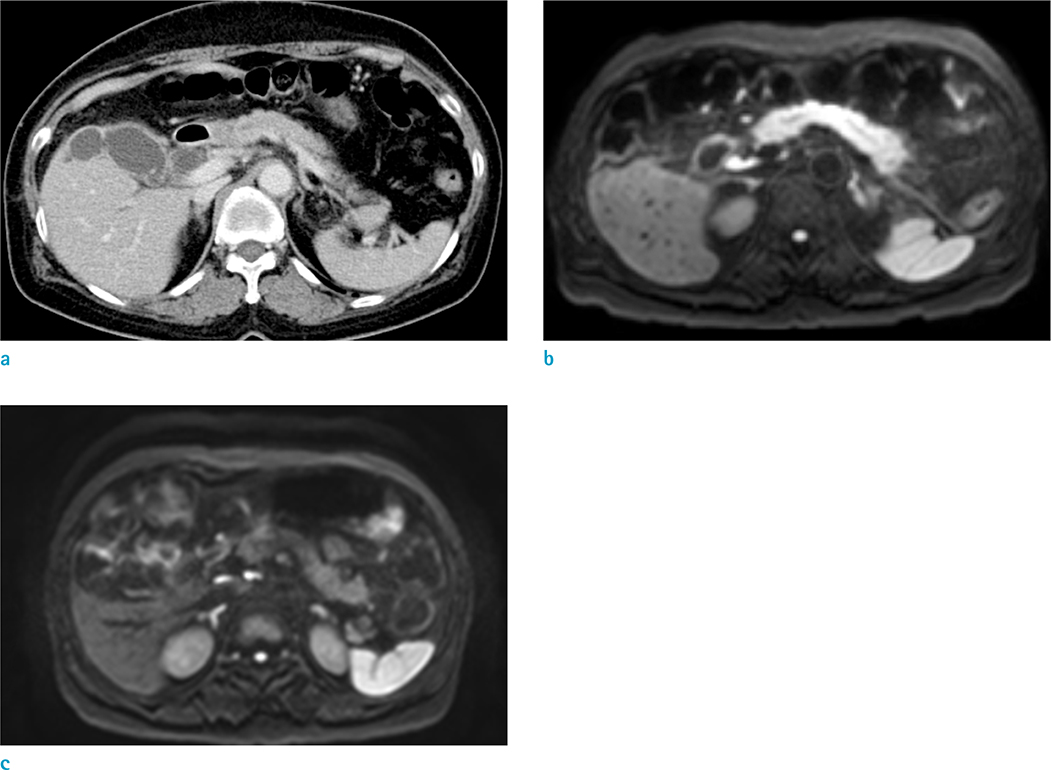
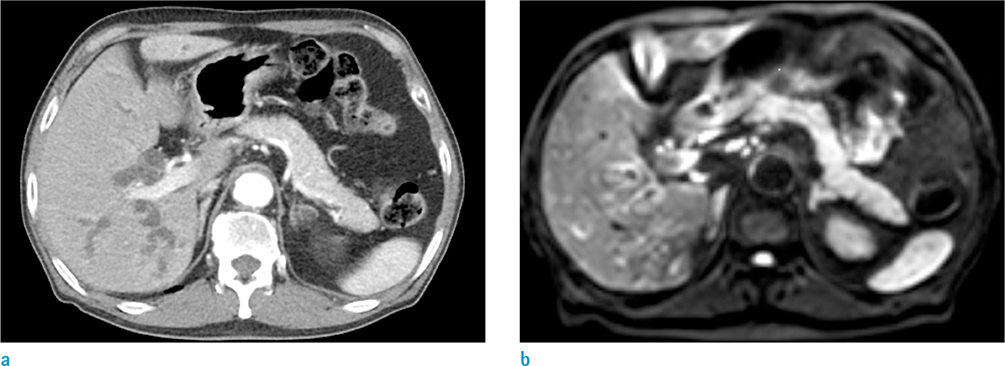
203 patients who underwent abdomen CT and subsequent DWI to do a workup for epigastric pain were analyzed. Two blinded radiologists independently performed an interval reading based on CT image sets first, then based on combined CT and DWI image sets. The diagnostic criterion on DWI was the increased signal intensity in the pancreas to that of the spleen. For quantitative analysis, the third radiologist measured ADC value of the pancreas in each patient.
RESULTS
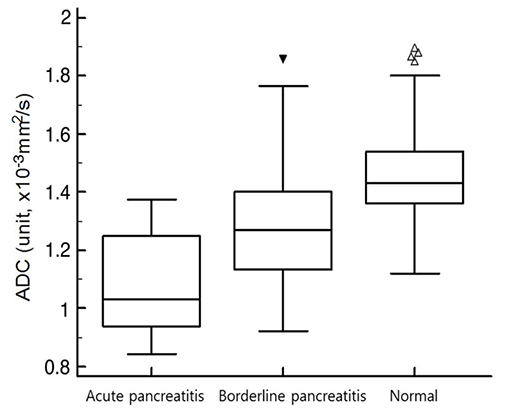
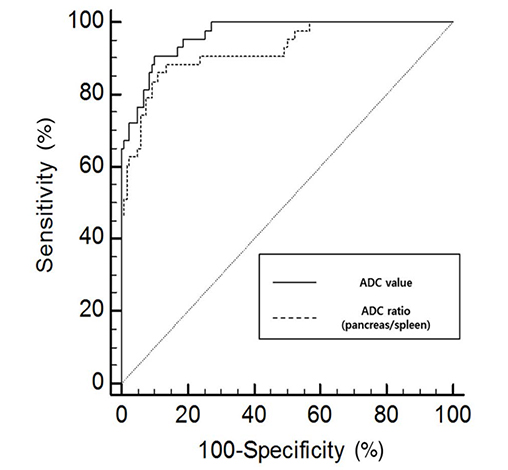
For AP (n = 43), the sensitivity for detecting pancreatic abnormality increased, from 42% to 70% for reader 1 (P < 0.05) and from 44% to 72% for reader 2 (P < 0.05). For borderline pancreatitis (n = 42), the sensitivity also increased, from 10% to 26% for reader 1 (P < 0.05) and from 7% to 29% for reader 2 (P < 0.05). The mean ADC values (unit, × 10⻳ mm²/s) were significantly different among the three groups (for AP, 1.09 ± 0.16; for borderline pancreatitis, 1.28 ± 0.2; for control, 1.46 ± 0.15, P < 0.05).
CONCLUSIONS
Sensitivity for detecting pancreatic abnormality increased significantly after adding DWI to CT in patients with clinically suspected AP.
Figure
Reference
-
1. Yadav D, Lowenfels AB. The epidemiology of pancreatitis and pancreatic cancer. Gastroenterology. 2013; 144:1252–1261.2. Kim CD. Current status of acute pancreatitis in Korea. Korean J Gastroenterol. 2003; 42:1–11.3. Arvanitakis M, Koustiani G, Gantzarou A, et al. Staging of severity and prognosis of acute pancreatitis by computed tomography and magnetic resonance imaging-a comparative study. Dig Liver Dis. 2007; 39:473–482.4. Lankisch PG, Assmus C, Lehnick D, Maisonneuve P, Lowenfels AB. Acute pancreatitis: does gender matter? Dig Dis Sci. 2001; 46:2470–2474.5. de Beaux AC, Palmer KR, Carter DC. Factors influencing morbidity and mortality in acute pancreatitis; an analysis of 279 cases. Gut. 1995; 37:121–126.6. Banks PA, Bollen TL, Dervenis C, et al. Classification of acute pancreatitis--2012: revision of the Atlanta classification and definitions by international consensus. Gut. 2013; 62:102–111.7. Hill MC, Barkin J, Isikoff MB, Silverstein W, Kalser M. Acute pancreatitis: clinical vs. CT findings. AJR Am J Roentgenol. 1982; 139:263–269.8. Balthazar EJ, Ranson JH, Naidich DP, Megibow AJ, Caccavale R, Cooper MM. Acute pancreatitis: prognostic value of CT. Radiology. 1985; 156:767–772.9. Balthazar EJ, Freeny PC, vanSonnenberg E. Imaging and intervention in acute pancreatitis. Radiology. 1994; 193:297–306.10. Viremouneix L, Monneuse O, Gautier G, et al. Prospective evaluation of nonenhanced MR imaging in acute pancreatitis. J Magn Reson Imaging. 2007; 26:331–338.11. Lecesne R, Taourel P, Bret PM, Atri M, Reinhold C. Acute pancreatitis: interobserver agreement and correlation of CT and MR cholangiopancreatography with outcome. Radiology. 1999; 211:727–735.12. Koh DM, Collins DJ. Diffusion-weighted MRI in the body: applications and challenges in oncology. AJR Am J Roentgenol. 2007; 188:1622–1635.13. Kamisawa T, Takuma K, Anjiki H, et al. Differentiation of autoimmune pancreatitis from pancreatic cancer by diffusion-weighted MRI. Am J Gastroenterol. 2010; 105:1870–1875.14. Yencilek E, Telli S, Tekesin K, et al. The efficacy of diffusion weighted imaging for detection of acute pancreatitis and comparison of subgroups according to Balthazar classification. Turk J Gastroenterol. 2014; 25:553–557.15. Hocaoglu E, Aksoy S, Akarsu C, Kones O, Inci E, Alis H. Evaluation of diffusion-weighted MR imaging in the diagnosis of mild acute pancreatitis. Clin Imaging. 2015; 39:463–467.16. Shinya S, Sasaki T, Nakagawa Y, Guiquing Z, Yamamoto F, Yamashita Y. Acute pancreatitis successfully diagnosed by diffusion-weighted imaging: a case report. World J Gastroenterol. 2008; 14:5478–5480.17. Thomas S, Kayhan A, Lakadamyali H, Oto A. Diffusion MRI of acute pancreatitis and comparison with normal individuals using ADC values. Emerg Radiol. 2012; 19:5–9.18. Yoshikawa T, Kawamitsu H, Mitchell DG, et al. ADC measurement of abdominal organs and lesions using parallel imaging technique. AJR Am J Roentgenol. 2006; 187:1521–1530.19. Shinya S, Sasaki T, Nakagawa Y, Guiquing Z, Yamamoto F, Yamashita Y. The efficacy of diffusion-weighted imaging for the detection and evaluation of acute pancreatitis. Hepatogastroenterology. 2009; 56:1407–1410.20. Kilickesmez O, Yirik G, Bayramoglu S, Cimilli T, Aydin S. Non-breath-hold high b-value diffusion-weighted MRI with parallel imaging technique: apparent diffusion coefficient determination in normal abdominal organs. Diagn Interv Radiol. 2008; 14:83–87.21. Ma J, Xia L, Ma X, Zhu L, Zhang G, Yang Y. Diagnostic value of diffusion weighted imaging in acute pancreatitis. Modern Diagnosis & Treatment. 2011; 4:193–196.22. Lee NK, Kim S, Kim GH, et al. Diffusion-weighted imaging of biliopancreatic disorders: correlation with conventional magnetic resonance imaging. World J Gastroenterol. 2012; 18:4102–4117.23. Oto A, Zhu F, Kulkarni K, Karczmar GS, Turner JR, Rubin D. Evaluation of diffusion-weighted MR imaging for detection of bowel inflammation in patients with Crohn's disease. Acad Radiol. 2009; 16:597–603.24. Ream JM, Dillman JR, Adler J, et al. MRI diffusion-weighted imaging (DWI) in pediatric small bowel Crohn disease: correlation with MRI findings of active bowel wall inflammation. Pediatr Radiol. 2013; 43:1077–1085.25. Tsuchiya K, Katase S, Yoshino A, Hachiya J. Diffusion-weighted MR imaging of encephalitis. AJR Am J Roentgenol. 1999; 173:1097–1099.26. Yadav D, Lowenfels AB. Trends in the epidemiology of the first attack of acute pancreatitis: a systematic review. Pancreas. 2006; 33:323–330.27. Bortoff GA, Chen MY, Ott DJ, Wolfman NT, Routh WD. Gallbladder stones: imaging and intervention. Radiographics. 2000; 20:751–766.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Value of PROPELLER Diffusion-Weighted Image in the Detection of Cholesteatoma
- Reversal of a Large Ischemic Lesion with Low Apparent Diffusion Coefficient Value by Rapid Spontaneous Recanalization
- A Case of Acute Recurrent Pancreatitis Caused by Proximal Migration of a Prophylactic Pancreatic Stent
- Langerhans Cell Histiocytosis with Pancreatic Involvement: Imaging Findings Including Diffusion-Weighted Imaging
- Diffusion Weighted Magnetic Resonance Imaging in a Patient with Acute Wernicke Encephalopathy