J Gynecol Oncol.
2015 Oct;26(4):311-319. 10.3802/jgo.2015.26.4.311.
Palonosetron versus granisetron in combination with aprepitant for the prevention of chemotherapy-induced nausea and vomiting in patients with gynecologic cancer
- Affiliations
-
- 1Department of Obstetrics and Gynecology, Osaka Medical College, Osaka, Japan. y-terai@poh.osaka-med.ac.jp
- KMID: 2345922
- DOI: http://doi.org/10.3802/jgo.2015.26.4.311
Abstract
OBJECTIVE
There is no research regarding the appropriate antiemetic agents for female patients, especially those receiving moderately emetogenic chemotherapy (MEC). We evaluated the antiemetic efficacy of a combination of 5-HT3 receptor with/without aprepitant in patients with gynecological cancer treated with the TC (paclitaxel and carboplatin) regimen of MEC.
METHODS
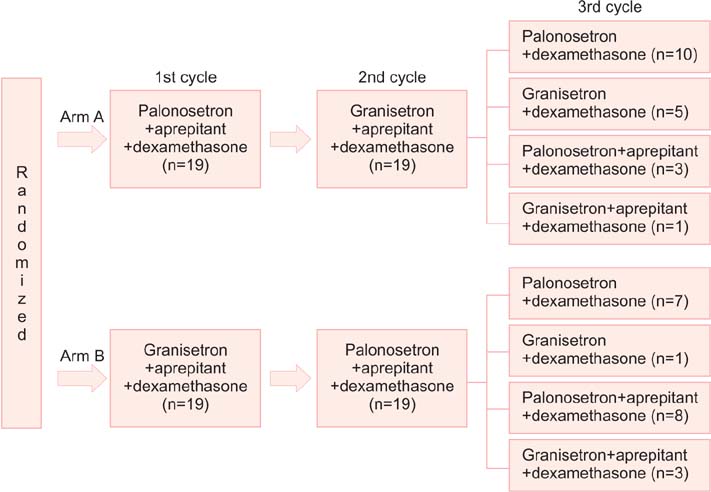
We enrolled 38 patients diagnosed with gynecologic cancer and scheduled to receive the TC regimen. The patients were randomly assigned to receive a 5-HT3 receptor antagonist, either palonosetron in the first cycle followed by granisetron in the second cycle or vice versa. In the third cycle, all patients received a combination of the 5-HT3 receptor and dexamethasone with/without aprepitant.
RESULTS
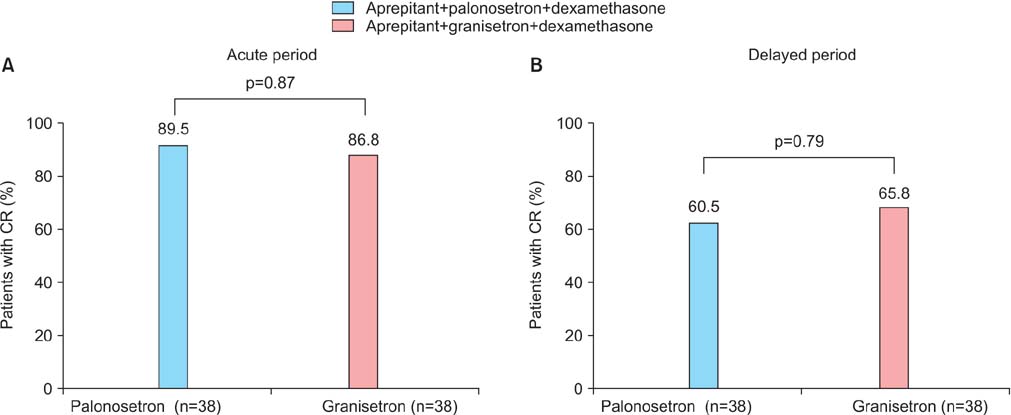
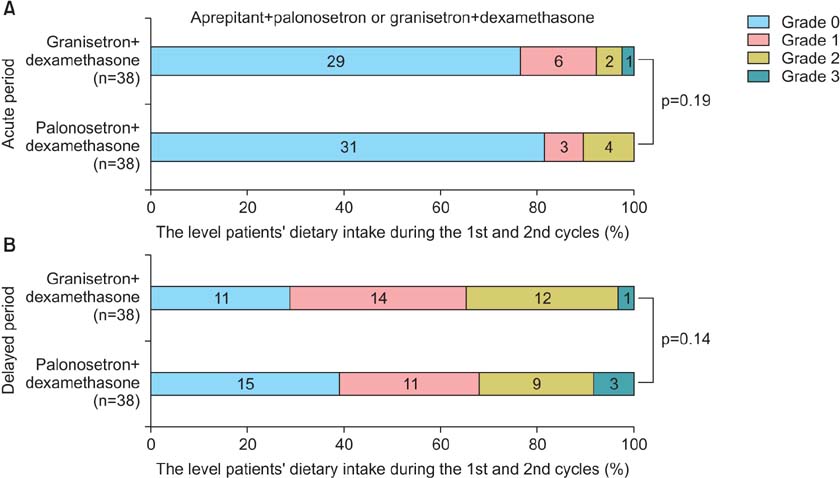
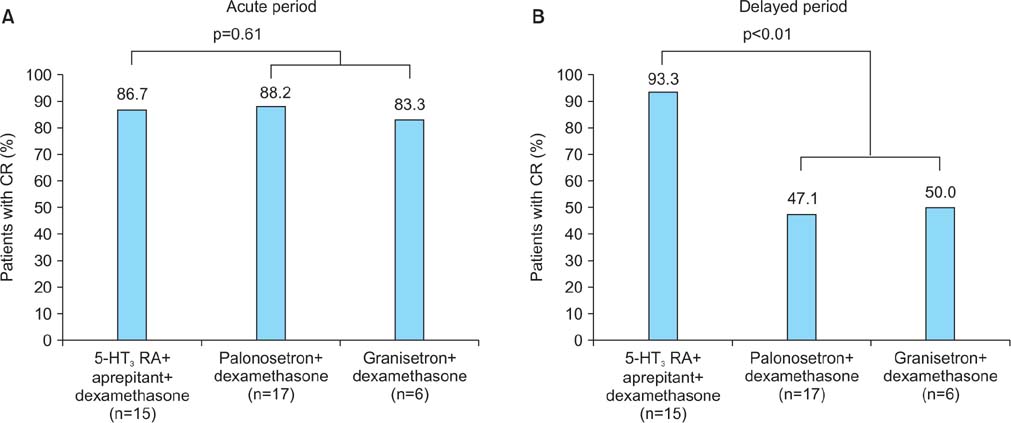
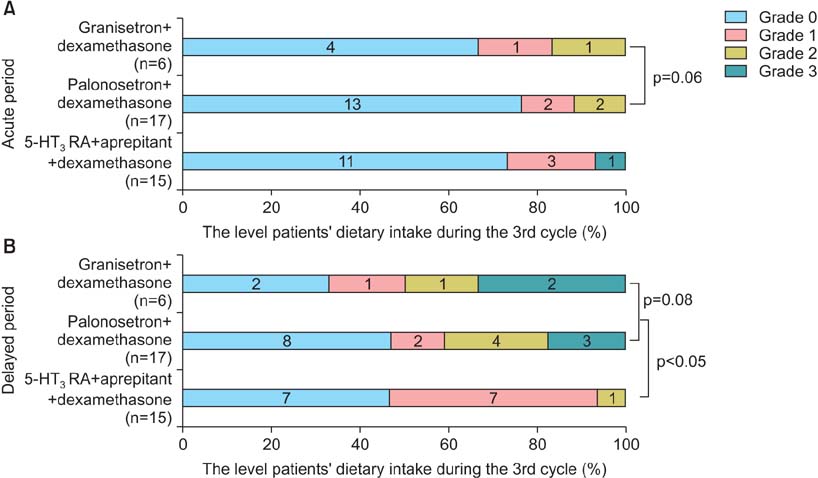
When three drugs were administered, palonosetron consistently produced an equivalent complete response (CR) rate to granisetron in the acute phase (89.5% vs. 86.8%, p=0.87) and delayed phase (60.5% vs. 65.8%, p=0.79). With regard to the change in dietary intake, palonosetron exhibited similar efficacy to granisetron in the acute phase (92.1% vs. 89.4%, p=0.19) and delayed phase (65.7% vs. 68.4%, p=0.14). However, in the delayed phase, the addition of aprepitant therapy with a 5-HT3 receptor antagonist and dexamethasone produced a higher CR rate than a 5-HT3 receptor antagonist with dexamethasone (93.3% vs. 47.8%, p<0.001) and allowed the patients to maintain a higher level of dietary intake (93.3% vs. 56.5%, p<0.001).
CONCLUSION
The addition of aprepitant therapy was more effective than the control therapy of a 5-HT3 receptor antagonist, and dexamethasone in gynecological cancer patients treated with the TC regimen.
Keyword
MeSH Terms
-
Adult
Aged
Antiemetics/*administration & dosage
Antineoplastic Combined Chemotherapy Protocols/adverse effects
Carboplatin/administration & dosage/adverse effects
Cross-Over Studies
Diet
Drug Administration Schedule
Female
Genital Neoplasms, Female/*drug therapy
Granisetron/administration & dosage
Humans
Isoquinolines/administration & dosage
Middle Aged
Morpholines/administration & dosage
Nausea/chemically induced/*prevention & control
Paclitaxel/administration & dosage/adverse effects
Quinuclidines/administration & dosage
Serotonin 5-HT3 Receptor Antagonists
Vomiting/chemically induced/*prevention & control
Antiemetics
Carboplatin
Granisetron
Isoquinolines
Morpholines
Quinuclidines
Serotonin 5-HT3 Receptor Antagonists
Paclitaxel
Figure
Cited by 1 articles
-
Efficacy of palonosetron plus dexamethasone in preventing chemotherapy-induced nausea and emesis in patients receiving carboplatin-based chemotherapy for gynecologic cancers: a phase II study by the West Japan Gynecologic Oncology Group (WJGOG 131)
Shin Nishio, Satomi Aihara, Mototsugu Shimokawa, Akira Fujishita, Shuichi Taniguchi, Toru Hachisuga, Shintaro Yanazume, Hiroaki Kobayashi, Fumihiro Murakami, Fumitaka Numa, Kohei Kotera, Naofumi Okura, Naoyuki Toki, Masatoshi Yokoyama, Kimio Ushijima
J Gynecol Oncol. 2018;29(5):. doi: 10.3802/jgo.2018.29.e77.
Reference
-
1. Hesketh PJ. Chemotherapy-induced nausea and vomiting. N Engl J Med. 2008; 358:2482–2494.2. Roila F, Herrstedt J, Aapro M, Gralla RJ, Einhorn LH, Ballatori E, et al. Guideline update for MASCC and ESMO in the prevention of chemotherapy- and radiotherapy-induced nausea and vomiting: results of the Perugia consensus conference. Ann Oncol. 2010; 21:Suppl 5. v232–v243.3. Basch E, Prestrud AA, Hesketh PJ, Kris MG, Feyer PC, Somerfield MR, et al. Antiemetics: American Society of Clinical Oncology clinical practice guideline update. J Clin Oncol. 2011; 29:4189–4198.4. Japan Society of Clinical Oncology. Japan Society of Clinical Oncology guideline for antiemetic, ver. 1. Tokyo: Japan Society of Clinical Oncology;2010.5. Tonato M, Roila F, Del Favero A. Methodology of antiemetic trials: a review. Ann Oncol. 1991; 2:107–114.6. Kaizer L, Warr D, Hoskins P, Latreille J, Lofters W, Yau J, et al. Effect of schedule and maintenance on the antiemetic efficacy of ondansetron combined with dexamethasone in acute and delayed nausea and emesis in patients receiving moderately emetogenic chemotherapy: a phase III trial by the National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol. 1994; 12:1050–1057.7. The Italian Group for Antiemetic Research. Dexamethasone alone or in combination with ondansetron for the prevention of delayed nausea and vomiting induced by chemotherapy. N Engl J Med. 2000; 342:1554–1559.8. Pater JL, Lofters WS, Zee B, Dempsey E, Walde D, Moquin JP, et al. The role of the 5-HT3 antagonists ondansetron and dolasetron in the control of delayed onset nausea and vomiting in patients receiving moderately emetogenic chemotherapy. Ann Oncol. 1997; 8:181–185.9. Tian W, Wang Z, Zhou J, Zhang S, Wang J, Chen Q, et al. Randomized, double-blind, crossover study of palonosetron compared with granisetron for the prevention of chemotherapy-induced nausea and vomiting in a Chinese population. Med Oncol. 2011; 28:71–78.10. Saito M, Aogi K, Sekine I, Yoshizawa H, Yanagita Y, Sakai H, et al. Palonosetron plus dexamethasone versus granisetron plus dexamethasone for prevention of nausea and vomiting during chemotherapy: a double-blind, double-dummy, randomised, comparative phase III trial. Lancet Oncol. 2009; 10:115–124.11. Ohzawa H, Miki A, Hozumi Y, Miyazaki C, Sagara Y, Tanaka Y, et al. Comparison between the antiemetic effects of palonosetron and granisetron in breast cancer patients treated with anthracycline-based regimens. Oncol Lett. 2015; 9:119–124.12. Sekine I, Segawa Y, Kubota K, Saeki T. Risk factors of chemotherapy-induced nausea and vomiting: index for personalized antiemetic prophylaxis. Cancer Sci. 2013; 104:711–717.13. Grote T, Hajdenberg J, Cartmell A, Ferguson S, Ginkel A, Charu V. Combination therapy for chemotherapy-induced nausea and vomiting in patients receiving moderately emetogenic chemotherapy: palonosetron, dexamethasone, and aprepitant. J Support Oncol. 2006; 4:403–408.14. Molassiotis A, Stamataki Z, Kontopantelis E. Development and preliminary validation of a risk prediction model for chemotherapy-related nausea and vomiting. Support Care Cancer. 2013; 21:2759–2767.15. Tanioka M, Kitao A, Matsumoto K, Shibata N, Yamaguchi S, Fujiwara K, et al. A randomised, placebo-controlled, double-blind study of aprepitant in nondrinking women younger than 70 years receiving moderately emetogenic chemotherapy. Br J Cancer. 2013; 109:859–865.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Aprepitant in combination with palonosetron for the prevention of postoperative nausea and vomiting in female patients using intravenous patient-controlled analgesia
- Prophylactic antiemetics therapy against gynecologic cancer chemotherapy
- Efficacy according to Dexamethasone Dose in Combination with Aprepitant and Granisetron during Cisplatin-based Chemotherapy
- Effect of Granisetron Plus Dexamethasone in the Prevention of Delayed Nausea and Vomiting
- Comparative Study of an Ondansetron and a Ramosetron an Aprepitant in the Control of Nausea and Vomiting in Gynecologinc Cancer Patient with Chemotherapy