J Korean Med Sci.
2016 Feb;31(2):222-230. 10.3346/jkms.2016.31.2.222.
Improving Effect of the Acute Administration of Dietary Fiber-Enriched Cereals on Blood Glucose Levels and Gut Hormone Secretion
- Affiliations
-
- 1Department of Internal Medicine, Seoul National University College of Medicine, Seoul, Korea. ymchomd@snu.ac.kr
- KMID: 2360046
- DOI: http://doi.org/10.3346/jkms.2016.31.2.222
Abstract
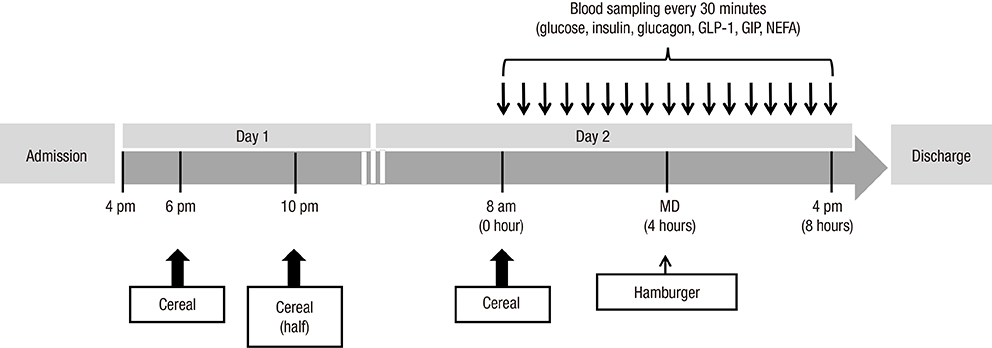
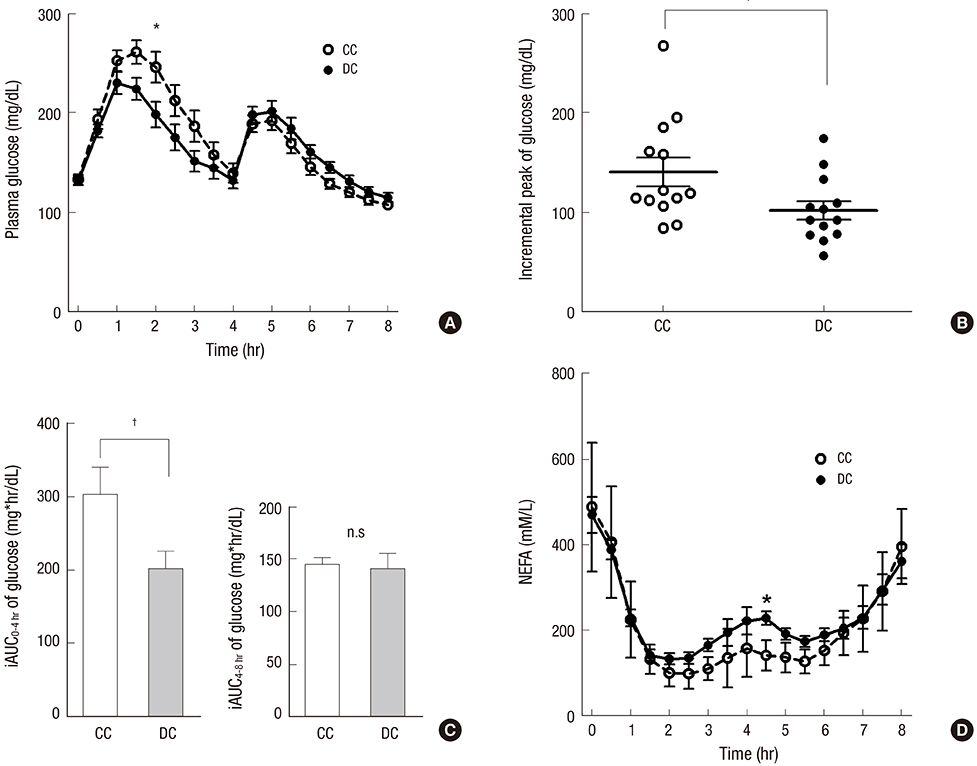
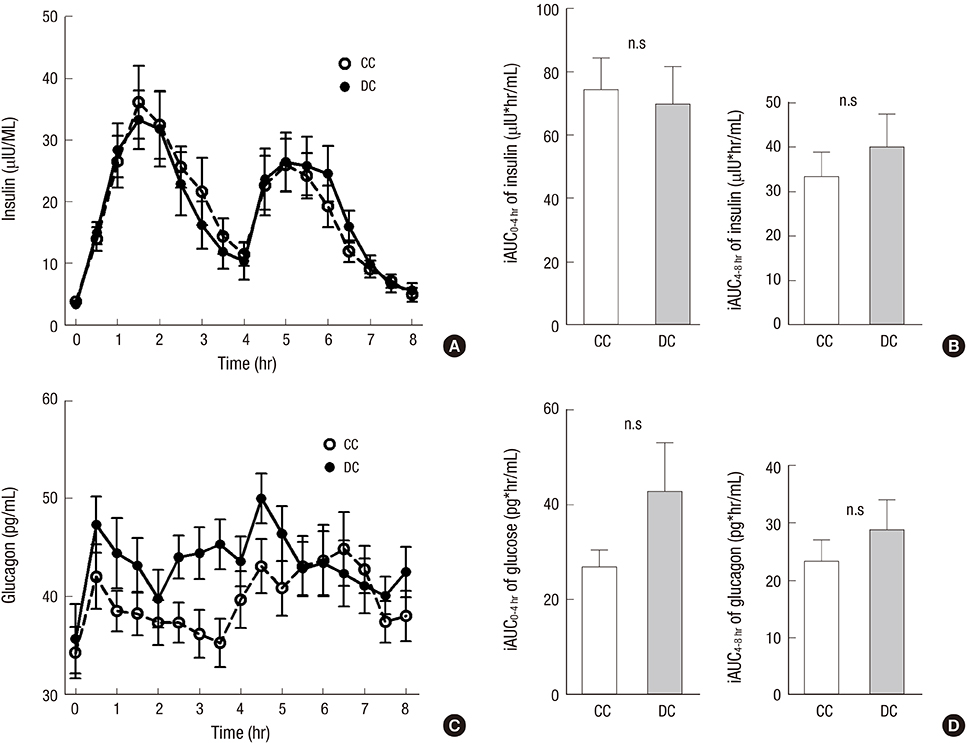
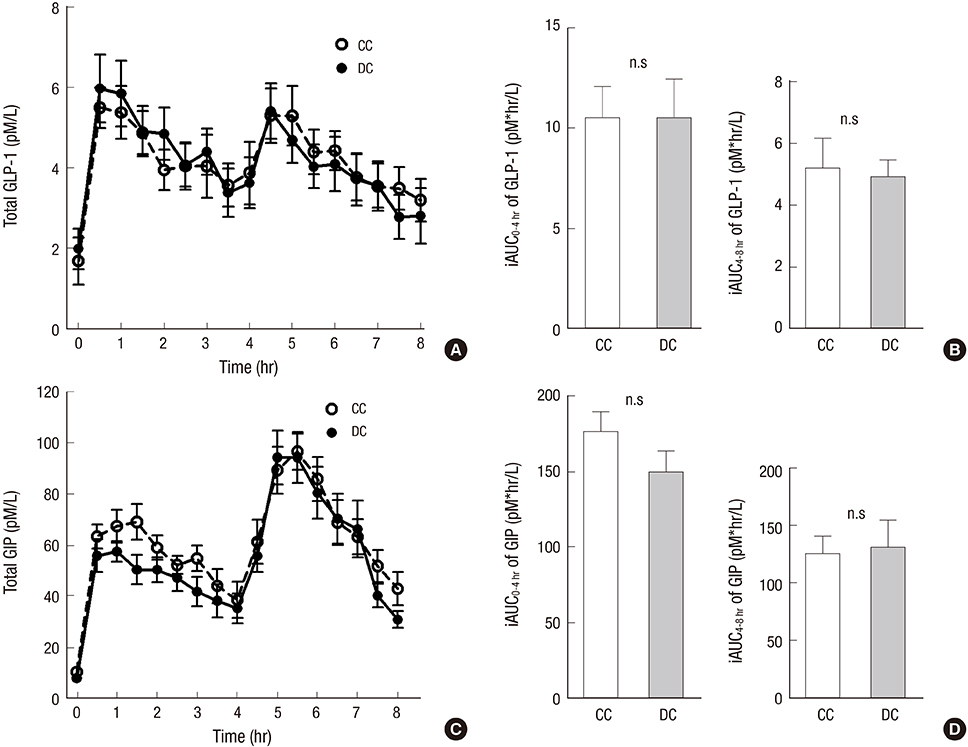
- Dietary fiber improves hyperglycemia in patients with type 2 diabetes through its physicochemical properties and possible modulation of gut hormone secretion, such as glucagon-like peptide 1 (GLP-1). We assessed the effect of dietary fiber-enriched cereal flakes (DC) on postprandial hyperglycemia and gut hormone secretion in patients with type 2 diabetes. Thirteen participants ate isocaloric meals based on either DC or conventional cereal flakes (CC) in a crossover design. DC or CC was provided for dinner, night snack on day 1 and breakfast on day 2, followed by a high-fat lunch. On day 2, the levels of plasma glucose, GLP-1, glucose-dependent insulinotropic polypeptide (GIP), and insulin were measured. Compared to CC, DC intake exhibited a lower post-breakfast 2-hours glucose level (198.5±12.8 vs. 245.9±15.2 mg/dL, P<0.05) and a lower incremental peak of glucose from baseline (101.8±9.1 vs. 140.3±14.3 mg/dL, P<0.001). The incremental area under the curve (iAUC) of glucose after breakfast was lower with DC than with CC (P<0.001). However, there were no differences in the plasma insulin, glucagon, GLP-1, and GIP levels. In conclusion, acute administration of DC attenuates postprandial hyperglycemia without any significant change in the representative glucose-regulating hormones in patients with type 2 diabetes (ClinicalTrials.gov. NCT 01997281).
MeSH Terms
-
Adult
Aged
Area Under Curve
Blood Glucose/*analysis
Cross-Over Studies
Diabetes Mellitus, Type 2/complications/diagnosis/*diet therapy
Dietary Fiber/*therapeutic use
Female
Gastric Inhibitory Polypeptide/blood
Glucagon/blood
Glucagon-Like Peptide 1/*blood
Hemoglobin A, Glycosylated/analysis
Humans
Hyperglycemia/complications/diagnosis
Insulin/blood
Intestines/metabolism
Male
Middle Aged
ROC Curve
Blood Glucose
Gastric Inhibitory Polypeptide
Glucagon
Glucagon-Like Peptide 1
Hemoglobin A, Glycosylated
Insulin
Figure
Cited by 2 articles
-
Effect of Nutrient Preload and Food Order on Glucose, Insulin, and Gut Hormones
Jae Hyun Bae, Young Min Cho
J Korean Diabetes. 2018;19(4):193-199. doi: 10.4093/jkd.2018.19.4.193.Premeal Consumption of a Protein-Enriched, Dietary Fiber-Fortified Bar Decreases Total Energy Intake in Healthy Individuals
Chang Ho Ahn, Jae Hyun Bae, Young Min Cho
Diabetes Metab J. 2019;43(6):879-892. doi: 10.4093/dmj.2018.0202.
Reference
-
1. Kahn SE, Cooper ME, Del Prato S. Pathophysiology and treatment of type 2 diabetes: perspectives on the past, present, and future. Lancet. 2014; 383:1068–1083.2. DeFronzo RA, Eldor R, Abdul-Ghani M. Pathophysiologic approach to therapy in patients with newly diagnosed type 2 diabetes. Diabetes Care. 2013; 36:Suppl 2. S127–S138.3. DECODE Study Group. the European Diabetes Epidemiology Group. Glucose tolerance and cardiovascular mortality: comparison of fasting and 2-hour diagnostic criteria. Arch Intern Med. 2001; 161:397–405.4. Sorkin JD, Muller DC, Fleg JL, Andres R. The relation of fasting and 2-h postchallenge plasma glucose concentrations to mortality: data from the Baltimore Longitudinal Study of Aging with a critical review of the literature. Diabetes Care. 2005; 28:2626–2632.5. Cavalot F, Petrelli A, Traversa M, Bonomo K, Fiora E, Conti M, Anfossi G, Costa G, Trovati M. Postprandial blood glucose is a stronger predictor of cardiovascular events than fasting blood glucose in type 2 diabetes mellitus, particularly in women: lessons from the San Luigi Gonzaga Diabetes Study. J Clin Endocrinol Metab. 2006; 91:813–819.6. Pratley RE, Weyer C. The role of impaired early insulin secretion in the pathogenesis of Type II diabetes mellitus. Diabetologia. 2001; 44:929–945.7. Gerich JE. Pathogenesis and treatment of type 2 (noninsulin-dependent) diabetes mellitus (NIDDM). Horm Metab Res. 1996; 28:404–412.8. Shah P, Vella A, Basu A, Basu R, Schwenk WF, Rizza RA. Lack of suppression of glucagon contributes to postprandial hyperglycemia in subjects with type 2 diabetes mellitus. J Clin Endocrinol Metab. 2000; 85:4053–4059.9. Ceriello A, Quagliaro L, Piconi L, Assaloni R, Da Ros R, Maier A, Esposito K, Giugliano D. Effect of postprandial hypertriglyceridemia and hyperglycemia on circulating adhesion molecules and oxidative stress generation and the possible role of simvastatin treatment. Diabetes. 2004; 53:701–710.10. Hanefeld M, Koehler C, Schaper F, Fuecker K, Henkel E, Temelkova-Kurktschiev T. Postprandial plasma glucose is an independent risk factor for increased carotid intima-media thickness in non-diabetic individuals. Atherosclerosis. 1999; 144:229–235.11. Williams SB, Goldfine AB, Timimi FK, Ting HH, Roddy MA, Simonson DC, Creager MA. Acute hyperglycemia attenuates endothelium-dependent vasodilation in humans in vivo. Circulation. 1998; 97:1695–1701.12. Kawano H, Motoyama T, Hirashima O, Hirai N, Miyao Y, Sakamoto T, Kugiyama K, Ogawa H, Yasue H. Hyperglycemia rapidly suppresses flow-mediated endothelium-dependent vasodilation of brachial artery. J Am Coll Cardiol. 1999; 34:146–154.13. Shiraiwa T, Kaneto H, Miyatsuka T, Kato K, Yamamoto K, Kawashima A, Kanda T, Suzuki M, Imano E, Matsuhisa M, et al. Post-prandial hyperglycemia is an important predictor of the incidence of diabetic microangiopathy in Japanese type 2 diabetic patients. Biochem Biophys Res Commun. 2005; 336:339–345.14. Abbatecola AM, Rizzo MR, Barbieri M, Grella R, Arciello A, Laieta MT, Acampora R, Passariello N, Cacciapuoti F, Paolisso G. Postprandial plasma glucose excursions and cognitive functioning in aged type 2 diabetics. Neurology. 2006; 67:235–240.15. Stein SA, Lamos EM, Davis SN. A review of the efficacy and safety of oral antidiabetic drugs. Expert Opin Drug Saf. 2013; 12:153–175.16. Tahrani AA, Bailey CJ, Del Prato S, Barnett AH. Management of type 2 diabetes: new and future developments in treatment. Lancet. 2011; 378:182–197.17. Zhou J, Martin RJ, Tulley RT, Raggio AM, McCutcheon KL, Shen L, Danna SC, Tripathy S, Hegsted M, Keenan MJ. Dietary resistant starch upregulates total GLP-1 and PYY in a sustained day-long manner through fermentation in rodents. Am J Physiol Endocrinol Metab. 2008; 295:E1160–E1166.18. Kim SH, Hong SB, Suh YJ, Choi YJ, Nam M, Lee HW, Park IB, Chon S, Woo JT, Baik SH, et al. Association between nutrient intake and obesity in type 2 diabetic patients from the Korean National Diabetes Program: a cross-sectional study. J Korean Med Sci. 2012; 27:1188–1195.19. Silva FM, Kramer CK, de Almeida JC, Steemburgo T, Gross JL, Azevedo MJ. Fiber intake and glycemic control in patients with type 2 diabetes mellitus: a systematic review with meta-analysis of randomized controlled trials. Nutr Rev. 2013; 71:790–801.20. Tremaroli V, Bäckhed F. Functional interactions between the gut microbiota and host metabolism. Nature. 2012; 489:242–249.21. Brown AJ, Goldsworthy SM, Barnes AA, Eilert MM, Tcheang L, Daniels D, Muir AI, Wigglesworth MJ, Kinghorn I, Fraser NJ, et al. The orphan G protein-coupled receptors GPR41 and GPR43 are activated by propionate and other short chain carboxylic acids. J Biol Chem. 2003; 278:11312–11319.22. Tolhurst G, Heffron H, Lam YS, Parker HE, Habib AM, Diakogiannaki E, Cameron J, Grosse J, Reimann F, Gribble FM. Short-chain fatty acids stimulate glucagon-like peptide-1 secretion via the G-protein-coupled receptor FFAR2. Diabetes. 2012; 61:364–371.23. Cho YM, Fujita Y, Kieffer TJ. Glucagon-like peptide-1: glucose homeostasis and beyond. Annu Rev Physiol. 2014; 76:535–559.24. Massimino SP, McBurney MI, Field CJ, Thomson AB, Keelan M, Hayek MG, Sunvold GD. Fermentable dietary fiber increases GLP-1 secretion and improves glucose homeostasis despite increased intestinal glucose transport capacity in healthy dogs. J Nutr. 1998; 128:1786–1793.25. Tarini J, Wolever TM. The fermentable fibre inulin increases postprandial serum short-chain fatty acids and reduces free-fatty acids and ghrelin in healthy subjects. Appl Physiol Nutr Metab. 2010; 35:9–16.26. Freeland KR, Wilson C, Wolever TM. Adaptation of colonic fermentation and glucagon-like peptide-1 secretion with increased wheat fibre intake for 1 year in hyperinsulinaemic human subjects. Br J Nutr. 2010; 103:82–90.27. Nilsson AC, Ostman EM, Holst JJ, Björck IM. Including indigestible carbohydrates in the evening meal of healthy subjects improves glucose tolerance, lowers inflammatory markers, and increases satiety after a subsequent standardized breakfast. J Nutr. 2008; 138:732–739.28. Ebbeling CB, Ludwig DS. Treating obesity in youth: should dietary glycemic load be a consideration? Adv Pediatr. 2001; 48:179–212.29. Sands AL, Leidy HJ, Hamaker BR, Maguire P, Campbell WW. Consumption of the slow-digesting waxy maize starch leads to blunted plasma glucose and insulin response but does not influence energy expenditure or appetite in humans. Nutr Res. 2009; 29:383–390.30. Li M, Piao JH, Tian Y, Li WD, Li KJ, Yang XG. Postprandial glycaemic and insulinaemic responses to GM-resistant starch-enriched rice and the production of fermentation-related H2 in healthy Chinese adults. Br J Nutr. 2010; 103:1029–1034.31. Rosén LA, Ostman EM, Björck IM. Effects of cereal breakfasts on postprandial glucose, appetite regulation and voluntary energy intake at a subsequent standardized lunch; focusing on rye products. Nutr J. 2011; 10:7.32. Jenkins DJ, Wolever TM, Leeds AR, Gassull MA, Haisman P, Dilawari J, Goff DV, Metz GL, Alberti KG. Dietary fibres, fibre analogues, and glucose tolerance: importance of viscosity. BMJ. 1978; 1:1392–1394.33. Frost GS, Brynes AE, Dhillo WS, Bloom SR, McBurney MI. The effects of fiber enrichment of pasta and fat content on gastric emptying, GLP-1, glucose, and insulin responses to a meal. Eur J Clin Nutr. 2003; 57:293–298.34. Bodinham CL, Al-Mana NM, Smith L, Robertson MD. Endogenous plasma glucagon-like peptide-1 following acute dietary fibre consumption. Br J Nutr. 2013; 110:1429–1433.35. Robertson MD, Currie JM, Morgan LM, Jewell DP, Frayn KN. Prior short-term consumption of resistant starch enhances postprandial insulin sensitivity in healthy subjects. Diabetologia. 2003; 46:659–665.36. Johansson EV, Nilsson AC, Östman EM, Björck IM. Effects of indigestible carbohydrates in barley on glucose metabolism, appetite and voluntary food intake over 16 h in healthy adults. Nutr J. 2013; 12:46.37. Lee KU, Lee HK, Koh CS, Min HK. Artificial induction of intravascular lipolysis by lipid-heparin infusion leads to insulin resistance in man. Diabetologia. 1988; 31:285–290.38. Ludwig DS. The glycemic index: physiological mechanisms relating to obesity, diabetes, and cardiovascular disease. JAMA. 2002; 287:2414–2423.39. Robertson MD, Bickerton AS, Dennis AL, Vidal H, Frayn KN. Insulin-sensitizing effects of dietary resistant starch and effects on skeletal muscle and adipose tissue metabolism. Am J Clin Nutr. 2005; 82:559–567.40. Bodinham CL, Smith L, Wright J, Frost GS, Robertson MD. Dietary fibre improves first-phase insulin secretion in overweight individuals. PLoS One. 2012; 7:e40834.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Premeal Consumption of a Protein-Enriched, Dietary Fiber-Fortified Bar Decreases Total Energy Intake in Healthy Individuals
- Rice Cakes Containing Dietary Fiber Supplemented with or without Artemisia Annua and Gynura Procumbens Merr. Alleviated the Risk Factors of Metabolic Syndrome
- Gut Hormone Response to Diet
- Diet Therapy for Postprandial Hyperglycemia in Patients with Diabetes
- Calcium-sensing receptor-mediated L-tryptophan-induced secretion of cholecystokinin and glucose-dependent insulinotropic peptide in swine duodenum