Korean J Physiol Pharmacol.
2016 Nov;20(6):565-571. 10.4196/kjpp.2016.20.6.565.
The effects of paeoniflorin injection on soluble triggering receptor expressed on myeloid-1 (sTREM-1) levels in severe septic rats
- Affiliations
-
- 1ICU, Tianjin TEDA Hospital, Tianjin 300457, China. xrliu1234@163.com
- KMID: 2364260
- DOI: http://doi.org/10.4196/kjpp.2016.20.6.565
Abstract
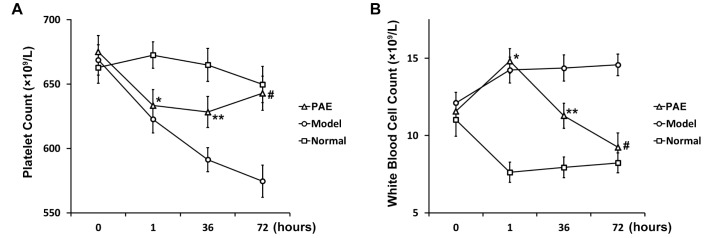
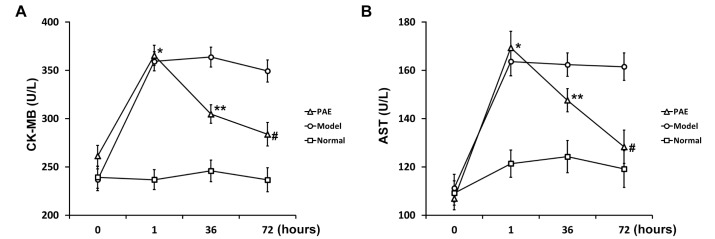
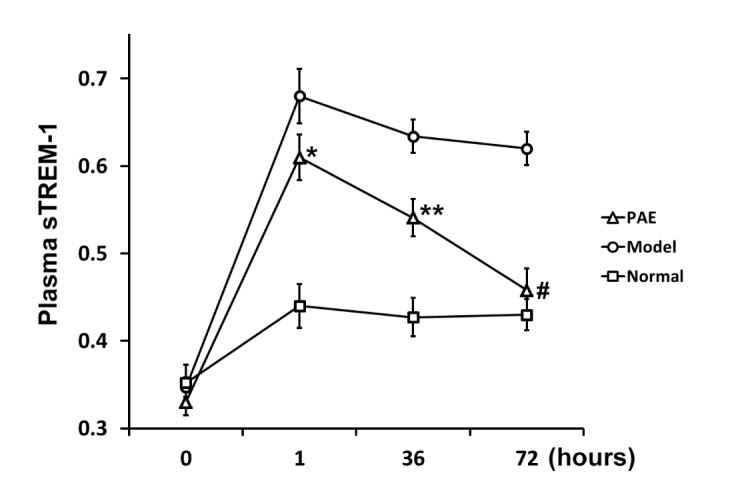
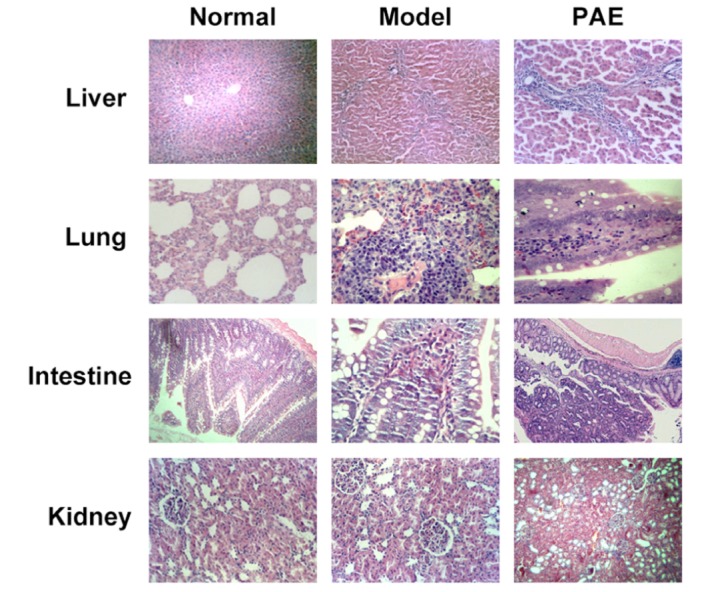
- Paeoniflorin (PAE) is the most abundant compound in Xuebijing injection widely used to treat sepsis. We aimed to investigate effect of PAE on expression of soluble triggering receptor expressed on myeloid cells-1 (sTREM-1) in a rat model of sepsis. Wistar rats were divided into Normal, Model, and PAE groups (n=20 each). Endotoxin was administrated at 5 mg/ml/kg in Model and PAE rats to establish rat sepsis model. 1 h after endotoxin administration, PAE was administrated at 4 ml/kg in PAE group once per day for 3 days. Routine blood tests and biochemical indexes were assessed, including aspartate aminotransferase (AST) and creatine kinase-MB (CK-MB). The plasma sTREM-1 level was measured using quantitative ELISA. At the end of experiment, the small intestine, liver, kidney and lung were subjected to pathological examinations. A rat model of sepsis-induced multiple organ dysfunction syndrome (MODS) was established successfully with endotoxin administration (5 mg/ml/kg), evidenced by histo-pathological examinations, routine blood tests and biochemical indexes: platelet count decreased and white blood cell count increased (p<0.05), CK-MB and AST increased (p<0.05). PAE treatment significantly reduced the plasma levels of AST, CK-MB, and sTREM-1, compared to Model group (p<0.05). Meanwhile, sepsis-induced damages in the liver, lung, stomach and intestinal mucosa were also markedly ameliorated by PAE treatment. PAE demonstrated a significantly protective effect in a rat model of sepsis by decreasing plasma sTREM-1 level, reducing inflammation, preventing MODS and protecting organ functions.
Keyword
MeSH Terms
Figure
Reference
-
1. Angus DC, van der Poll T. Severe sepsis and septic shock. N Engl J Med. 2013; 369:840–851. PMID: 23984731.
Article2. Angus DC, Linde-Zwirble WT, Lidicker J, Clermont G, Carcillo J, Pinsky MR. Epidemiology of severe sepsis in the United States: analysis of incidence, outcome, and associated costs of care. Crit Care Med. 2001; 29:1303–1310. PMID: 11445675.
Article3. Prucha M, Bellingan G, Zazula R. Sepsis biomarkers. Clin Chim Acta. 2015; 440:97–103. PMID: 25447700.
Article4. Gibot S, Cravoisy A, Levy B, Bene MC, Faure G, Bollaert PE. Soluble triggering receptor expressed on myeloid cells and the diagnosis of pneumonia. N Engl J Med. 2004; 350:451–458. PMID: 14749453.
Article5. Gibot S. Clinical review: role of triggering receptor expressed on myeloid cells-1 during sepsis. Crit Care. 2005; 9:485–489. PMID: 16277737.6. Knapp S, Gibot S, de Vos A, Versteeg HH, Colonna M, van der Poll T. Cutting edge: expression patterns of surface and soluble triggering receptor expressed on myeloid cells-1 in human endotoxemia. J Immunol. 2004; 173:7131–7134. PMID: 15585833.
Article7. Lemarié J, Barraud D, Gibot S. Host response biomarkers in sepsis: overview on sTREM-1 detection. Methods Mol Biol. 2015; 1237:225–239. PMID: 25319790.
Article8. Dai X, Zeng Z, Fu C, Zhang S, Cai Y, Chen Z. Diagnostic value of neutrophil gelatinase-associated lipocalin, cystatin C, and soluble triggering receptor expressed on myeloid cells-1 in critically ill patients with sepsis-associated acute kidney injury. Crit Care. 2015; 19:223. PMID: 25944130.
Article9. Ravetti CG, Moura AD, Vieira ÉL, Pedroso ÊR, Teixeira AL. sTREM-1 predicts intensive care unit and 28-day mortality in cancer patients with severe sepsis and septic shock. J Crit Care. 2015; 30:440.e7–440.e13.
Article10. Arízaga-Ballesteros V, Alcorta-García MR, Lázaro-Martínez LC, Amézquita-Gómez JM, Alanís-Cajero JM, Villela L, Castorena-Torres F, Lara-Díaz VJ. Can sTREM-1 predict septic shock & death in late-onset neonatal sepsis? A pilot study. Int J Infect Dis. 2015; 30:27–32. PMID: 25461656.11. Hou SY, Feng XH, Lin CL, Tan YF. Efficacy of Xuebijing for coagulopathy in patients with sepsis. Saudi Med J. 2015; 36:164–169. PMID: 25719579.
Article12. Jiang M, Zhou M, Han Y, Xing L, Zhao H, Dong L, Bai G, Luo G. Identification of NF-κB Inhibitors in Xuebijing injection for sepsis treatment based on bioactivity-integrated UPLC-Q/TOF. J Ethnopharmacol. 2013; 147:426–433. PMID: 23524166.
Article13. He XD, Wang Y, Wu Q, Wang HX, Chen ZD, Zheng RS, Wang ZS, Wang JB, Yang Y. Xuebijing protects rats from sepsis challenged with acinetobacter baumannii by promoting annexin A1 expression and inhibiting proinflammatory cytokines secretion. Evid Based Complement Alternat Med. 2013; 2013:804940. PMID: 24369483.14. Gao YL, Chai YF, Yao YM. Advancement in the research of mechanism of immune dysfunction in sepsis and the regulatory effects of Xuebijing injection. Zhonghua Shao Shang Za Zhi. 2013; 29:162–165. PMID: 23985206.15. Shao M, Liu B, Wang JQ, Tao XG, Zhou SS, Jin K, Zhang CP. Effect of Xuebijing injection on T helper 17 and CD4+ CD25+ regulatory T cells in patients with sepsis. Zhongguo Wei Zhong Bing Ji Jiu Yi Xue. 2011; 23:430–434. PMID: 21787474.16. Zhu XQ, Wang L, Liu QQ, Yao YM. Protective effects of Xuebijing injection on kidney in rats with sepsis. Zhongguo Wei Zhong Bing Ji Jiu Yi Xue. 2006; 18:680–683. PMID: 17092422.17. Zhang SW, Sun CD, Wen Y, Yin CH. Effect of treatment with Xuebijing injection on serum inflammatory mediators and Th1/2 of spleen in rats with sepsis. Zhongguo Wei Zhong Bing Ji Jiu Yi Xue. 2006; 18:673–676. PMID: 17092419.18. Guo SS, Gao YJ, Tian XC, Jin YH, Liu FZ, Cui XL. Effect of xuebijing oral effervescent tablet on endotoxin induced fever and disseminated intravascular coagulation rabbit model. Yao Xue Xue Bao. 2013; 48:1241–1246. PMID: 24187830.19. Yao XQ, Zhang YH, Sun CH. Protective effects of Xuebijing effervescent tablet on vital organs in rats with toxic injury induced by endotoxin. Zhongguo Wei Zhong Bing Ji Jiu Yi Xue. 2012; 24:357–359. PMID: 23019734.20. Liu YK, He JB, Chen HE, Chen D, Miao YF, Ying L, You X, Wang WT. Effect of Xuebijing Injection on TLR4-NF-κB-TNF-α pathway of rats' myocardial anoxia/reoxygenation. Zhongguo Zhong Xi Yi Jie He Za Zhi. 2014; 34:1463–1468. PMID: 25632747.21. He J, Tan Z, Zhang M, Guo L. Effect of Xuebijing injection on hemodynamics and endothelial function in patients with severe sepsis: a prospective study. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue. 2015; 27:127–132. PMID: 25665612.22. Ji L, Huang H, Jiang M, Bai G, Luo G. Simultaneous HPLC determination of 11 essential compounds in Xuebijing injection. Zhongguo Zhong Yao Za Zhi. 2010; 35:2395–2398. PMID: 21141485.23. Yin D, Liu YY, Wang TX, Hu ZZ, Qu WM, Chen JF, Cheng NN, Huang ZL. Paeoniflorin exerts analgesic and hypnotic effects via adenosine A1 receptors in a mouse neuropathic pain model. Psychopharmacology (Berl). 2016; 233:281–293. [Epub ahead of print]. PMID: 26514553.
Article24. Liu C, Cheng Z, Wang Y, Dai X, Zhang J, Xue D. Paeoniflorin exerts a nephroprotective effect on concanavalin A-induced damage through inhibition of macrophage infiltration. Diagn Pathol. 2015; 10:120. PMID: 26204936.
Article25. Zhang HR, Peng JH, Cheng XB, Shi BZ, Zhang MY, Xu RX. Paeoniflorin atttenuates amyloidogenesis and the inflammatory responses in a transgenic mouse model of alzheimer's disease. Neurochem Res. 2015; 40:1583–1592. PMID: 26068144.
Article26. Li P, Li Z. Neuroprotective effect of paeoniflorin on H2O2-induced apoptosis in PC12 cells by modulation of reactive oxygen species and the inflammatory response. Exp Ther Med. 2015; 9:1768–1772. PMID: 26136891.
Article27. Chen Z, Ma X, Zhu Y, Zhao Y, Wang J, Li R, Chen C, Wei S, Jiao W, Zhang Y, Li J, Wang L, Wang R, Liu H, Shen H, Xiao X. Paeoniflorin ameliorates ANIT-induced cholestasis by activating Nrf2 through an PI3K/Akt-dependent pathway in rats. Phytother Res. 2015; 29:1768–1775. PMID: 26269092.
Article28. Zhang LG, Wang LJ, Shen QQ, Wang HF, Zhang Y, Shi CG, Zhang SC, Zhang MY. Paeoniflorin improves regional cerebral blood flow and suppresses inflammatory factors in the hippocampus of rats with vascular dementia. Chin J Integr Med. 2015; [Epub ahead of print].
Article29. HU S, Sheng ZY, Zhou BT. Studies on the animal models of two-phase late onset of multiple organ dystrophy syndrome. Chin J Traumatol. 1996; 12:102–106.30. Determann RM, Millo JL, Gibot S, Korevaar JC, Vroom MB, van der Poll T, Garrard CS, Schultz MJ. Serial changes in soluble triggering receptor expressed on myeloid cells in the lung during development of ventilator-associated pneumonia. Intensive Care Med. 2005; 31:1495–1500. PMID: 16195904.
Article31. Yao YM, Luan YY, Zhang QH, Sheng ZY. Pathophysiological aspects of sepsis: an overview. Methods Mol Biol. 2015; 1237:5–15. PMID: 25319775.
Article32. Barati M, Bashar FR, Shahrami R, Zadeh MH, Taher MT, Nojomi M. Soluble triggering receptor expressed on myeloid cells 1 and the diagnosis of sepsis. J Crit Care. 2010; 25:362.e1–362.e6.
Article33. Su L, Han B, Liu C, Liang L, Jiang Z, Deng J, Yan P, Jia Y, Feng D, Xie L. Value of soluble TREM-1, procalcitonin, and C-reactive protein serum levels as biomarkers for detecting bacteremia among sepsis patients with new fever in intensive care units: a prospective cohort study. BMC Infect Dis. 2012; 12:157. PMID: 22809118.
Article34. Wang HX, Chen B. Diagnostic role of soluble triggering receptor expressed on myeloid cell-1 in patients with sepsis. World J Emerg Med. 2011; 2:190–194. PMID: 25215008.
Article35. Cui Y, Zhang YX, Li CX, Li Y, Shen HL. The effects of nitrogen monoxide/inducible nitric oxide synthase on renal injury in septic rats and the renal protective effects of Xuebijing injection. Zhongguo Wei Zhong Bing Ji Jiu Yi Xue. 2009; 21:497–498. PMID: 19695177.36. Shen J, Lin XJ, Cui BK, Chi PD, Zeng QY, Zhao QY. The protective effect of Xuebijing injection pretreatment on hepatic ischemia reperfusion injury and coagulopathy after excision of liver cancer. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue. 2013; 25:743–748. PMID: 24620386.37. Ma JF, Xuan LZ, Wu W, Zhu DM. Effect of Xuebijing injection on rabbits ischemia/reperfusion injury induced by femoral arterial disease. Zhongguo Wei Zhong Bing Ji Jiu Yi Xue. 2012; 24:233–236. PMID: 22464578.38. Lu Y, Sheng HL, Wang LH. Effects of Xuebijing injection on the expression of sTREM-1 in severe spetic patients. Chin J TCM WM Crit Care. 2013; 20:337–340.39. Liu MW, Wang YH, Qian CY, Li H. Xuebijing exerts protective effects on lung permeability leakage and lung injury by upregulating Toll-interacting protein expression in rats with sepsis. Int J Mol Med. 2014; 34:1492–1504. PMID: 25269519.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Diagnostic Utility of Pleural Fluid Soluble Triggering Receptor Expressed on Myeloid Cells 1 Protein in Patients with Exudative Pleural Effusion
- Correlation between Soluble Triggering Receptor Expressed on Myeloid Cells-1 and Endoscopic Activity in Intestinal Behcet's Disease
- Soluble Triggering Receptor Expressed on Myeloid cells-1: Role in the Diagnosis of Pleural Effusions
- Prognostic Utility of the Soluble Triggering Receptor Expressed on Myeloid Cells-1 in Patients with Acute Respiratory Distress Syndrome
- A New Triggering Receptor Expressed on Myeloid Cells (TREM) Family Member, TLT-6, is Involved in Activation and Proliferation of Macrophages