J Korean Acad Periodontol.
2009 Aug;39(Suppl):213-222.
The Effects of Hydroxyapatite-Chitosan Membrane on Bone Regeneration in Rat Calvarial Defects
- Affiliations
-
- 1Department of Periodontology, Research Institute for Periodontal Regeneration, Yonsei University, College of Dentistry, Korea. shchoi726@yuhs.ac
- 2Department and Research Institute of Dental Biomaterials and Bioengineering, Yonsei University, College of Dentistry, Korea.
Abstract
-
PURPOSE: Recently, interest in chitosan has increased due to its excellent biological properties such as biocompatibility, antibacterial effect, and rapid healing capacity. On the other hand, hydroxyapatite is used as a bone substitute in the fields of orthopedics and dentistry. The hydroxyapatite-chitosan (HA-CS) complex containing hydroxyapatite nanoparticles was developed for synergy of both biomaterials. The objective of this study was to evaluate the effect of hydroxyapatite (HA)-chitosan (CS) membrane on bone regeneration in the rat calvarial defect.
METHODS
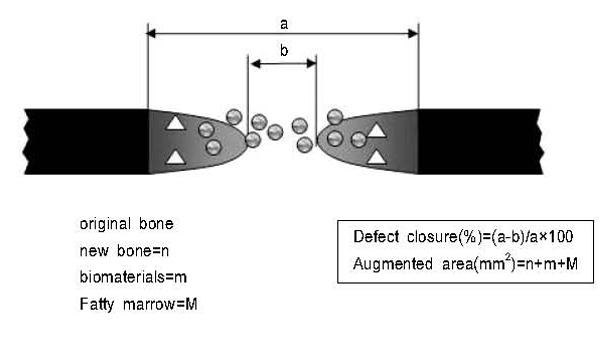
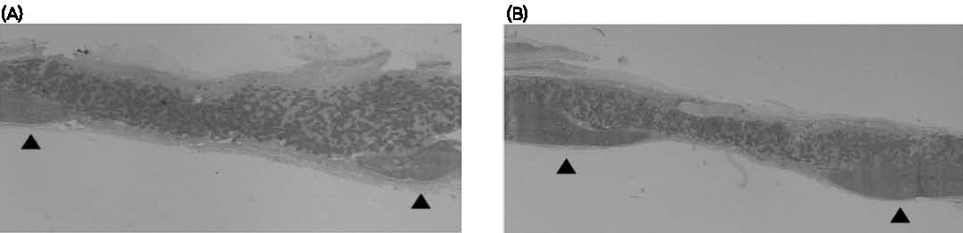
Eight-millimeter critical-sized calvarial defects were created in 70 male Sprague-Dawley rats. The animals were divided into 7 groups of 10 animals and received either 1) chitosan (CS) 100% membrane, 2) hydroxyapatite (HA) 30%/ CS 70% membrane, 3) HA 30% / CS 70%, pressed membrane, 4) HA 40%/ CS 60% membrane, 5) HA 50%/ CS 50% membrane, 6) HA 50%/ CS 50%, pressed membrane, or 7) a sham - surgery control. The amount of newly formed bone from the surface of the rat calvarial defects was measured using histomorphometry, following 2- or 8- week healing intervals.
RESULTS
Surgical implantation of the HA - CS membrane resulted in enhanced local bone formation at both 2 and 8 weeks compared to the control group. The HA - CS membrane would be significantly more effective than the chitosan membrane in early bone formation.
CONCLUSIONS
Concerning the advantages of biomaterials, the HA-CS membrane would be an effective biomaterial for regeneration of periodontal bone. Further studies will be required to improve the mechanical properties to develop a more rigid scaffold for the HA-CS membrane.
Keyword
MeSH Terms
Figure
Reference
-
1. Magnusson I, Batich C, Collins BR. New attachment formation following controlled tissue regeneration using biodegradable membranes. J Periodontol. 1998. 59:1–6.
Article2. Aquirre-Zorzano LA, Estefania-Cundin E, GilLozano J. Periodontal regeneration of infrabony defectsusing resorbable membrane: determinant of the healing response. An observational clinical study. Int J Periodontics Rest Dent. 1999. 19:363–371.3. Zitzmann NU, Naef R, Schaurer P. Resorbable versus nonresorbable membranes in combination with Bio-Oss for guided bone regeneration. Int J Oral Maxillofac Implant. 1997. 12:844–852.4. Becker W, Becker BD, Mellonig J. A prospective multi-center study evaluation periodontal regeneration for class II furcation invasions and intrabony defects after treatment with a bioabsorbable barrier membrane: 1-year results. J Periodontol. 1996. 67:641–649.
Article5. Hong SB, Kwon YH, Lee MS, Herr Y. Comparative study on tissue responses of 3 resorbable membranes in rats. J Korean Acad Periodontol. 2002. 32:475–488.
Article6. Amano K, Ito E. The action of lysozyme on partially deacetylate chitin. European J Biochem. 1978. 85:97–104.7. Pangburn SH, Trescony PV, Heller J. Lysozyme degradation of partially deacetylated chitin, its films and hydrogels. Biomaterials. 1982. 3:105–108.
Article8. Reynolds BL. Wound healing III: Artificial maturation of arrested regenerate with an acetylated amino sugar. The American Surgeon. 1960. 26:113–117.9. Kim OS, Chung HJ. Effects of chitosan on human gingival fibroblasts in vitro. J Korean Acad Periodontol. 2002. 32:235–247.
Article10. Paik JW, Lee HJ, Yoo YJ. The effects of chitosan on the human periodontal ligament fibroblasts in vitro. J Korean Acad Periodontol. 2001. 31:823–832.
Article11. Park JS, Choi SH, Moon IS. Eight-week histological analysis on the effect of chitosan on surgically created one-wall intrabony defects in beagle dogs. J Clin Periodontol. 2003. 30:443–453.
Article12. Salata LA, Craig GT, Brook IM. Bone healing following the use of hydroxyapatite or ionomericbone substitutes alone or combined with a guided bone regeneration technique: an animal study. Int J Oral Maxillofac Implants. 1998. 13:44–51.13. Zhang C, Hu YY, Cui FZ, Zhang SM, Ruan DK. A study on a tissue-engineered bone using rhBMP-2 induced periosteal cells with a porous nano-hydroxyapatite/collagen/poly(L-lactic acid) scaffold. Biomed Mater. 2006. 2:56–62.
Article14. Wang HL, MacNeal RL, Shieh AT. Utilization of a resorbable collagen membrane in repairing gingival recession defects. Pract Periodont Aesthet Dent. 1996. 8:441–448.15. Wang HL, O'Neal RB, Tomas CL. Evaluation of and absorbale collagen membrane in treating class II furcation defects. J Periodontol. 1994. 65:1029–1036.
Article16. Polson AM, Garrett S, Stoller NH. Guided tissue regeneration in human furcation defects after using biodegradable barrier. : A multi-center feasibility study. J Periodontol. 1995. 66:377–385.17. Bouchard P, Giovannoli JL, Mattout C. Clinical evaluation of a bioabsorble regenerative material in mandibular class II furcation therapy. J Clin Periodontol. 1997. 24:511–518.
Article18. Jung UW, Song KY, Kim CS, Lee YK, Cho KS, Kim CK, Choi SH. Effects of a chitosan membrane coated with polylactic and polyglycolic acid on bone regeneration in a rat calvarial defect. Biomed Mater. 2007. 3:S101–S105.
Article19. Webster TJ, Ergun C, Doremus RH. Enhanced functions of osteoblasts on nanophase ceramics. Biomaterials. 2000. 21:1803–1810.
Article20. Zhang YF, Cheng XR, Chen Y. Three-dimensional nanohydroxyapatite/chitosan scaffolds as potential tissue engineered periodontal tissue. J Biomater Appl. 2007. 21:333–349.
Article21. Kong L, Gao Y, Cao W. Preparation and characterization of nano-hydroxyapatite/chitosan composite scaffolds. J Biomed Mater Res A. 2005. 75:275–282.
Article22. Wei G, Ma PX. Structure and properties of nano-hydroxyapatite/polymer composite scaffolds for bone tissue engineering. Biomaterials. 2004. 25:4749–4757.
Article23. Schmitz JP, Hollinger JO. The critical size defect as an experimental model for craniomandibulofacial nonunoins. Clin Orthop. 1986. 205:299–308.24. Caton J, Mota L, Gandini L. Non-human primate models for testing the efficacy and safety of periodontal regeneration procedures. J Periodontol. 1994. 65:1143–1150.
Article25. Selvig KA. Discussion: Animal models in reconstructive therapy. J Periodontol. 1994. 65:1169–1172.
Article26. Jung UW, Suh JJ, Choi SH. The bone regenerative effects of chitosan on the calvarial critical size defect in Sprague-Dawley rats. J Korean Acad Periodontol. 2000. 30:851–868.
Article27. Pang EK, Paik JW, Kim SK. Effects of chitosan on human periodontal ligament fibroblasts in vitro and on bone formation in rat calvarial defects. J Periodontol. 2005. 76:1526–1533.
Article28. Oonishi H, Hench LL, Wilson J. Comparative bone growth behavior in granules of bioceramic materials of various sizes. J Biomed Mater Res. 1999. 44:31–43.
Article29. Manjubala I, Sivakumar M, Sureshkumar RV. Bioactivity and osseointegration study of calcium phosphate ceramic of different chemical composition. J Biomed Mater Res. 2002. 63:200–208.
Article30. Liljensten E, Adolfsson E, Strid KG. Resorbable and nonresorbable hydroxyapatite granules as bone graft substitutes in rabbit cortical defects. Clin Implant Dent Relat Res. 2003. 5:95–101.
Article31. Andrade JC, Camilli JA, Kawachi EY. Behavior of dense and porous hydroxyapatite implants and tissue response in rat femoral defects. J Biomed Mater Res. 2002. 62:30–36.
Article32. Takeshita F, Ayukawa Y, lyama S. Histological comparison of early wound healing following dense hydroxyapatite granule grafting and barrier placement in surgically-created bone defects neighboring implants. J Periodontol. 1997. 68:924–932.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Effect On Guided Bone Regeneration Of The Chitosan Membrane
- A study on the biodegradable novel chitosan nanofiber membrane as a possible tool for guided bone regeneration
- Study of chitosan's effects on periodontal tissue regeneration: a meta-analysis of the histomorphometry
- Effect of chitosan in primary rat calvarial cell
- Guided bone regenerative effect of chitosan and chitosan-cellulose membranes