J Korean Surg Soc.
2012 May;82(5):271-280.
Gene expression profiling of papillary thyroid carcinomas in Korean patients by oligonucleotide microarrays
- Affiliations
-
- 1Center for Thyroid Cancer, Research Institute and Hospital, National Cancer Center, Goyang, Korea.
- 2Division of Endocrinology and Metabolism, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea. swkimmd@skku.edu
Abstract
- PURPOSE
The incidence of papillary thyroid carcinomas (PTCs) is rapidly increasing in Korea. Analyzing the gene expression profiling (GEP) of PTCs will facilitate the advent of new methods in diagnosis, prognostication, and treatment. We performed this study to find the GEP of Korean PTCs.
METHODS
We performed oligonucleotide microarray analysis with 19 PTCs and 7 normal thyroid glands. Differentially expressed genes were selected using a t-test (|fold| >3) and adjusted Benjamini-Hochberg false discovery rate P-value < 0.01. Quantitative reverse transcription-polymerase chain reaction (QRT-PCR) was used to validate microarray data. A classification model was developed by support vector machine (SVM) algorithm to diagnose PTCs based on molecular signatures.
RESULTS
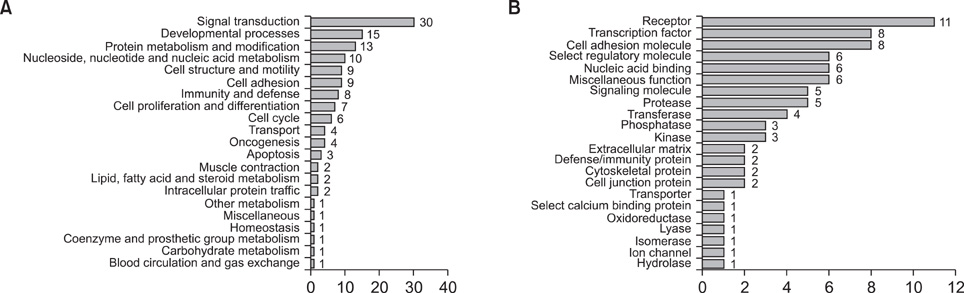
We identified 79 differentially expressed genes (70 up-regulated and 9 down-regulated) according to the criteria. QRT-PCR for five genes (CDH3, NGEF, PROS1, TGFA, MET) was confirmatory of the microarray data. Hierarchical cluster analysis and a classification model by the SVM algorithm accurately differentiated PTCs from normal thyroid gland based on GEP.
CONCLUSION
A disease classification model showed excellent accuracy in diagnosing PTCs, thus showing the possibility of molecular diagnosis in the future. This GEP could serve as baseline data for further investigation in the management of PTCs based on molecular signatures.
MeSH Terms
Figure
Reference
-
1. Ministry of Health & Welfare. National Cancer Center. Cancer facts and figures 2011 in the Republic Korea. 2011. Goyang: National Cancer Center.2. Yu GP, Li JC, Branovan D, McCormick S, Schantz SP. Thyroid cancer incidence and survival in the national cancer institute surveillance, epidemiology, and end results race/ethnicity groups. Thyroid. 2010. 20:465–473.3. Davies L, Welch HG. Increasing incidence of thyroid cancer in the United States, 1973-2002. JAMA. 2006. 295:2164–2167.4. Nishiyama RH. Clark OH, Duh QY, Kebebew E, editors. Pathology of tumors of the thyroid gland. Textbook of endocrine surgery. 2005. 2nd ed. Philadelphia: Elsevire Saunders;223–247.5. Paik S, Shak S, Tang G, Kim C, Baker J, Cronin M, et al. A multigene assay to predict recurrence of tamoxifen-treated, node-negative breast cancer. N Engl J Med. 2004. 351:2817–2826.6. Reiner A, Yekutieli D, Benjamini Y. Identifying differentially expressed genes using false discovery rate controlling procedures. Bioinformatics. 2003. 19:368–375.7. Wasenius VM, Hemmer S, Kettunen E, Knuutila S, Franssila K, Joensuu H. Hepatocyte growth factor receptor, matrix metalloproteinase-11, tissue inhibitor of metalloproteinase-1, and fibronectin are up-regulated in papillary thyroid carcinoma: a cDNA and tissue microarray study. Clin Cancer Res. 2003. 9:68–75.8. Huang Y, Prasad M, Lemon WJ, Hampel H, Wright FA, Kornacker K, et al. Gene expression in papillary thyroid carcinoma reveals highly consistent profiles. Proc Natl Acad Sci U S A. 2001. 98:15044–15049.9. Aldred MA, Huang Y, Liyanarachchi S, Pellegata NS, Gimm O, Jhiang S, et al. Papillary and follicular thyroid carcinomas show distinctly different microarray expression profiles and can be distinguished by a minimum of five genes. J Clin Oncol. 2004. 22:3531–3539.10. Carpi A, Mechanick JI, Saussez S, Nicolini A. Thyroid tumor marker genomics and proteomics: diagnostic and clinical implications. J Cell Physiol. 2010. 224:612–619.11. Rhodes DR, Yu J, Shanker K, Deshpande N, Varambally R, Ghosh D, et al. Large-scale meta-analysis of cancer microarray data identifies common transcriptional profiles of neoplastic transformation and progression. Proc Natl Acad Sci U S A. 2004. 101:9309–9314.12. Griffith OL, Melck A, Jones SJ, Wiseman SM. Meta-analysis and meta-review of thyroid cancer gene expression profiling studies identifies important diagnostic biomarkers. J Clin Oncol. 2006. 24:5043–5051.13. Kim HS, Kim DH, Kim JY, Jeoung NH, Lee IK, Bong JG, et al. Microarray analysis of papillary thyroid cancers in Korean. Korean J Intern Med. 2010. 25:399–407.14. Giordano S, Di Renzo MF, Narsimhan RP, Cooper CS, Rosa C, Comoglio PM. Biosynthesis of the protein encoded by the c-met proto-oncogene. Oncogene. 1989. 4:1383–1388.15. Mineo R, Costantino A, Frasca F, Sciacca L, Russo S, Vigneri R, et al. Activation of the hepatocyte growth factor (HGF)-Met system in papillary thyroid cancer: biological effects of HGF in thyroid cancer cells depend on Met expression levels. Endocrinology. 2004. 145:4355–4365.16. Benjamini Y, Lipkind D, Horev G, Fonio E, Kafkafi N, Golani I. Ten ways to improve the quality of descriptions of whole-animal movement. Neurosci Biobehav Rev. 2010. 34:1351–1365.17. Bergstrom JD, Westermark B, Heldin NE. Epidermal growth factor receptor signaling activates met in human anaplastic thyroid carcinoma cells. Exp Cell Res. 2000. 259:293–299.18. Benjamini Y, Yekutieli D. Quantitative trait Loci analysis using the false discovery rate. Genetics. 2005. 171:783–790.19. Thomassen M, Tan Q, Kruse TA. Gene expression meta-analysis identifies chromosomal regions and candidate genes involved in breast cancer metastasis. Breast Cancer Res Treat. 2009. 113:239–249.20. Carvallo L, Munoz R, Bustos F, Escobedo N, Carrasco H, Olivares G, et al. Non-canonical Wnt signaling induces ubiquitination and degradation of Syndecan4. J Biol Chem. 2010. 285:29546–29555.21. Bashyam MD, Bair R, Kim YH, Wang P, Hernandez-Boussard T, Karikari CA, et al. Array-based comparative genomic hybridization identifies localized DNA amplifications and homozygous deletions in pancreatic cancer. Neoplasia. 2005. 7:556–562.22. Gorres KL, Raines RT. Prolyl 4-hydroxylase. Crit Rev Biochem Mol Biol. 2010. 45:106–124.23. Teodoro JG, Parker AE, Zhu X, Green MR. p53-mediated inhibition of angiogenesis through up-regulation of a collagen prolyl hydroxylase. Science. 2006. 313:968–971.24. Conacci-Sorrell M, Zhurinsky J, Ben-Ze'ev A. The cadherin-catenin adhesion system in signaling and cancer. J Clin Invest. 2002. 109:987–991.25. Turashvili G, McKinney SE, Goktepe O, Leung SC, Huntsman DG, Gelmon KA, et al. P-cadherin expression as a prognostic biomarker in a 3992 case tissue microarray series of breast cancer. Mod Pathol. 2011. 24:64–81.26. Zhang CC, Yan Z, Zhang Q, Kuszpit K, Zasadny K, Qiu M, et al. PF-03732010: a fully human monoclonal antibody against P-cadherin with antitumor and antimetastatic activity. Clin Cancer Res. 2010. 16:5177–5188.27. Rodrigues NR, Theodosiou AM, Nesbit MA, Campbell L, Tandle AT, Saranath D, et al. Characterization of Ngef, a novel member of the Dbl family of genes expressed predominantly in the caudate nucleus. Genomics. 2000. 65:53–61.28. Yukinawa N, Oba S, Kato K, Taniguchi K, Iwao-Koizumi K, Tamaki Y, et al. A multi-class predictor based on a probabilistic model: application to gene expression profiling-based diagnosis of thyroid tumors. BMC Genomics. 2006. 7:190.29. Krause K, Eszlinger M, Gimm O, Karger S, Engelhardt C, Dralle H, et al. TFF3-based candidate gene discrimination of benign and malignant thyroid tumors in a region with borderline iodine deficiency. J Clin Endocrinol Metab. 2008. 93:1390–1393.30. Nikolova DN, Zembutsu H, Sechanov T, Vidinov K, Kee LS, Ivanova R, et al. Genome-wide gene expression profiles of thyroid carcinoma: identification of molecular targets for treatment of thyroid carcinoma. Oncol Rep. 2008. 20:105–121.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Expression of p53 and MDM2 Proteins in Thyroid Carcinomas
- Thyroid Tumorigenesis
- Microarray Applications in Cancer Research
- Comparison of oligonucleotide-microarray and serial analysis of gene expression (SAGE) in transcript profiling analysis of megakaryocytes derived from CD34+ cells
- Expression of Down Stream Molecules of RET (p-ERK, p-p38 MAPK, p-JNK and p-AKT) in Papillary Thyroid Carcinomas