J Vet Sci.
2016 Mar;17(1):13-20. 10.4142/jvs.2016.17.1.13.
Reduction of adult hippocampal neurogenesis is amplified by aluminum exposure in a model of type 2 diabetes
- Affiliations
-
- 1Department of Anatomy and Cell Biology, College of Veterinary Medicine, Seoul National University, Seoul 08826, Korea. ysyoon@snu.ac.kr
- 2BK21 PLUS Program for Creative Veterinary Science Research, and Research Institute for Veterinary Science, Seoul National University, Seoul 08826, Korea.
- 3Department of Anatomy, College of Veterinary Medicine, Kangwon National University, Chuncheon 24341, Korea.
- 4Korea Mouse Phenotyping Center, Seoul National University, Seoul 08826, Korea.
- KMID: 2363330
- DOI: http://doi.org/10.4142/jvs.2016.17.1.13
Abstract
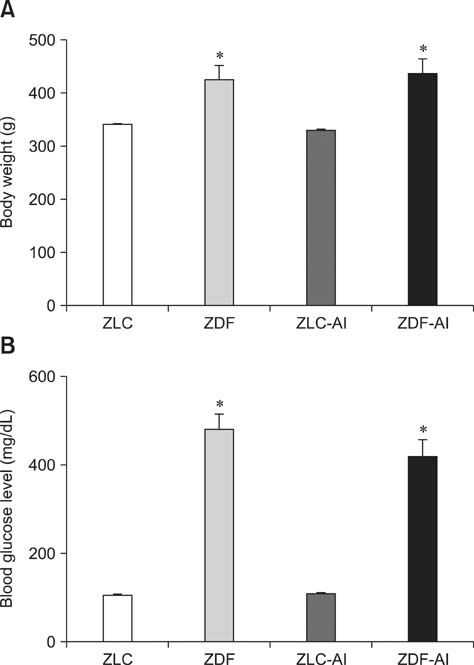
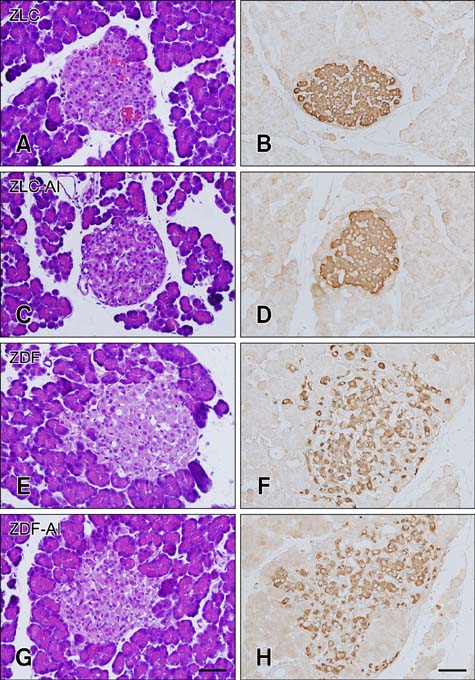
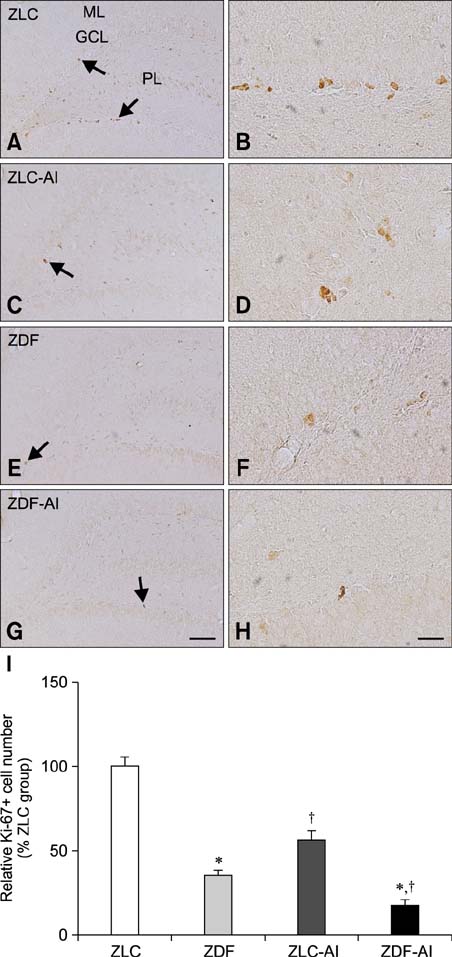
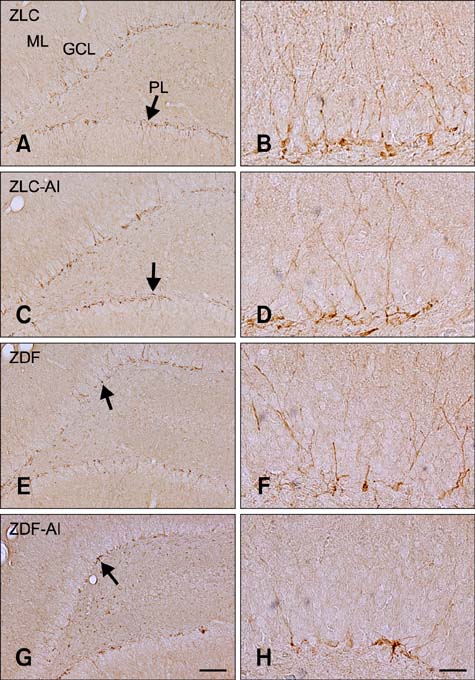
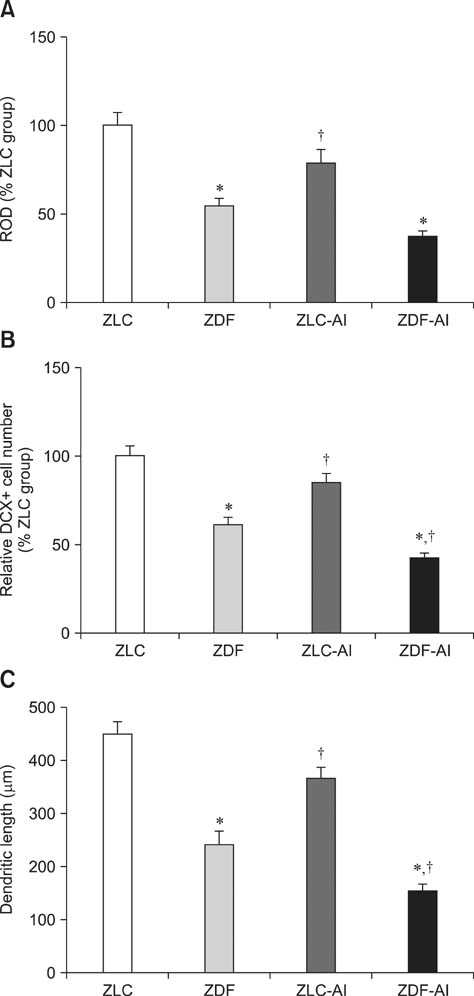
- In this study, we investigated the effects of chronic aluminum (Al) exposure for 10 weeks on cell proliferation and neuroblast differentiation in the hippocampus of type 2 diabetic rats. Six-week-old Zucker diabetic fatty (ZDF) and Zucker lean control (ZLC) rats were selected and randomly divided into Al- and non-Al-groups. Al was administered via drinking water for 10 weeks, after which the animals were sacrificed at 16 weeks of age. ZDF rats in both Al- and non-Al-groups showed increases in body weight and blood glucose levels compared to ZLC rats. Al exposure did not significantly affect body weight, blood glucose levels or pancreatic β-cells and morphology of the pancreas in either ZLC or ZDF rats. However, exposure to Al reduced cell proliferation and neuroblast differentiation in both ZLC and ZDF rats. Exposure to Al resulted in poor development of the dendritic processes of neuroblasts in both ZLC and ZDF rats. Furthermore, onset and continuation of diabetes reduced cell proliferation and neuroblast differentiation, and Al exposure amplified reduction of these parameters. These results suggest that Al exposure via drinking water aggravates the impairment in hippocampal neurogenesis that is typically observed in type 2 diabetic animals.
MeSH Terms
-
Aluminum/*toxicity
Animals
Blood Glucose/analysis
Cell Differentiation/drug effects
Cell Proliferation/drug effects
Diabetes Mellitus, Experimental/pathology
Diabetes Mellitus, Type 2/*pathology
Disease Models, Animal
Hippocampus/*drug effects
Neurogenesis/*drug effects
Random Allocation
Rats, Zucker
Aluminum
Blood Glucose
Figure
Cited by 1 articles
-
Effects of aluminum on the reduction of neural stem cells, proliferating cells, and differentiating neuroblasts in the dentate gyrus of D-galactose-treated mice via increasing oxidative stress
Sung Min Nam, Jong Whi Kim, Dae Young Yoo, Woosuk Kim, Hyo Young Jung, Jung Hoon Choi, In Koo Hwang, Je Kyung Seong, Yeo Sung Yoon
J Vet Sci. 2016;17(2):127-136. doi: 10.4142/jvs.2016.17.2.127.
Reference
-
1. Beauquis J, Saravia F, Coulaud J, Roig P, Dardenne M, Homo-Delarche F, De Nicola A. Prominently decreased hippocampal neurogenesis in a spontaneous model of type 1 diabetes, the nonobese diabetic mouse. Exp Neurol. 2008; 210:359–367.
Article2. Becaria A, Lahiri DK, Bondy SC, Chen D, Hamadeh A, Li H, Taylor R, Campbell A. Aluminum and copper in drinking water enhance inflammatory or oxidative events specifically in the brain. J Neuroimmunol. 2006; 176:16–23.
Article3. Beckman JA, Creager MA, Libby P. Diabetes and atherosclerosis: epidemiology, pathophysiology, and management. JAMA. 2002; 287:2570–2581.4. Biessels GJ, Kamal A, Ramakers GM, Urban IJ, Spruijt BM, Erkelens DW, Gispen WH. Place learning and hippocampal synaptic plasticity in streptozotocin-induced diabetic rats. Diabetes. 1996; 45:1259–1266.
Article5. Corsetti JP, Sparks JD, Peterson RG, Smith RL, Sparks CE. Effect of dietary fat on the development of non-insulin dependent diabetes mellitus in obese Zucker diabetic fatty male and female rats. Atherosclerosis. 2000; 148:231–241.
Article6. Deng W, Aimone JB, Gage FH. New neurons and new memories: how does adult hippocampal neurogenesis affect learning and memory? Nat Rev Neurosci. 2010; 11:339–350.
Article7. DeVoto E, Yokel RA. The biological speciation and toxicokinetics of aluminum. Environ Health Perspect. 1994; 102:940–951.8. Döbrössy MD, Drapeau E, Aurousseau C, Le Moal M, Piazza PV, Abrous DN. Differential effects of learning on neurogenesis: learning increases or decreases the number of newly born cells depending on their birth date. Mol Psychiatry. 2003; 8:974–982.
Article9. Dubois B, Albert ML. Amnestic MCI or prodromal Alzheimer's disease? Lancet Neurol. 2004; 3:246–248.
Article10. El-Rahman SS. Neuropathology of aluminum toxicity in rats (glutamate and GABA impairment). Pharmacol Res. 2003; 47:189–194.
Article11. Etgen GJ, Oldham BA. Profiling of Zucker diabetic fatty rats in their progression to the overt diabetic state. Metabolism. 2000; 49:684–688.
Article12. Flaten TP. Aluminium as a risk factor in Alzheimer's disease, with emphasis on drinking water. Brain Res Bull. 2001; 55:187–196.
Article13. Helkala EL, Laulumaa V, Soininen H, Riekkinen PJ. Recall and recognition memory in patients with Alzheimer's and Parkinson's diseases. Ann Neurol. 1988; 24:214–217.
Article14. Hwang IK, Yi SS, Song W, Won MH, Yoon YS, Seong JK. Effects of age and treadmill exercise in chronic diabetic stages on neuronal differentiation in a rat model of type 2 diabetes. Brain Res. 2010; 1341:63–71.
Article15. Jin K, Peel AL, Mao XO, Xie L, Cottrell BA, Henshall DC, Greenberg DA. Increased hippocampal neurogenesis in Alzheimer's disease. Proc Natl Acad Sci U S A. 2004; 101:343–347.
Article16. Jing Y, Wang Z, Song Y. Quantitative study of aluminuminduced changes in synaptic ultrastructure in rats. Synapse. 2004; 52:292–298.
Article17. Julka D, Vasishta RK, Gill KD. Distribution of aluminum in different brain regions and body organs of rat. Biol Trace Elem Res. 1996; 52:181–192.
Article18. Kakimoto T, Kimata H, Iwasaki S, Fukunari A, Utsumi H. Automated recognition and quantification of pancreatic islets in Zucker diabetic fatty rats treated with exendin-4. J Endocrinol. 2013; 216:13–20.
Article19. Kitamura T, Inokuchi K. Role of adult neurogenesis in hippocampal-cortical memory consolidation. Mol Brain. 2014; 7:13.
Article20. Luitse MJ, Biessels GJ, Rutten GE, Kappelle LJ. Diabetes, hyperglycaemia, and acute ischaemic stroke. Lancet Neurol. 2012; 11:261–271.
Article21. Luo Y, Nie J, Gong QH, Lu YF, Wu Q, Shi JS. Protective effects of icariin against learning and memory deficits induced by aluminium in rats. Clin Exp Pharmacol Physiol. 2007; 34:792–795.
Article22. Nam SM, Kim JW, Yoo DY, Kim W, Jung HY, Hwang IK, Seong JK, Yoon YS. Additive or synergistic effects of aluminum on the reduction of neural stem cells, cell proliferation, and neuroblast differentiation in the dentate gyrus of high-fat diet-fed mice. Biol Trace Elem Res. 2014; 157:51–59.
Article23. Paxinos G, Watson C. The Rat Brain in Stereotaxic Coordinates. 6th ed. Amsterdam: Academic Press;2007.24. Pennanen C, Kivipelto M, Tuomainen S, Hartikainen P, Hänninen T, Laakso MP, Hallikainen M, Vanhanen M, Nissinen A, Helkala EL, Vainio P, Vanninen R, Partanen K, Soininen H. Hippocampus and entorhinal cortex in mild cognitive impairment and early AD. Neurobiol Aging. 2004; 25:303–310.
Article25. Pick A, Clark J, Kubstrup C, Levisetti M, Pugh W, Bonner-Weir S, Polonsky KS. Role of apoptosis in failure of beta-cell mass compensation for insulin resistance and beta-cell defects in the male Zucker diabetic fatty rat. Diabetes. 1998; 47:358–364.
Article26. Plaschke K, Kopitz J, Siegelin M, Schliebs R, Salkovic-Petrisic M, Riederer P, Hoyer S. Insulin-resistant brain state after intracerebroventricular streptozotocin injection exacerbates Alzheimer-like changes in Tg2576 AβPP-overexpressing mice. J Alzheimers Dis. 2010; 19:691–704.
Article27. Praticò D, Uryu K, Sung S, Tang S, Trojanowski JQ, Lee VM. Aluminum modulates brain amyloidosis through oxidative stress in APP transgenic mice. FASEB J. 2002; 16:1138–1140.
Article28. Ribes D, Colomina MT, Vicens P, Domingo JL. Impaired spatial learning and unaltered neurogenesis in a transgenic model of Alzheimer's disease after oral aluminum exposure. Curr Alzheimer Res. 2010; 7:401–408.
Article29. Rondeau V, Jacqmin-Gadda H, Commenges D, Helmer C, Dartigues JF. Aluminum and silica in drinking water and the risk of Alzheimer's disease or cognitive decline: findings from 15-year follow-up of the PAQUID cohort. Am J Epidemiol. 2009; 169:489–496.
Article30. Sánchez-Iglesias S, Soto-Otero R, Iglesias-González J, Barciela-Alonso MC, Bermejo-Barrera P, Méndez-Alvarez E. Analysis of brain regional distribution of aluminium in rats via oral and intraperitoneal administration. J Trace Elem Med Biol. 2007; 21:Suppl 1. 31–34.
Article31. Stranahan AM, Arumugam TV, Cutler RG, Lee K, Egan JM, Mattson MP. Diabetes impairs hippocampal function through glucocorticoid-mediated effects on new and mature neurons. Nat Neurosci. 2008; 11:309–317.
Article32. Van Alstyne R, McDowell LR, Davis PA, Wilkinson NS, O'Connor GA. Effects of an aluminum-water treatment residual on performance and mineral status of feeder lambs. Small Rumin Res. 2007; 73:77–86.
Article33. Vukovic J, Borlikova GG, Ruitenberg MJ, Robinson GJ, Sullivan RKP, Walker TL, Bartlett PF. Immature doublecortin-positive hippocampal neurons are important for learning but not for remembering. J Neurosci. 2013; 33:6603–6613.
Article34. Walton JR, Wang MX. APP expression, distribution and accumulation are altered by aluminum in a rodent model for Alzheimer's disease. J Inorg Biochem. 2009; 103:1548–1554.
Article35. Wang M, Chen JT, Ruan DY, Xu YZ. Vasopressin reverses aluminum-induced impairment of synaptic plasticity in the rat dentate gyrus in vivo. Brain Res. 2001; 899:193–200.
Article36. World Health Organization. Aluminium. Guidelines for Drinking-water Quality. Addendum to Vol. 2. Health criteria and other supporting information. 2nd ed. Geneva: World Health Organization;1998.37. Xu W, Caracciolo B, Wang HX, Winblad B, Bäckman L, Qiu C, Fratiglioni L. Accelerated progression from mild cognitive impairment to dementia in people with diabetes. Diabetes. 2010; 59:2928–2935.
Article38. Yau SY, Lau BW, Tong JB, Wong R, Ching YP, Qiu G, Tang SW, Lee TM, So KF. Hippocampal neurogenesis and dendritic plasticity support running-improved spatial learning and depression-like behaviour in stressed rats. PLoS One. 2011; 6:e24263.
Article39. Yokel RA, McNamara PJ. Influence of renal impairment, chemical form, and serum protein binding on intravenous and oral aluminum kinetics in the rabbit. Toxicol Appl Pharmacol. 1988; 95:32–43.
Article40. Yumoto S, Kakimi S, Ohsaki A, Ishikawa A. Demonstration of aluminum in amyloid fibers in the cores of senile plaques in the brains of patients with Alzheimer's disease. J Inorg Biochem. 2009; 103:1579–1584.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Repeated restraint stress promotes hippocampal neuronal cell ciliogenesis and proliferation in mice
- Effects of aluminum on the reduction of neural stem cells, proliferating cells, and differentiating neuroblasts in the dentate gyrus of D-galactose-treated mice via increasing oxidative stress
- Neurogenesis and Psychiatry: Focusing on Mood Disorders
- Normal Adult Hippocampal Neurogenesis in SRG3-overexpressing Transgenic Mice
- Odor Enrichment Increases Hippocampal Neuron Numbers in Mouse