J Korean Orthop Assoc.
2008 Dec;43(6):775-782.
Effect of Adrenomedullin on Proliferation and Mitogen-Activated Protein Kinase Activity in Osteosarcoma Cell Lines (MG63, U2OS)
- Affiliations
-
- 1Department of Orthopaedic Surgery, Chonbuk National University Medical School, Research Institute of Clinical Science, Jeonju, Korea. jrkeem@chonbuk.ac.kr
Abstract
-
PURPOSE: The aims investigated how how human osteosarcoma cell proliferation and the MAP kinases cascade are regulated, in the MG63 and U2OS human osteosarcoma cell lines after stimulating them with adrenomedullin (AM) with particular focus on extracellular signal-regulated kinase 1/2 (ERK 1 and 2) activation.
MATERIALS AND METHODS
A cell proliferation assay was used to examined the effects of AM on the osteosarcoma cell lines (MG63 and U2OS). The effects of AM on ERK1/2 were examed by Western blot analysis. The roles of ERK 1/2 in the AM-induced proliferative signaling pathways in the two cell types were examed using PD98059, a selective inhibitor of the mitogen activated protein-ERK kinase (MEK) pathway.
RESULTS
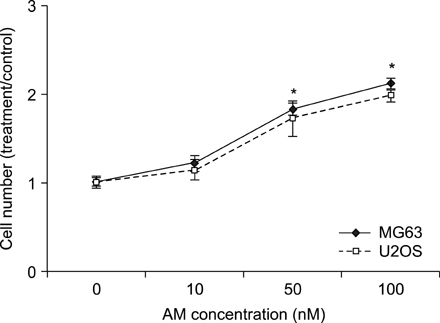
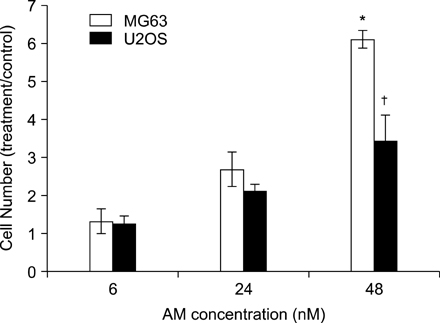
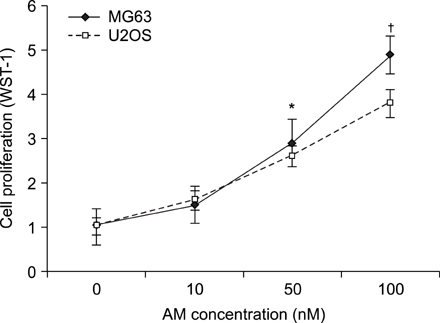
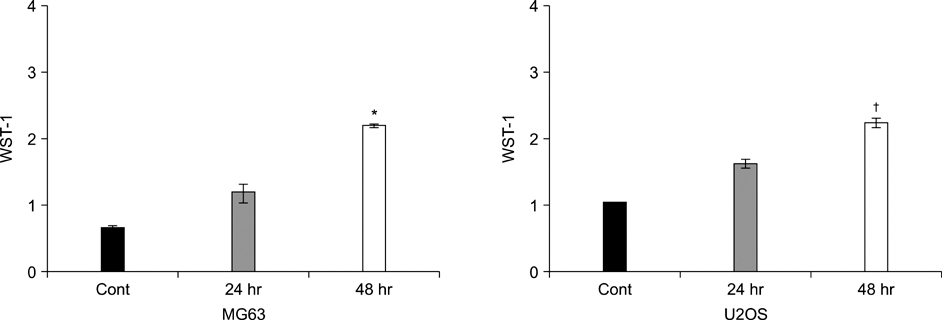
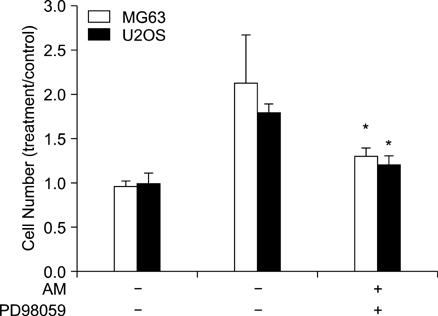
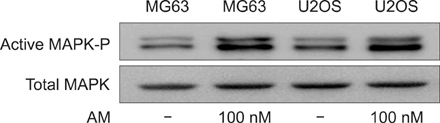
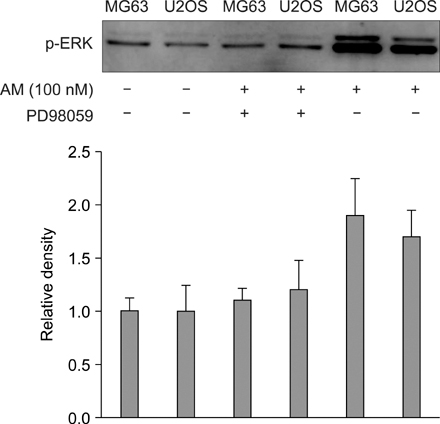
The addition of AM to the medium containing the osteosarcoma MG63 and U2OS cells induced proliferation in a dose-dependent manner. AM strongly activated ERK 1/2 mediated cell proliferation signaling, which was prevented using PD98059.
CONCLUSION
These results suggest that AM plays an important role in the proliferation of human osteosarcoma MG63 and U2OS cells, and ERK kinase pathway plays a signal transduction role in AM treated human osteosarcoma MG63 and U2OS cell lines.
MeSH Terms
Figure
Reference
-
1. Chen P, Xie H, Well A. Mitogenic signaling from the EGF receptor is attenuated by a phospholipase C-gamma/protein kinase C feedback mechanism. Mol Biol Cell. 1996. 76:871–881.
Article2. Harris AL. Hypoxia - a key regulatory factor in tumor growth. Nat Rev Cancer. 2002. 2:38–47.3. Hinson JP, Thomson LM, Kapas S. Adrenomedullin and CGRP receptors mediate different effects in the rat adrenal cortex. Endocr Res. 1998. 24:725–728.
Article4. Kitamura K, Kangawa K, Kawamoto M, et al. Adrenomeullin: a novel hypotensive peptide isolated from human pheochromocytoma. Biochem Biophys Res Commun. 1993. 192:553–560.5. Lai CF, Chaudhary L, Fausto A, et al. ERK is essential for growth, differentiation, intergrin expression and cell function in human osteoblastic cells. J Bio Chem. 2001. 276:14443–14450.6. Lajeunesse D, Frondoza C, Schoffield B, Sacktor B. Osteocalcin secretion by the human osteosarcoma cell line MG-63. J Bone Miner Res. 1990. 5:915–922.
Article7. Lou J, Tu Y, Li S, Manske PR. Involvement of ERK in BMP-2 induced osteoblastic differentiation of mesenchymal progenitor cell line C3H10T1/2. Biochem Biophys Res Commun. 2000. 268:757–762.
Article8. Martinez A, Miller Mj, Unsworth EJ, Siegfried JM, Cuttitta F. Expression of adrenomedullin in normal human lung and in pulmonary tumors. Endocrinology. 1996. 136:4099–4105.
Article9. Neary JT. MAPK cascades in cell growth and death. News Physiol Sci. 1997. 12:286–293.
Article10. Nikitenko LL, Smith DM, Bicknell R, Rees MC. Transcriptional regulation of the CRLR gene in human microvascular endothelial cells by hypoxia. FASEB J. 2003. 17:1499–1501.
Article11. Nikitenko LL, Smith DM, Hague S, Brucknell R, Rees MC. Adrenomedullin and the microvasculature. Trends Pharm Sci. 2002. 23:101–103.
Article12. Quafik L, Sauze S, Boudouresque F, et al. Neutralization of adrenomedullin inhibits the growth of human glioblastoma cell lines in vitro and suppresses tumor xenograft growth in vivo. Am J Pathol. 2002. 160:1279–1292.13. Satoh F, Takahashi K, Murakami O, et al. Adrenomedullin in human brain, adrenal glands and tumor tissues of pheochromocytoma, ganglioneuroblastoma and neuroblastoma. J Clin Endocrinol Matab. 1995. 80:1750–1752.
Article14. Seger R, Krebs EG. The MAPK signaling cascade. FASEB J. 1995. 9:726–735.
Article15. Zimmermann U, Fischer JA, Frei K, Fischer AH, Reinscheid RK, Muff R. Identification of adrenomedullin receptors in cultured rat astrocytes and in neuroblastoma x glioma hybrid cells (NG108-15). Brain Res. 1996. 724:238–245.16. Zudaire E, Martinez A, Cuttitta F. Adrenomedullin and cancer. Regul Pept. 2003. 112:175–183.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- ERK1/2 Activation Modulates Matrix Metalloproteinase 2 Activity and Ezrin Expression in Osteosarcoma Cell Lines
- Mitogen-activated Protein Kinase Signaling in Inflammation-related Carcinogenesis
- Role of Mitogen Activated Protein Kinase and PI 3-kinase on Cell Proliferation in Pancreatic Cancer Cell Lines with K-ras Mutation
- The Role of CTGF in Osteosarcoma Progression
- Effects of the Peroxisome Proliferator-Activated Receptor Ligand Troglitazone in Osteosarcoma Cell Lines