J Korean Rheum Assoc.
2009 Sep;16(3):213-222.
IL-10 Inihibits VEGF Production in the Synovial Fibroblasts of RA Patients via Down-regulation of the ERK and AP-1 Pathways
- Affiliations
-
- 1Rheumatism Research Center, Catholic Institutes of Medical Science, The Catholic University of Korea, College of Medicine, Seoul, Korea. iammila@catholic.ac.kr
Abstract
OBJECTIVE
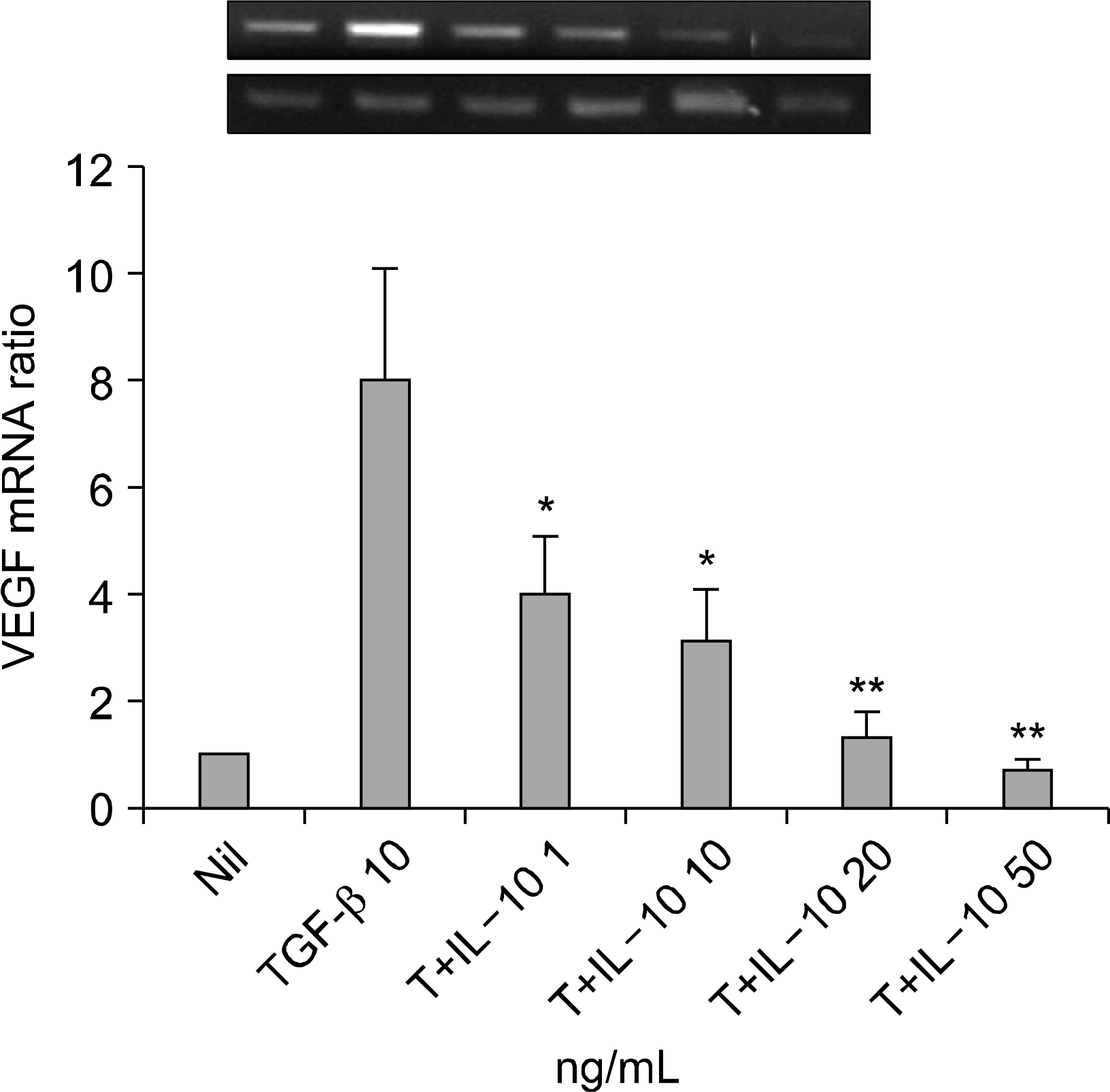
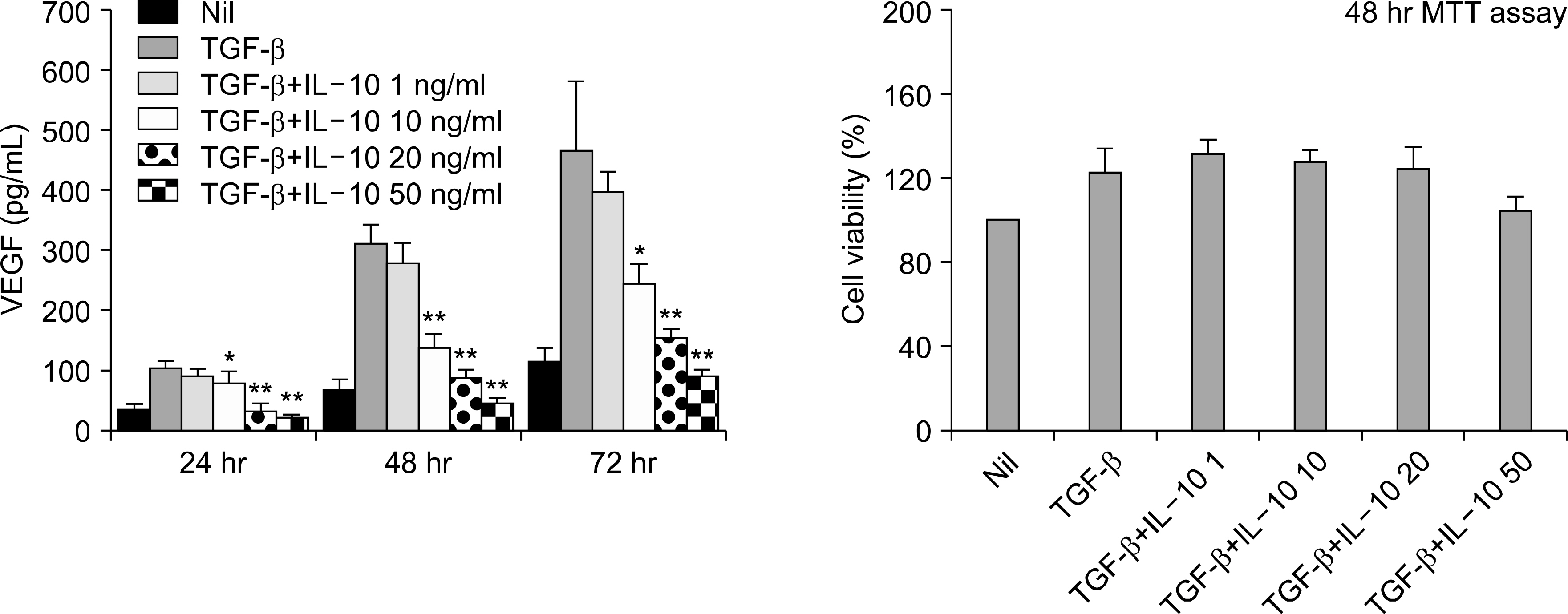
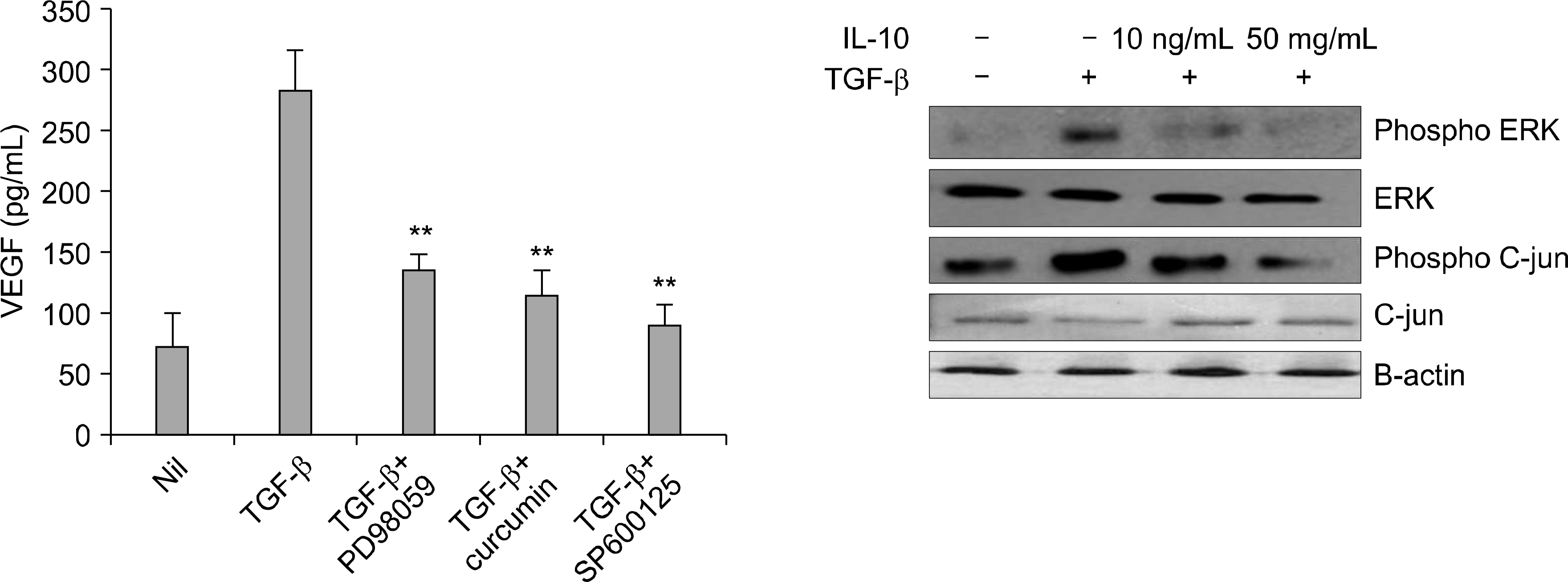
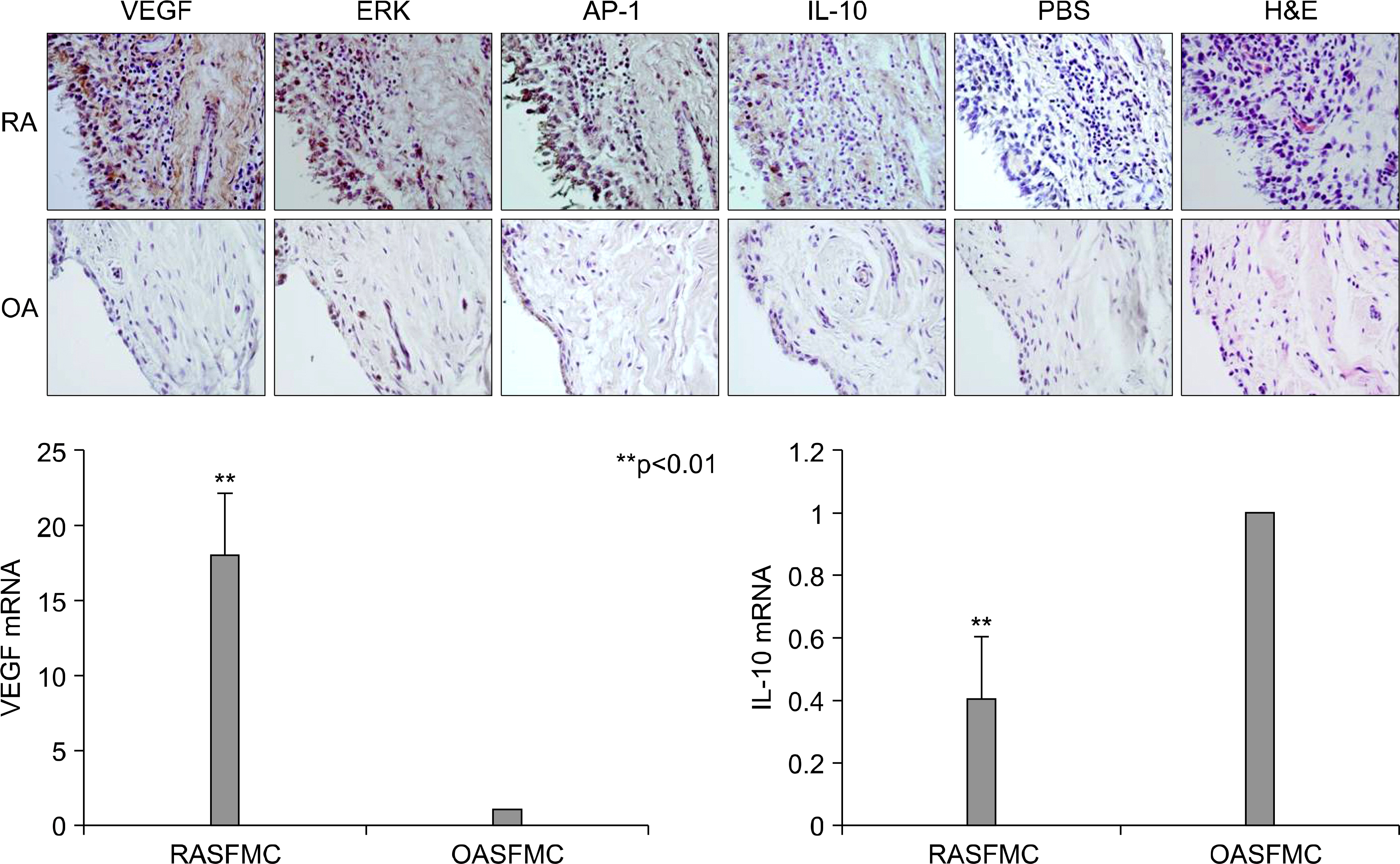
Interleukin (IL)-10 has been demonstrated to have anti-inflammatory and anti-tumour activity. Because aberrant angiogenesis is a significant pathogenic component of tumor growth and chronic inflammation, we investigated the effect of IL-10 on the production of vascular endothelial growth factor (VEGF) by the synovial fibroblasts derived from the patients with rheumatoid arthritis (RA). METHODS: Fibroblast-like synoviocytes (FLS) were cultured with transforming growth factor (TGF-beta) alone or with IL-10. The level of VEGF was measured by RT-PCR and enzyme-linked immunosorbent assay (using the 24, 48 and 72 h culture supernatants). The FLSs were cultured with TGF-b for 48 hr in the presence of PD98059 (an ERK inhibitor), curcumin and SP600125 (a JNK and Ap-1 inhibitor, respectively). The level of VEGF in the supernatants was measured by ELISA. Cell viability was assessed using MTT assay. The expressions of VEGF, ERK, AP-1 and IL-10 in the synovial tissue were quantified by immunohistochemistry. RESULTS: IL-10 exhibited the inhibitory effect on VEGF production when the FLSs were stimulated with TGF-beta. ERK and AP-1 inhibitors inhibited the TGF-beta induced VEGF production. Moreover, TGF-beta increased the phosphorylation of ERK and C-Jun, which was significantly inhibited by the IL-10. CONCLUSION: IL-10 may exert an antiangiogenic effect by inhibiting the ERK- and AP-1 mediated VEGF expression in rheumatoid synovial fibroblasts.
Keyword
MeSH Terms
-
Anthracenes
Arthritis, Rheumatoid
Cell Survival
Curcumin
Down-Regulation
Enzyme-Linked Immunosorbent Assay
Fibroblasts
Flavonoids
Humans
Immunohistochemistry
Inflammation
Interleukin-10
Interleukins
Phosphorylation
Transcription Factor AP-1
Transforming Growth Factor beta
Transforming Growth Factors
Vascular Endothelial Growth Factor A
Anthracenes
Curcumin
Flavonoids
Interleukin-10
Interleukins
Transcription Factor AP-1
Transforming Growth Factor beta
Transforming Growth Factors
Vascular Endothelial Growth Factor A
Figure
Reference
-
References
1. Koch A. Angiogenesis: implications for rheumatoid arthritis. Arthritis Rheum. 1998; 41:951–62.
Article2. Harris ED Jr. Recent insights into the pathogenesis of the proliferative lesion in rheumatoid arthritis. Arthritis Rheum. 1976; 19:68–72.
Article3. Walsh DA. Angiogenesis and arthritis. Rheumatology (Oxford). 1999; 38:103–12.
Article4. Hong KH, Cho ML, Min SY, Shin YJ, Yoo SA, Choi JJ, et al. Effect of interleukin-4 on vascular endothelial growth factor production in rheumatoid synovial fibroblasts. Clin Exp Immunol. 2007; 147:573–9.
Article5. Lee SS, Joo YS, Kim WU, Min DJ, Min JK, Park SH, et al. Vascular endothelial growth factor levels in the serum and synovial fluid of patients with rheumatoid arthritis. Clin Exp Rheumatol. 2001; 19:321–4.6. Maeno N, Takei S, Imanaka H, Takasaki I, Kitajima I, Maruyama I, et al. Increased circulating vascular endothelial growth factor is correlated with disease activity in polyarticular juvenile rheumatoid arthritis. J Rheumatol. 1999; 26:2244–8.7. Jackson JR, Minton JAL, Ho ML, Wei N, Winkler JD. Expression of vascular endothelial growth factor in synovial fibroblasts is induced by hypoxia and interleukin-1β. J Rheumatol. 1997; 24:1253–9.8. Berse B, Hunt JA, Diegel RJ, Morganelli P, Yeo K, Brown F, et al. Hypoxia augments cytokine [transforming growth factor-beta (TGF-beta) and interleukin (IL)-1]-induced vascular endothelial growth factor secretion by human synovial fibroblasts. Clin Exp Immunol. 1999; 115:176–82.9. Marumo T, Schini-Kerth VB, Busse R. Vascular endothelial growth factor activates nuclear factor-κB and induces monocyte chemoattractant protein-1 in bovine retinal endothelial cells. Diabetes. 1999; 48:1131–7.10. Lee TH, Avraham H, Lee SH, Avraham S. Vascular endothelial growth factor modulates neutrophil transendothelial migration via upregulation of interleukin-8 in human brain microvascular endothelial cells. J Biol Chem. 2002; 277:10445–51.
Article11. Sone H, Kawakami Y, Sakauchi M, Nakamura Y, Takahashi H, Shimano H, et al. Neutralization of vascular endothelial growth factor prevents collageninduced arthritis and ameliorates established disease in mice. Biochem Biophys Res Commun. 2001; 281:562–8.
Article12. Huang S, Ullrich SE, Bar-Eli M. Regulation of tumor growth and metastasis by interleukin-10: the melanoma experience. J Interferon Cytokine Res. 1999; 19:697–703.
Article13. Silvestre JS, Mallat Z, Duriez M, Tamarat R, Bureau MF, Scherman D, et al. Antiangiogenic effect of interleukin-10 in ischemia-induced angiogenesis in mice hindlimb. Circ Res. 2000; 87:448–52.
Article14. Cervenak L, Morbidelli L, Donati D, Donnini S, Kambayashi T, Wilson JL, et al. Abolished angio-genicity and tumorigenicity of Burkitt lymphoma by interleukin-10. Blood. 2000; 96:2568–73.
Article15. Finlay GA, Thannickal VJ, Fanburg BL, Paulson KE. Transforming growth factor beta1 induced activation of the ERK pathway and AP 1 in human lung fibroblasts requires the autocrine induction of basic fibroblast growth factor. J Biol Chem. 2000; 8:27650–6.16. Firestein GS, Zvaifler NJ. How important are T cells in chronic rheumatoid synovitis: T cell-independent mechanisms from beginning to end. Arthritis Rheum. 2002; 46:298–308.17. Dvorak HF, Brown LF, Detmar M, Dvorak AM. Vascular permeability factor/vascular endothelial growth factor, microvascular hyperpemeability and angiogenesis. Am J Pathol. 1995; 146:1029–35.18. Senger DR, Galli SJ, Dvorak AM, Perruzzi CA, Harvey VS, Dvorak HF. Tumor cells secrete a vascular permeability factor that promotes accumulation of ascitic fluid. Science. 1983; 219:983–96.19. Fava RA, Olsen NJ, Spencer-Green G, Yeo KT, Yeo TK, Berse B, et al. Vascular permeability factor/endothelial growth factor (VPF/VEGF): accumulation and expression in human synovial fluids and rheumatoid synovial tissue. J Immunol. 1994; 180:341–7.
Article20. Nagashima M, Yoshino S, Ishiwata T, Asano G. Role of vascular endothelial growth factor in angiogenesis of rheumatoid arthrits. J Rheumatol. 1995; 22:1624–30.21. Sakuma M, Hatsushika K, Koyama K, Katoh R, Ando T, Watanabe Y, et al. TGF-beta type I receptor kinase inhibitor downregulates rheumatoid synoviocytes and prevents the arthritis induced by type II collagen antibody. Int Immunol. 2007; 19:117–26.22. Pajkrt D, Camoglio L, Tiel-van Buul MC, de Bruin K, Cutler DL, Affrime MB, et al. Attenuation of proinflammatory response by recombinant IL-10 in human endotoxemia: effect of timing of recombinant human IL-10 administration. J Immunol. 1997; 158:3971–7.23. Di Carlo E, Coletti A, Modesti A, Giovarelli M, Forni G, Musiani P. Local release of interleukin-10 by transfected mouse adenocarcinoma cells exhibit pro-and antiinflammatory activity and results in a delayed tumor rejection. Eur Cytokine Netw. 1998; 9:61–8.24. Cho ML, Cho CS, Min SY, Kim SH, Lee SS, Kim WU, et al. Cyclosporine inhibition of vascular endothelial growth factor production in rheumatoid synovial fibroblasts. Arthritis Rheum. 2002; 46:1202–9.
Article25. Wang CR, Shiau AL, Chen SY, Lin LL, Tai MH, Shieh GS, et al. Amelioration of collageninduced arthritis in rats by adenovirus-mediated PTEN gene transfer. Arthritis Rheum. 2008; 58:1650–6.
Article26. Wang CH, Chen LN, Zhu P, Fan CM, Wang YH, Jia JF. CD147 stimulates the angiogenesis in rheumatoid synovium via the activation of vascular endothelial growth factor. J Mol Cell Immunol. 2007; 23:426–8.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- IL-15 Induced an Increased SDF-1 Expression in the Synovial Fibroblasts of Patients with Rheumatoid Arthritis
- Engagement of Toll-Like Receptor 3 Induces Vascular Endothelial Growth Factor and Interleukin-8 in Human Rheumatoid Synovial Fibroblasts
- Hypoxia/reoxygenation Regulates Expression of Tumor Suppressor PTEN in Rheumatoid Synovial Fibroblasts
- Regulation of Interleukin-17 Production in Patients with Rheumatoid Arthritis by Phosphoinositide 3-kinase (PI3K)/ Akt and Nuclear Factor KappaB (NF-kappaB) Dependent Signal Transduction Pathway
- The Th17 and Autoimmune Arthritis