Korean J Radiol.
2016 Feb;17(1):117-126. 10.3348/kjr.2016.17.1.117.
Pre-Operative Perfusion Skewness and Kurtosis Are Potential Predictors of Progression-Free Survival after Partial Resection of Newly Diagnosed Glioblastoma
- Affiliations
-
- 1Department of Radiology, Dankook University Hospital, Cheonan 31116, Korea.
- 2Department of Radiology and Research Institute of Radiology, University of Ulsan College of Medicine, Asan Medical Center, Seoul 05505, Korea. radhskim@gmail.com
- KMID: 2351171
- DOI: http://doi.org/10.3348/kjr.2016.17.1.117
Abstract
OBJECTIVE
To determine whether pre-operative perfusion skewness and kurtosis derived from normalized cerebral blood volume (nCBV) histograms are associated with progression-free survival (PFS) of patients after partial resection of newly diagnosed glioblastoma.
MATERIALS AND METHODS
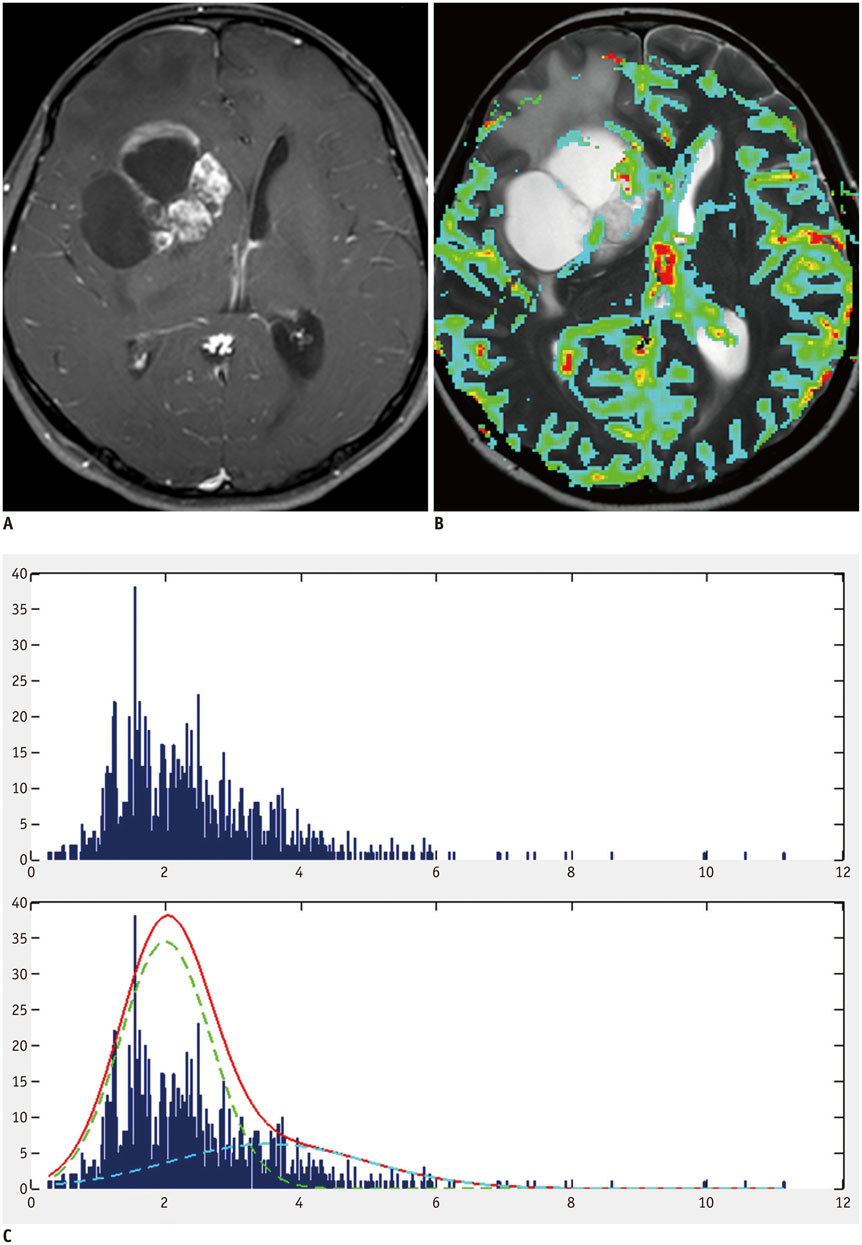
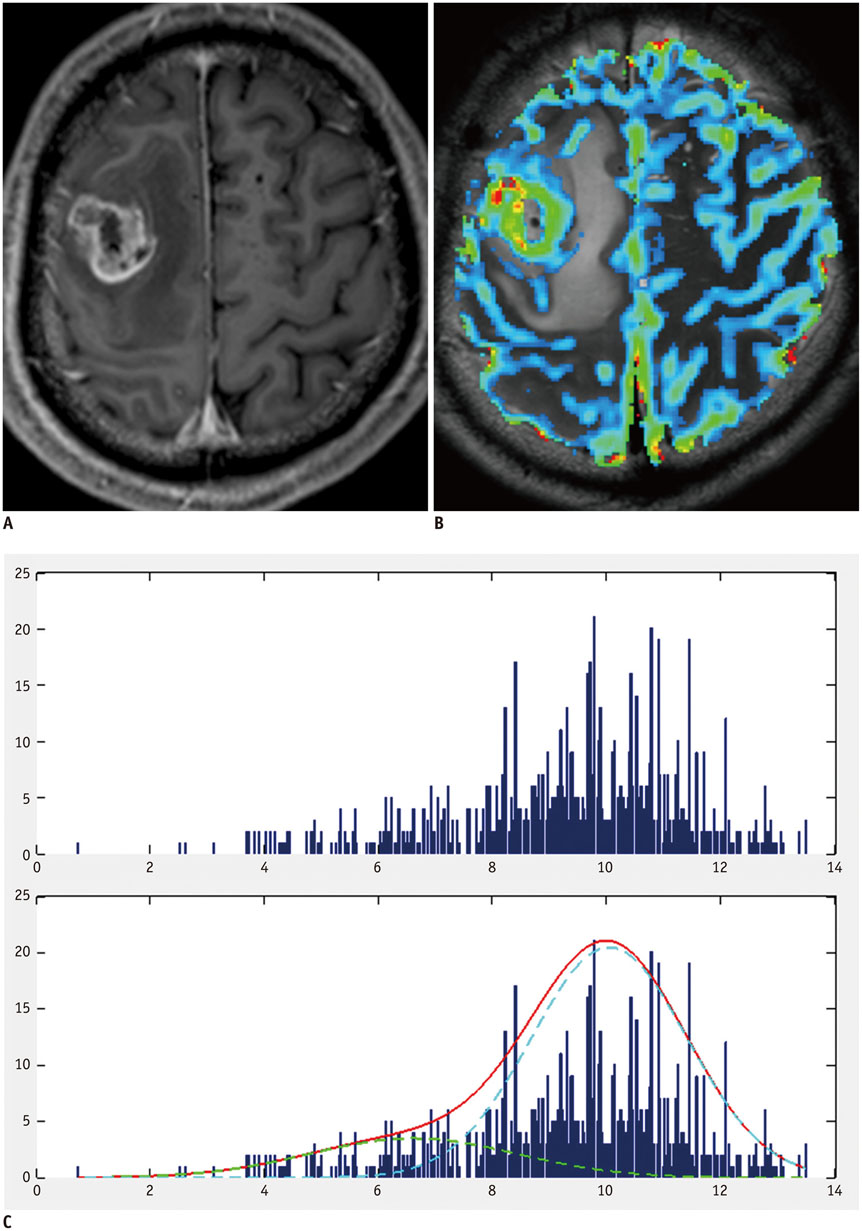
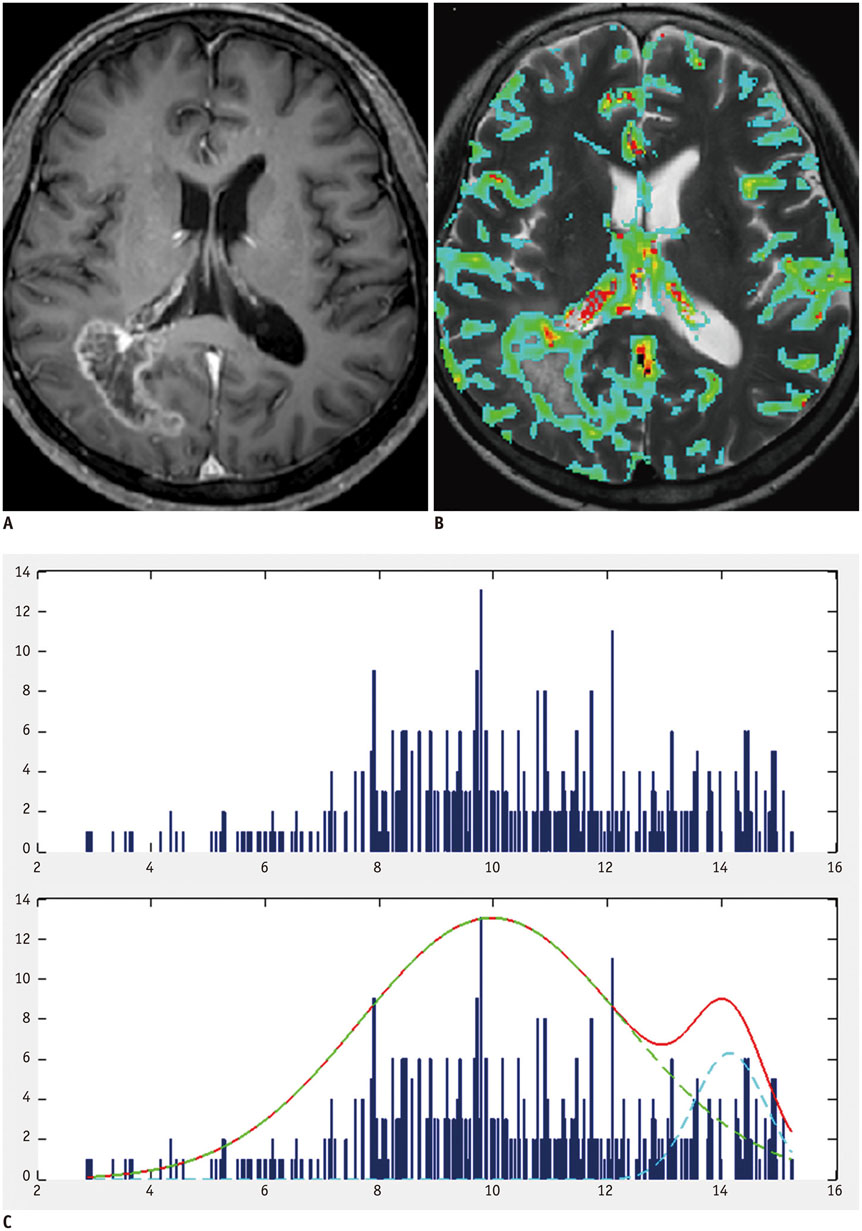
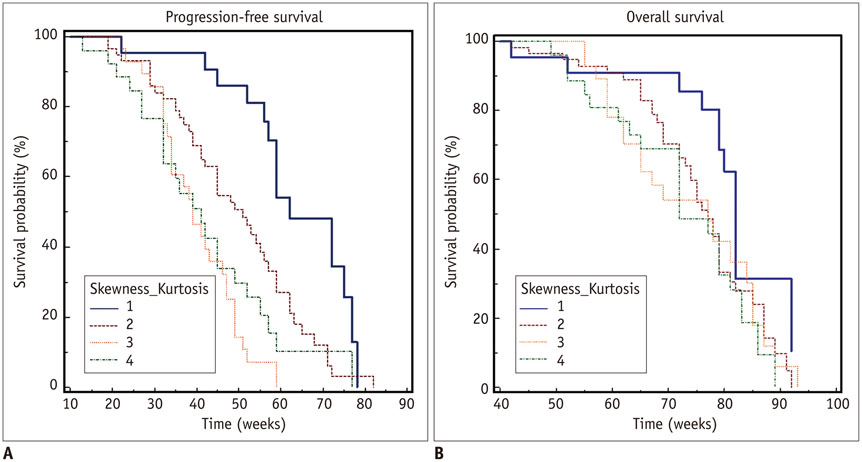
A total of 135 glioblastoma patients who had undergone partial resection of tumor (resection of < 50% of pre-operative tumor volume or surgical biopsy) confirmed with immediate postsurgical MRI and examined with both conventional MRI and dynamic susceptibility contrast (DSC) perfusion MRI before the surgery were retrospectively reviewed in this study. They had been followed up post-surgical chemoradiotherapy for tumor progression. Using histogram analyses of nCBV derived from pre-operative DSC perfusion MRI, patients were sub-classified into the following four groups: positive skewness and leptokurtosis (group 1); positive skewness and platykurtosis (group 2); negative skewness and leptokurtosis (group 3); negative skewness and platykurtosis (group 4). Kaplan-Meier analysis and multivariable Cox proportional hazards regression analysis were performed to determine whether clinical and imaging covariates were associated with PFS or overall survival (OS) of these patients.
RESULTS
According to the Kaplan-Meier method, median PFS of group 1, 2, 3, and 4 was 62, 51, 39, and 41 weeks, respectively, with median OS of 82, 77, 77, and 72 weeks, respectively. In multivariable analyses with Cox proportional hazards regression, pre-operative skewness/kurtosis pattern (hazard ratio: 2.98 to 4.64; p < 0.001), Karnofsky performance scale score (hazard ratio: 1.04; p = 0.003), and post-operative tumor volume (hazard ratio: 1.04; p = 0.02) were independently associated with PFS but not with OS.
CONCLUSION
Higher skewness and kurtosis of nCBV histogram before surgery were associated with longer PFS in patients with newly diagnosed glioblastoma after partial tumor resection.
MeSH Terms
-
Adult
Aged
Aged, 80 and over
Brain Neoplasms/*mortality/pathology/*surgery
Chemoradiotherapy
Disease-Free Survival
Female
Glioblastoma/*mortality/pathology/*surgery
Humans
Image Interpretation, Computer-Assisted
Image Processing, Computer-Assisted
Kaplan-Meier Estimate
Magnetic Resonance Imaging/methods
Male
Middle Aged
Regression Analysis
Retrospective Studies
Statistical Distributions
Tumor Burden
Young Adult
Figure
Cited by 1 articles
-
Influence of B1-Inhomogeneity on Pharmacokinetic Modeling of Dynamic Contrast-Enhanced MRI: A Simulation Study
Bumwoo Park, Byung Se Choi, Yu Sub Sung, Dong-Cheol Woo, Woo Hyun Shim, Kyung Won Kim, Yoon Seok Choi, Sang Joon Pae, Ji-Yeon Suh, Hyungjoon Cho, Jeong Kon Kim
Korean J Radiol. 2017;18(4):585-596. doi: 10.3348/kjr.2017.18.4.585.
Reference
-
1. Smith JS, Jenkins RB. Genetic alterations in adult diffuse glioma: occurrence, significance, and prognostic implications. Front Biosci. 2000; 5:D213–D231.2. Cao Y, Tsien CI, Nagesh V, Junck L, Ten Haken R, Ross BD, et al. Survival prediction in high-grade gliomas by MRI perfusion before and during early stage of RT [corrected]. Int J Radiat Oncol Biol Phys. 2006; 64:876–885.3. Law M, Young RJ, Babb JS, Peccerelli N, Chheang S, Gruber ML, et al. Gliomas: predicting time to progression or survival with cerebral blood volume measurements at dynamic susceptibility-weighted contrast-enhanced perfusion MR imaging. Radiology. 2008; 247:490–498.4. Jahng GH, Li KL, Ostergaard L, Calamante F. Perfusion magnetic resonance imaging: a comprehensive update on principles and techniques. Korean J Radiol. 2014; 15:554–577.5. Lee EK, Choi SH, Yun TJ, Kang KM, Kim TM, Lee SH, et al. Prediction of response to concurrent chemoradiotherapy with temozolomide in glioblastoma: application of immediate post-operative dynamic susceptibility contrast and diffusion-weighted MR imaging. Korean J Radiol. 2015; 16:1341–1348.6. Kim HS, Kim JH, Kim SH, Cho KG, Kim SY. Posttreatment high-grade glioma: usefulness of peak height position with semiquantitative MR perfusion histogram analysis in an entire contrast-enhanced lesion for predicting volume fraction of recurrence. Radiology. 2010; 256:906–915.7. Emblem KE, Bjornerud A. An automatic procedure for normalization of cerebral blood volume maps in dynamic susceptibility contrast-based glioma imaging. AJNR Am J Neuroradiol. 2009; 30:1929–1932.8. Emblem KE, Nedregaard B, Nome T, Due-Tonnessen P, Hald JK, Scheie D, et al. Glioma grading by using histogram analysis of blood volume heterogeneity from MR-derived cerebral blood volume maps. Radiology. 2008; 247:808–817.9. Law M, Young R, Babb J, Pollack E, Johnson G. Histogram analysis versus region of interest analysis of dynamic susceptibility contrast perfusion MR imaging data in the grading of cerebral gliomas. AJNR Am J Neuroradiol. 2007; 28:761–766.10. Emblem KE, Scheie D, Due-Tonnessen P, Nedregaard B, Nome T, Hald JK, et al. Histogram analysis of MR imaging-derived cerebral blood volume maps: combined glioma grading and identification of low-grade oligodendroglial subtypes. AJNR Am J Neuroradiol. 2008; 29:1664–1670.11. Wen PY, Macdonald DR, Reardon DA, Cloughesy TF, Sorensen AG, Galanis E, et al. Updated response assessment criteria for high-grade gliomas: response assessment in neuro-oncology working group. J Clin Oncol. 2010; 28:1963–1972.12. Rosen BR, Belliveau JW, Vevea JM, Brady TJ. Perfusion imaging with NMR contrast agents. Magn Reson Med. 1990; 14:249–265.13. Ostergaard L, Weisskoff RM, Chesler DA, Gyldensted C, Rosen BR. High resolution measurement of cerebral blood flow using intravascular tracer bolus passages. Part I: Mathematical approach and statistical analysis. Magn Reson Med. 1996; 36:715–725.14. Boxerman JL, Schmainda KM, Weisskoff RM. Relative cerebral blood volume maps corrected for contrast agent extravasation significantly correlate with glioma tumor grade, whereas uncorrected maps do not. AJNR Am J Neuroradiol. 2006; 27:859–867.15. Pope WB, Kim HJ, Huo J, Alger J, Brown MS, Gjertson D, et al. Recurrent glioblastoma multiforme: ADC histogram analysis predicts response to bevacizumab treatment. Radiology. 2009; 252:182–189.16. Nowosielski M, Recheis W, Goebel G, Güler O, Tinkhauser G, Kostron H, et al. ADC histograms predict response to antiangiogenic therapy in patients with recurrent high-grade glioma. Neuroradiology. 2011; 53:291–302.17. Bisdas S, Kirkpatrick M, Giglio P, Welsh C, Spampinato MV, Rumboldt Z. Cerebral blood volume measurements by perfusion-weighted MR imaging in gliomas: ready for prime time in predicting short-term outcome and recurrent disease? AJNR Am J Neuroradiol. 2009; 30:681–688.18. Barajas RF Jr, Hodgson JG, Chang JS, Vandenberg SR, Yeh RF, Parsa AT, et al. Glioblastoma multiforme regional genetic and cellular expression patterns: influence on anatomic and physiologic MR imaging. Radiology. 2010; 254:564–576.19. Oh J, Henry RG, Pirzkall A, Lu Y, Li X, Catalaa I, et al. Survival analysis in patients with glioblastoma multiforme: predictive value of choline-to-N-acetylaspartate index, apparent diffusion coefficient, and relative cerebral blood volume. J Magn Reson Imaging. 2004; 19:546–554.20. Law M, Oh S, Babb JS, Wang E, Inglese M, Zagzag D, et al. Low-grade gliomas: dynamic susceptibility-weighted contrast-enhanced perfusion MR imaging--prediction of patient clinical response. Radiology. 2006; 238:658–667.21. Lev MH, Ozsunar Y, Henson JW, Rasheed AA, Barest GD, Harsh GR 4th, et al. Glial tumor grading and outcome prediction using dynamic spin-echo MR susceptibility mapping compared with conventional contrast-enhanced MR: confounding effect of elevated rCBV of oligodendrogliomas [corrected]. AJNR Am J Neuroradiol. 2004; 25:214–221.22. Price SJ, Jena R, Burnet NG, Hutchinson PJ, Dean AF, Peña A, et al. Improved delineation of glioma margins and regions of infiltration with the use of diffusion tensor imaging: an image-guided biopsy study. AJNR Am J Neuroradiol. 2006; 27:1969–1974.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of Cadherin-11 Expression on the Prognosis of a Newly Diagnosed Primary Glioblastoma
- Validation of the Effectiveness and Safety of Temozolomide during and after Radiotherapy for Newly Diagnosed Glioblastomas: 10-year Experience of a Single Institution
- Recurrence pattern of glioblastoma treated with intensity-modulated radiation therapy versus three-dimensional conformal radiation therapy
- Temozolomide during and after Radiotherapy for Newly Diagnosed Glioblastomas : A Prospective Multicenter Study of Korean Patients
- Supramaximal Resection for Glioblastoma: Redefining the Extent of Resection Criteria and Its Impact on Survival