J Vet Sci.
2016 Mar;17(1):63-70. 10.4142/jvs.2016.17.1.63.
Gap junction blockage promotes cadmium-induced apoptosis in BRL 3A derived from Buffalo rat liver cells
- Affiliations
-
- 1College of Veterinary Medicine, Yangzhou University, Yangzhou 225009, China. liuzongping@yzu.edu.cn
- 2Jiangsu Co-innovation Center for Prevention and Control of Important Animal Infectious Diseases and Zoonoses, Yangzhou 225009, China.
- 3Department of Life Science, Zaozhuang College, Zaozhuang 277160, China.
- KMID: 2363336
- DOI: http://doi.org/10.4142/jvs.2016.17.1.63
Abstract
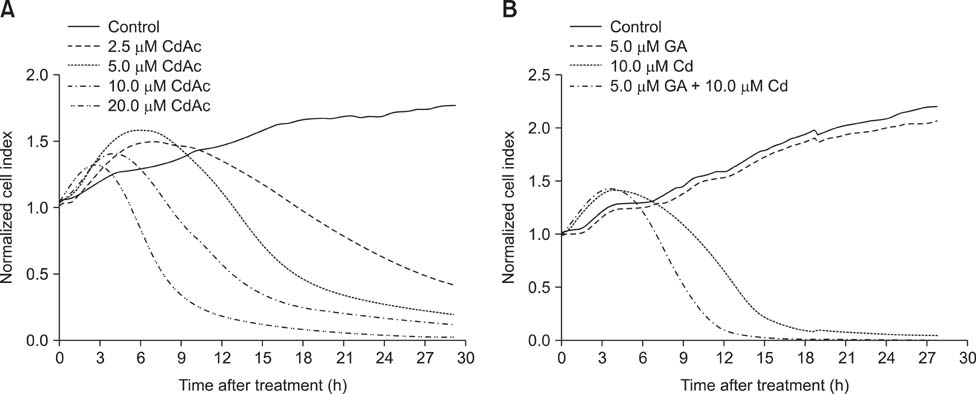
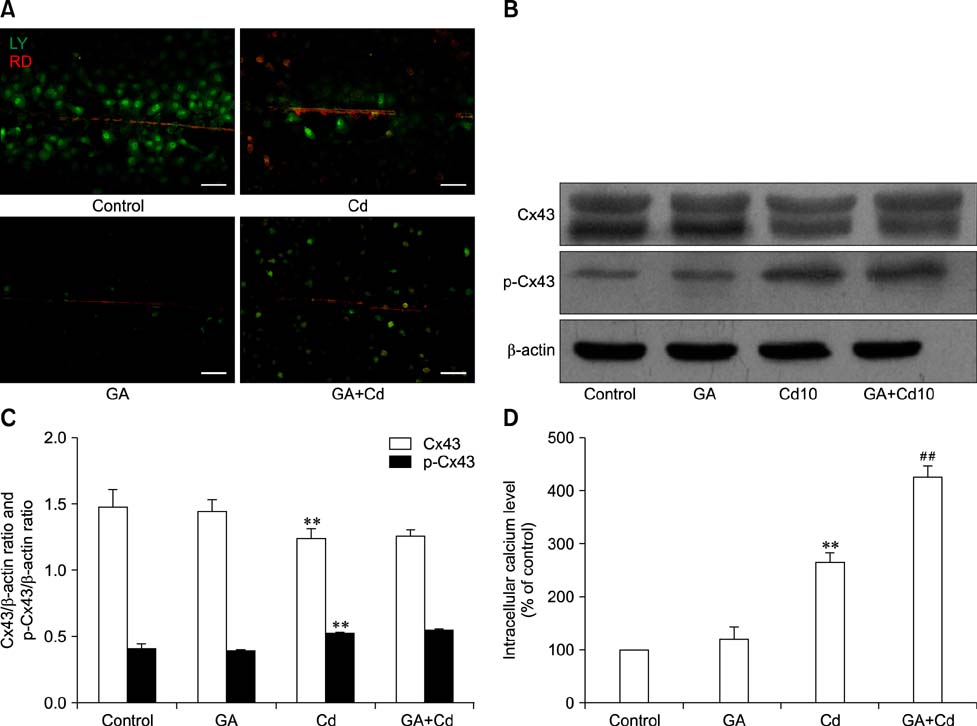
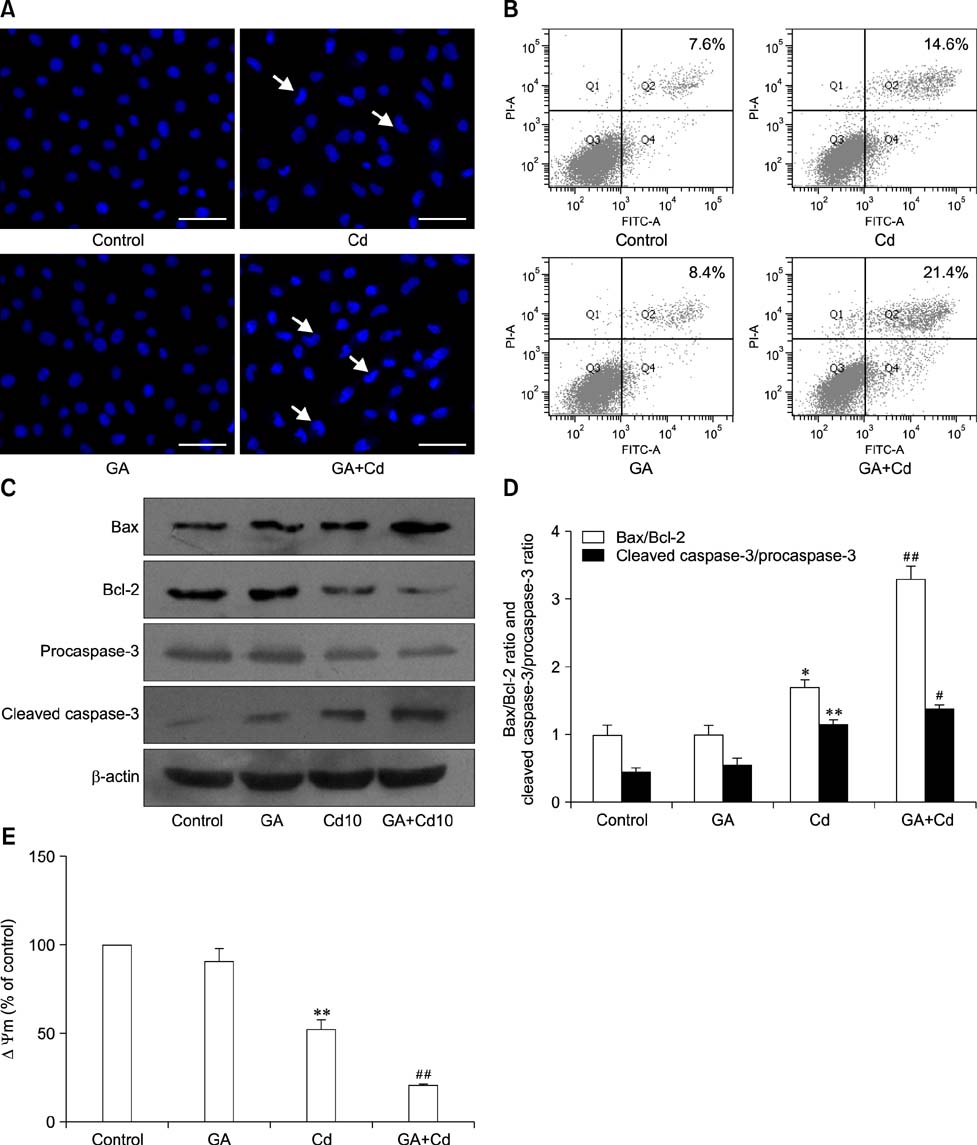
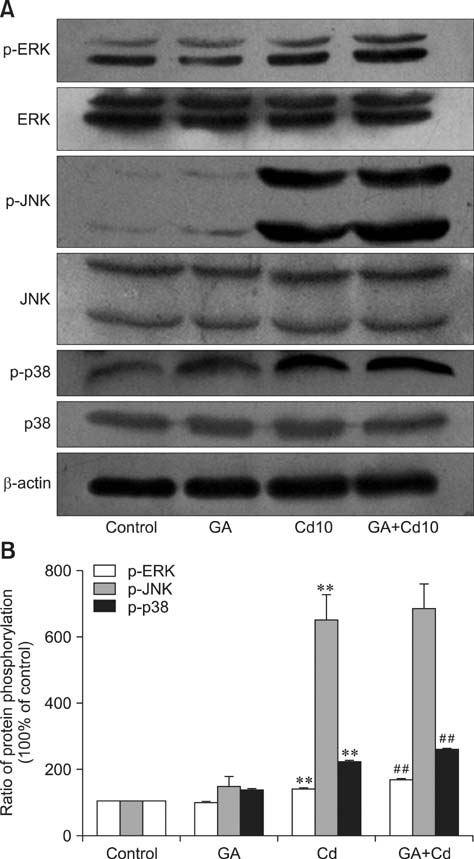
- Gap junctions mediate direct communication between cells; however, toxicological cascade triggered by nonessential metals can abrogate cellular signaling mediated by gap junctions. Although cadmium (Cd) is known to induce apoptosis in organs and tissues, the mechanisms that underlie gap junction activity in Cd-induced apoptosis in BRL 3A rat liver cells has yet to be established. In this study, we showed that Cd treatment decreased the cell index (a measure of cellular electrical impedance) in BRL 3A cells. Mechanistically, we found that Cd exposure decreased expression of connexin 43 (Cx43), increased expression of p-Cx43 and elevated intracellular free Ca2+ concentration, corresponding to a decrease in gap junctional intercellular communication. Gap junction blockage pretreatment with 18β-glycyrrhizic acid (GA) promoted Cd-induced apoptosis, involving changes in expression of Bax, Bcl-2, caspase-3 and the mitochondrial transmembrane electrical potential (Δψm). Additionally, GA was found to enhance ERK and p38 activation during Cd-induced activation of mitogen-activated protein kinases, but had no significant effect on JNK activation. Our results indicated the apoptosis-related proteins and the ERK and p38 signaling pathways may participate in gap junction blockage promoting Cd-induced apoptosis in BRL 3A cells.
Keyword
MeSH Terms
-
Animals
Apoptosis/*drug effects
Cadmium/*toxicity
Calcium/metabolism
Cell Communication/drug effects
Connexin 43/genetics
Enzyme Activation/drug effects
Gap Junctions/*drug effects
Gene Expression Regulation/drug effects
Hepatocytes/cytology/*drug effects
Rats
Signal Transduction/drug effects
Cadmium
Calcium
Connexin 43
Figure
Reference
-
1. Bertin G, Averbeck D. Cadmium: cellular effects, modifications of biomolecules, modulation of DNA repair and genotoxic consequences (a review). Biochimie. 2006; 88:1549–1559.
Article2. Boucherie S, Decaens C, Verbavatz JM, Grosse B, Erard M, Merola F, Cassio D, Combettes L. Cadmium disorganises the scaffolding of gap and tight junction proteins in the hepatic cell line WIF B9. Biol Cell. 2013; 105:561–575.
Article3. Decrock E, Vinken M, Bol M, D'Herde K, Rogiers V, Vandenabeele P, Krysko DV, Bultynck G, Leybaert L. Calcium and connexin-based intercellular communication, a deadly catch? Cell Calcium. 2011; 50:310–321.
Article4. Fang X, Huang T, Zhu Y, Yan Q, Chi Y, Jiang JX, Wang P, Matsue H, Kitamura M, Yao J. Connexin43 hemichannels contribute to cadmium-induced oxidative stress and cell injury. Antioxid Redox Signal. 2011; 14:2427–2439.
Article5. Garrod DR, Merritt AJ, Nie Z. Desmosomal adhesion: structural basis, molecular mechanism and regulation (Review). Mol Membr Biol. 2002; 19:81–94.
Article6. Giepmans BN. Gap junctions and connexin-interacting proteins. Cardiovasc Res. 2004; 62:233–245.
Article7. Harris AL. Emerging issues of connexin channels: biophysics fills the gap. Q Rev Biophys. 2001; 34:325–472.
Article8. Huang RP, Hossain MZ, Huang R, Gano J, Fan Y, Boynton AL. Connexin 43 (cx43) enhances chemotherapy-induced apoptosis in human glioblastoma cells. Int J Cancer. 2001; 92:130–138.
Article9. Jeong SH, Habeebu SSM, Klaassen CD. Cadmium decreases gap junctional intercellular communication in mouse liver. Toxicol Sci. 2000; 57:156–166.
Article10. Joseph P. Mechanisms of cadmium carcinogenesis. Toxicol Appl Pharmacol. 2009; 238:272–279.
Article11. Kameritsch P, Khandoga N, Pohl U, Pogoda K. Gap junctional communication promotes apoptosis in a connexin-type-dependent manner. Cell Death Dis. 2013; 4:e584.
Article12. Kim KN, Ham YM, Moon JY, Kim MJ, Jung YH, Jeon YJ, Lee NH, Kang N, Yang HM, Kim D, Hyun CG. Acanthoic acid induces cell apoptosis through activation of the p38 MAPK pathway in HL-60 human promyelocytic leukaemia. Food Chem. 2012; 135:2112–2117.
Article13. Lemarié A, Lagadic-Gossmann D, Morzadec C, Allain N, Fardel O, Vernhet L. Cadmium induces caspase-independent apoptosis in liver Hep3B cells: role for calcium in signaling oxidative stress-related impairment of mitochondria and relocation of endonuclease G and apoptosis-inducing factor. Free Radic Biol Med. 2004; 36:1517–1531.
Article14. Leung HW, Yang WH, Lai MY, Lin CJ, Lee HZ. Inhibition of 12-lipoxygenase during baicalein-induced human lung nonsmall carcinoma H460 cell apoptosis. Food Chem Toxicol. 2007; 45:403–411.
Article15. Li L, Jiang C, Hao P, Li W, Song C, Song B. Changes of gap junctional cell-cell communication in overactive detrusor in rats. Am J Physiol Cell Physiol. 2007; 293:C1627–C1635.
Article16. Lin D, Takemoto DJ. Protection from ataxia-linked apoptosis by gap junction inhibitors. Biochem Biophys Res Commun. 2007; 362:982–987.
Article17. Lin H, Ogawa K, Imanaga I, Tribulova N. Alterations of connexin 43 in the diabetic rat heart. Adv Cardiol. 2006; 42:243–254.
Article18. Liu W, Zhao H, Wang Y, Jiang C, Xia P, Gu J, Liu X, Bian J, Yuan Y, Liu Z. Calcium-calmodulin signaling elicits mitochondrial dysfunction and the release of cytochrome c during cadmium-induced apoptosis in primary osteoblasts. Toxicol Lett. 2014; 224:1–6.
Article19. Munshi A, Ramesh R. Mitogen-activated protein kinases and their role in radiation response. Genes Cancer. 2013; 4:401–408.
Article20. Nakase T, Fushiki S, Naus CC. Astrocytic gap junctions composed of connexin 43 reduce apoptotic neuronal damage in cerebral ischemia. Stroke. 2003; 34:1987–1993.
Article21. Ngezahayo A, Altmann B, Steffens M, Kolb HA. Gap junction coupling and apoptosis in GFSHR-17 granulosa cells. J Membr Biol. 2005; 204:137–144.
Article22. Nodin C, Nilsson M, Blomstrand F. Gap junction blockage limits intercellular spreading of astrocytic apoptosis induced by metabolic depression. J Neurochem. 2005; 94:1111–1123.
Article23. Nováková K, Babica P, Adamovský O, Bláha L. Modulation of gap-junctional intercellular communication by a series of cyanobacterial samples from nature and laboratory cultures. Toxicon. 2011; 58:76–84.
Article24. Pauly D, Worbs S, Kirchner S, Shatohina O, Dorner MB, Dorner BG. Real-time cytotoxicity assay for rapid and sensitive detection of ricin from complex matrices. PLoS One. 2012; 7:e35360.
Article25. Ryu MJ, Kim AD, Kang KA, Chung HS, Kim HS, Suh IS, Chang WY, Hyun JW. The green algae Ulva fasciata Delile extract induces apoptotic cell death in human colon cancer cells. In Vitro Cell Dev Biol Anim. 2013; 49:74–81.
Article26. Shieh JM, Huang TF, Hung CF, Chou KH, Tsai YJ, Wu WB. Activation of c-Jun N-terminal kinase is essential for mitochondrial membrane potential change and apoptosis induced by doxycycline in melanoma cells. Br J Pharmacol. 2010; 160:1171–1184.
Article27. Subramaniam S, Unsicker K. ERK and cell death: ERK1/2 in neuronal death. FEBS J. 2010; 277:22–29.
Article28. Swiergosz-Kowalewska R. Cadmium distribution and toxicity in tissues of small rodents. Microsc Res Tech. 2001; 55:208–222.
Article29. Theocharis SE, Margeli AP, Giannakou N, Drakopoulos DS, Mykoniatis MG. Cadmium-induced hepatotoxicity in three different rat strains. Toxicol Lett. 1994; 70:39–48.
Article30. Thévenod F, Lee WK. Cadmium and cellular signaling cascades: interactions between cell death and survival pathways. Arch Toxicol. 2013; 87:1743–1786.
Article31. Thévenod F, Lee WK. Toxicology of cadmium and its damage to mammalian organs. Met Ions Life Sci. 2013; 11:415–490.
Article32. Vinken M, Ceelen L, Vanhaecke T, Rogiers V. Inhibition of gap junctional intercellular communication by toxic metals. Chem Res Toxicol. 2010; 23:1862–1867.
Article33. Vinken M, Doktorova T, Decrock E, Leybaert L, Vanhaecke T, Rogiers V. Gap junctional intercellular communication as a target for liver toxicity and carcinogenicity. Crit Rev Biochem Mol Biol. 2009; 44:201–222.
Article34. Wang J, Zhu H, Liu X, Liu Z. Oxidative stress and Ca2+ signals involved on cadmium-induced apoptosis in rat hepatocyte. Biol Trace Elem Res. 2014; 161:180–189.
Article35. Wilson MR, Close TW, Trosko JE. Cell population dynamics (apoptosis, mitosis, and cell-cell communication) during disruption of homeostasis. Exp Cell Res. 2000; 254:257–268.
Article36. Yasui K, Kada K, Hojo M, Lee JK, Kamiya K, Toyama J, Opthof T, Kodama I. Cell-to-cell interaction prevents cell death in cultured neonatal rat ventricular myocytes. Cardiovasc Res. 2000; 48:68–76.
Article37. Yiran Z, Jiang C, Wang J, Yuan Y, Gu J, Bian J, Liu X, Liu Z. Oxidative stress and mitogen-activated protein kinase pathways involved in cadmium-induced BRL 3A cell apoptosis. Oxid Med Cell Longev. 2013; 2013:516051.
Article38. Zou H, Hu D, Han T, Zhao H, Xie J, Liu X, Wang Y, Gu J, Yuan Y, Bian J, Liu Z. Salidroside ameliorates Cd-induced calcium overload and gap junction dysfunction in BRL 3A rat liver cells. Biol Trace Elem Res. 2015; 164:90–98.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Development of Human Embryos in the Buffalo Rat Liver(BRL) Cell Coculture
- L-Theanine-Treated Adipose-Derived Mesenchymal Stem Cells Alleviate the Cytotoxicity Induced by N-Nitrosodiethylamine in Liver
- Induction Of Metallothionein And Toxicity In Acute Cadmium Intoxicated Rat
- Control of Mitochondrial Dynamics by Fas-induced Caspase-8 Activation in Hippocampal Neurons
- Changes of Tight Junction and Epithelial Cells after Treatment of Cadmium