Diagnosis and Treatment of Nontuberculous Mycobacterial Lung Disease: Clinicians' Perspectives
- Affiliations
-
- 1Division of Pulmonary and Critical Care Medicine, Department of Medicine, Ewha Womans University School of Medicine, Seoul, Korea.
- 2Division of Pulmonary and Critical Care Medicine, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- 3Division of Mycobacterial and Respiratory Infections, National Jewish Health, Denver, CO, USA. daleyc@njhealth.org
- KMID: 2320769
- DOI: http://doi.org/10.4046/trd.2016.79.2.74
Abstract
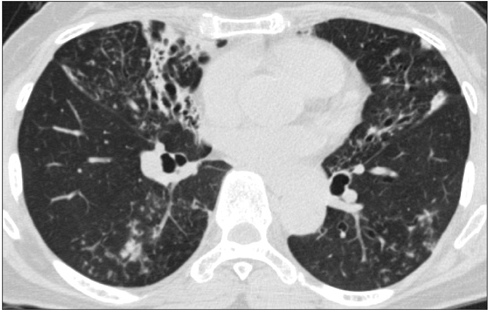
- Nontuberculous mycobacteria (NTM) are emerging pathogens that affect both immunocompromised and immunocompetent patients. The incidence and prevalence of NTM lung disease are increasing worldwide and rapidly becoming a major public health problem. For the diagnosis of NTM lung disease, patients suspected to have NTM lung disease are required to meet all clinical and microbiologic criteria. The development of molecular methods allows the characterization of new species and NTM identification at a subspecies level. Even after the identification of NTM species from respiratory specimens, clinicians should consider the clinical significance of such findings. Besides the limited options, treatment is lengthy and varies by species, and therefore a challenge. Treatment may be complicated by potential toxicity with discouraging outcomes. The decision to start treatment for NTM lung disease is not easy and requires careful individualized analysis of risks and benefits. Clinicians should be alert to those unique aspects of NTM lung disease concerning diagnosis with advanced molecular methods and treatment with limited options. Current recommendations and recent advances for diagnosis and treatment of NTM lung disease are summarized in this article.
Keyword
MeSH Terms
Figure
Cited by 6 articles
-
Changing Epidemiology of Nontuberculous Mycobacterial Lung Diseases in a Tertiary Referral Hospital in Korea between 2001 and 2015
Ryoung-Eun Ko, Seong Mi Moon, Soohyun Ahn, Byung Woo Jhun, Kyeongman Jeon, O Jung Kwon, Hee Jae Huh, Chang-Seok Ki, Nam Yong Lee, Won-Jung Koh
J Korean Med Sci. 2018;33(8):. doi: 10.3346/jkms.2018.33.e65.Is Cross-reactivity with Nontuberculous Mycobacteria a Systematic Problem in the Xpert MTB/RIF Assay?
Hee Jae Huh, Dong Joon Song, Chang-Seok Ki, Nam Yong Lee
Tuberc Respir Dis. 2019;82(1):88-89. doi: 10.4046/trd.2018.0075.Diagnosis of pulmonary tuberculosis
Byung Woo Jhun, Hee Jae Huh, Won-Jung Koh
J Korean Med Assoc. 2019;62(1):18-24. doi: 10.5124/jkma.2019.62.1.18.Down-Regulation of Serum High-Mobility Group Box 1 Protein in Patients with Pulmonary Tuberculosis and Nontuberculous Mycobacterial Lung Disease
Su-Young Kim, Won-Jung Koh, Hye Yun Park, Kyeongman Jeon, Soo-Youn Lee, Jae-Joon Yim, Sung Jae Shin
Tuberc Respir Dis. 2017;80(2):153-158. doi: 10.4046/trd.2017.80.2.153.Treatment of
Mycobacterium avium Complex Pulmonary Disease
Yong-Soo Kwon, Won-Jung Koh, Charles L. Daley
Tuberc Respir Dis. 2019;82(1):15-26. doi: 10.4046/trd.2018.0060.Infection Source and Epidemiology of Nontuberculous Mycobacterial Lung Disease
Doosoo Jeon
Tuberc Respir Dis. 2019;82(2):94-101. doi: 10.4046/trd.2018.0026.
Reference
-
1. Kendall BA, Winthrop KL. Update on the epidemiology of pulmonary nontuberculous mycobacterial infections. Semin Respir Crit Care Med. 2013; 34:87–94.2. Prevots DR, Marras TK. Epidemiology of human pulmonary infection with nontuberculous mycobacteria: a review. Clin Chest Med. 2015; 36:13–34.3. Koh WJ, Chang B, Jeong BH, Jeon K, Kim SY, Lee NY, et al. Increasing recovery of nontuberculous mycobacteria from respiratory specimens over a 10-year period in a tertiary referral hospital in South Korea. Tuberc Respir Dis. 2013; 75:199–204.4. Hoefsloot W, van Ingen J, Andrejak C, Angeby K, Bauriaud R, Bemer P, et al. The geographic diversity of nontuberculous mycobacteria isolated from pulmonary samples: an NTMNET collaborative study. Eur Respir J. 2013; 42:1604–1613.5. Griffith DE, Aksamit T, Brown-Elliott BA, Catanzaro A, Daley C, Gordin F, et al. An official ATS/IDSA statement: diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. Am J Respir Crit Care Med. 2007; 175:367–416.6. Marras TK, Prevots DR, Jamieson FB, Winthrop KL. Pulmonary MAC Outcomes Group. Opinions differ by expertise in Mycobacterium avium complex disease. Ann Am Thorac Soc. 2014; 11:17–22.7. Marras TK, Prevots DR, Jamieson FB, Winthrop KL. Pulmonary MAC Outcomes Group. Variable agreement among experts regarding Mycobacterium avium complex lung disease. Respirology. 2015; 20:348–351.8. Adjemian J, Prevots DR, Gallagher J, Heap K, Gupta R, Griffith D. Lack of adherence to evidence-based treatment guidelines for nontuberculous mycobacterial lung disease. Ann Am Thorac Soc. 2014; 11:9–16.9. van Ingen J. Microbiological diagnosis of nontuberculous mycobacterial pulmonary disease. Clin Chest Med. 2015; 36:43–54.10. Kwon YS, Koh WJ. Diagnosis of pulmonary tuberculosis and nontuberculous mycobacterial lung disease in Korea. Tuberc Respir Dis. 2014; 77:1–5.11. Koh WJ, Yu CM, Suh GY, Chung MP, Kim H, Kwon OJ, et al. Pulmonary TB and NTM lung disease: comparison of characteristics in patients with AFB smear-positive sputum. Int J Tuberc Lung Dis. 2006; 10:1001–1007.12. Kim YK, Hahn S, Uh Y, Im DJ, Lim YL, Choi HK, et al. Comparable characteristics of tuberculous and non-tuberculous mycobacterial cavitary lung diseases. Int J Tuberc Lung Dis. 2014; 18:725–729.13. Jeong YJ, Lee KS, Koh WJ, Han J, Kim TS, Kwon OJ. Nontuberculous mycobacterial pulmonary infection in immunocompetent patients: comparison of thin-section CT and histopathologic findings. Radiology. 2004; 231:880–886.14. Yuan MK, Chang CY, Tsai PH, Lee YM, Huang JW, Chang SC. Comparative chest computed tomography findings of non-tuberculous mycobacterial lung diseases and pulmonary tuberculosis in patients with acid fast bacilli smearpositive sputum. BMC Pulm Med. 2014; 14:65.15. Chu HQ, Li B, Zhao L, Huang DD, Zhang ZM, Xu JF, et al. Chest imaging comparison between non-tuberculous and tuberculosis mycobacteria in sputum acid fast bacilli smearpositive patients. Eur Rev Med Pharmacol Sci. 2015; 19:2429–2439.16. Koh WJ, Lee KS, Kwon OJ, Jeong YJ, Kwak SH, Kim TS. Bilateral bronchiectasis and bronchiolitis at thin-section CT: diagnostic implications in nontuberculous mycobacterial pulmonary infection. Radiology. 2005; 235:282–288.17. Lee G, Lee KS, Moon JW, Koh WJ, Jeong BH, Jeong YJ, et al. Nodular bronchiectatic Mycobacterium avium complex pulmonary disease: natural course on serial computed tomographic scans. Ann Am Thorac Soc. 2013; 10:299–306.18. Kim RD, Greenberg DE, Ehrmantraut ME, Guide SV, Ding L, Shea Y, et al. Pulmonary nontuberculous mycobacterial disease: prospective study of a distinct preexisting syndrome. Am J Respir Crit Care Med. 2008; 178:1066–1074.19. Kartalija M, Ovrutsky AR, Bryan CL, Pott GB, Fantuzzi G, Thomas J, et al. Patients with nontuberculous mycobacterial lung disease exhibit unique body and immune phenotypes. Am J Respir Crit Care Med. 2013; 187:197–205.20. Park IK, Olivier KN. Nontuberculous mycobacteria in cystic fibrosis and non-cystic fibrosis bronchiectasis. Semin Respir Crit Care Med. 2015; 36:217–224.21. Chu H, Zhao L, Xiao H, Zhang Z, Zhang J, Gui T, et al. Prevalence of nontuberculous mycobacteria in patients with bronchiectasis: a meta-analysis. Arch Med Sci. 2014; 10:661–668.22. Hollings NP, Wells AU, Wilson R, Hansell DM. Comparative appearances of non-tuberculous mycobacteria species: a CT study. Eur Radiol. 2002; 12:2211–2217.23. Chung MJ, Lee KS, Koh WJ, Lee JH, Kim TS, Kwon OJ, et al. Thin-section CT findings of nontuberculous mycobacterial pulmonary diseases: comparison between Mycobacterium avium-intracellulare complex and Mycobacterium abscessus infection. J Korean Med Sci. 2005; 20:777–783.24. van Ingen J. Diagnosis of nontuberculous mycobacterial infections. Semin Respir Crit Care Med. 2013; 34:103–109.25. Pfyffer GE. Mycobacterium: general characteristics, laboratory detection, and staining procedures. In : Jorgensen JH, Pfaller MA, Carroll KC, Funke G, Landry ML, Richter SS, editors. Manual of clinical microbiology. 11th ed. Washington,DC: ASM Press;2015. p. 543–572.26. Somoskövi A, Hotaling JE, Fitzgerald M, O'Donnell D, Parsons LM, Salfinger M. Lessons from a proficiency testing event for acid-fast microscopy. Chest. 2001; 120:250–257.27. Huh HJ, Koh WJ, Song DJ, Ki CS, Lee NY. Evaluation of the Cobas TaqMan MTB test for the detection of Mycobacterium tuberculosis complex according to acid-fast-bacillus smear grades in respiratory specimens. J Clin Microbiol. 2015; 53:696–698.28. Guglielmetti L, Mougari F, Lopes A, Raskine L, Cambau E. Human infections due to nontuberculous mycobacteria: the infectious diseases and clinical microbiology specialists' point of view. Future Microbiol. 2015; 10:1467–1483.29. Somoskovi A, Salfinger M. Nontuberculous mycobacteria in respiratory infections: advances in diagnosis and identification. Clin Lab Med. 2014; 34:271–295.30. Macheras E, Roux AL, Ripoll F, Sivadon-Tardy V, Gutierrez C, Gaillard JL, et al. Inaccuracy of single-target sequencing for discriminating species of the Mycobacterium abscessus group. J Clin Microbiol. 2009; 47:2596–2600.31. Frothingham R, Wilson KH. Sequence-based differentiation of strains in the Mycobacterium avium complex. J Bacteriol. 1993; 175:2818–2825.32. Ben Salah I, Adekambi T, Raoult D, Drancourt M. rpoB sequence-based identification of Mycobacterium avium complex species. Microbiology. 2008; 154(Pt 12):3715–3723.33. Zelazny AM, Root JM, Shea YR, Colombo RE, Shamputa IC, Stock F, et al. Cohort study of molecular identification and typing of Mycobacterium abscessus, Mycobacterium massiliense, and Mycobacterium bolletii. J Clin Microbiol. 2009; 47:1985–1995.34. Macheras E, Roux AL, Bastian S, Leao SC, Palaci M, Sivadon-Tardy V, et al. Multilocus sequence analysis and rpoB sequencing of Mycobacterium abscessus (sensu lato) strains. J Clin Microbiol. 2011; 49:491–499.35. Jang MA, Koh WJ, Huh HJ, Kim SY, Jeon K, Ki CS, et al. Distribution of nontuberculous mycobacteria by multigene sequence-based typing and clinical significance of isolated strains. J Clin Microbiol. 2014; 52:1207–1212.36. Koh WJ, Stout JE, Yew WW. Advances in the management of pulmonary disease due to Mycobacterium abscessus complex. Int J Tuberc Lung Dis. 2014; 18:1141–1148.37. Lee MR, Sheng WH, Hung CC, Yu CJ, Lee LN, Hsueh PR. Mycobacterium abscessus complex infections in humans. Emerg Infect Dis. 2015; 21:1638–1646.38. Cho YJ, Yi H, Chun J, Cho SN, Daley CL, Koh WJ, et al. The genome sequence of 'Mycobacterium massiliense' strain CIP 108297 suggests the independent taxonomic status of the Mycobacterium abscessus complex at the subspecies level. PLoS One. 2013; 8:e81560.39. Sassi M, Drancourt M. Genome analysis reveals three genomospecies in Mycobacterium abscessus. BMC Genomics. 2014; 15:359.40. Nash KA, Brown-Elliott BA, Wallace RJ Jr. A novel gene, erm(41), confers inducible macrolide resistance to clinical isolates of Mycobacterium abscessus but is absent from Mycobacterium chelonae. Antimicrob Agents Chemother. 2009; 53:1367–1376.41. Bastian S, Veziris N, Roux AL, Brossier F, Gaillard JL, Jarlier V, et al. Assessment of clarithromycin susceptibility in strains belonging to the Mycobacterium abscessus group by erm(41) and rrl sequencing. Antimicrob Agents Chemother. 2011; 55:775–781.42. Brown-Elliott BA, Vasireddy S, Vasireddy R, Iakhiaeva E, Howard ST, Nash K, et al. Utility of sequencing the erm(41) gene in isolates of Mycobacterium abscessus subsp. abscessus with low and intermediate clarithromycin MICs. J Clin Microbiol. 2015; 53:1211–1215.43. Choi GE, Shin SJ, Won CJ, Min KN, Oh T, Hahn MY, et al. Macrolide treatment for Mycobacterium abscessus and Mycobacterium massiliense infection and inducible resistance. Am J Respir Crit Care Med. 2012; 186:917–925.44. Seng P, Rolain JM, Fournier PE, La Scola B, Drancourt M, Raoult D. MALDI-TOF-mass spectrometry applications in clinical microbiology. Future Microbiol. 2010; 5:1733–1754.45. Buckwalter SP, Olson SL, Connelly BJ, Lucas BC, Rodning AA, Walchak RC, et al. Evaluation of matrix-assisted laser desorption ionization-time of flight mass spectrometry for identification of Mycobacterium species, nocardia species, and other aerobic actinomycetes. J Clin Microbiol. 2016; 54:376–384.46. Kodana M, Tarumoto N, Kawamura T, Saito T, Ohno H, Maesaki S, et al. Utility of the MALDI-TOF MS method to identify nontuberculous mycobacteria. J Infect Chemother. 2016; 22:32–35.47. Mediavilla-Gradolph MC, De Toro-Peinado I, Bermudez-Ruiz MP, Garcia-Martinez Mde L, Ortega-Torres M, Montiel Quezel-Guerraz N, et al. Use of MALDI-TOF MS for identification of nontuberculous Mycobacterium species isolated from clinical specimens. Biomed Res Int. 2015; 2015:854078.48. van Ingen J, Kuijper EJ. Drug susceptibility testing of nontuberculous mycobacteria. Future Microbiol. 2014; 9:1095–1110.49. Griffith DE, Brown-Elliott BA, Langsjoen B, Zhang Y, Pan X, Girard W, et al. Clinical and molecular analysis of macrolide resistance in Mycobacterium avium complex lung disease. Am J Respir Crit Care Med. 2006; 174:928–934.50. Clinical Laboratory Standards Institute. Susceptibility testing of mycobacteria, nocardiae, and other aerobic actinomycetes: approved standard. 2nd ed. Wayne: Clinical Laboratory Standards Institute;2011. CLSI No.M24-A2.51. Czaja CA, Levin AR, Cox CW, Vargas D, Daley CL, Cott GR. Improvement in quality of life after therapy for Mycobacterium abscessus group lung infection: a prospective cohort study. Ann Am Thorac Soc. 2016; 13:40–48.52. Egelund EF, Fennelly KP, Peloquin CA. Medications and monitoring in nontuberculous mycobacteria infections. Clin Chest Med. 2015; 36:55–66.53. Wallace RJ Jr, Brown-Elliott BA, McNulty S, Philley JV, Killingley J, Wilson RW, et al. Macrolide/azalide therapy for nodular/bronchiectatic Mycobacterium avium complex lung disease. Chest. 2014; 146:276–282.54. Jeong BH, Jeon K, Park HY, Kim SY, Lee KS, Huh HJ, et al. Intermittent antibiotic therapy for nodular bronchiectatic Mycobacterium avium complex lung disease. Am J Respir Crit Care Med. 2015; 191:96–103.55. Koh WJ, Hong G, Kim SY, Jeong BH, Park HY, Jeon K, et al. Treatment of refractory Mycobacterium avium complex lung disease with a moxifloxacin-containing regimen. Antimicrob Agents Chemother. 2013; 57:2281–2285.56. Jo KW, Kim S, Lee JY, Lee SD, Kim WS, Kim DS, et al. Treatment outcomes of refractory MAC pulmonary disease treated with drugs with unclear efficacy. J Infect Chemother. 2014; 20:602–606.57. Olivier KN, Shaw PA, Glaser TS, Bhattacharyya D, Fleshner M, Brewer CC, et al. Inhaled amikacin for treatment of refractory pulmonary nontuberculous mycobacterial disease. Ann Am Thorac Soc. 2014; 11:30–35.58. van Ingen J, Totten SE, Helstrom NK, Heifets LB, Boeree MJ, Daley CL. In vitro synergy between clofazimine and amikacin in treatment of nontuberculous mycobacterial disease. Antimicrob Agents Chemother. 2012; 56:6324–6327.59. Ferro BE, Meletiadis J, Wattenberg M, de Jong A, van Soolingen D, Mouton JW, et al. Clofazimine prevents the regrowth of Mycobacterium abscessus and Mycobacterium avium type strains exposed to amikacin and clarithromycin. Antimicrob Agents Chemother. 2015; 60:1097–1105.60. Jarand J, Davis JP, Cowie RL, Field SK, Fisher DA. Long term follow up of Mycobacterium avium complex lung disease in patients treated with regimens including clofazimine and/or rifampin. Chest. 2015; 10. 29. [Epub]. DOI: 10.1378/chest.15-0543.61. Santin M, Dorca J, Alcaide F, Gonzalez L, Casas S, Lopez M, et al. Long-term relapses after 12-month treatment for Mycobacterium kansasii lung disease. Eur Respir J. 2009; 33:148–152.62. Shitrit D, Baum GL, Priess R, Lavy A, Shitrit AB, Raz M, et al. Pulmonary Mycobacterium kansasii infection in Israel, 1999-2004: clinical features, drug susceptibility, and outcome. Chest. 2006; 129:771–776.63. Moon SM, Park HY, Jeon K, Kim SY, Chung MJ, Huh HJ, et al. Clinical significance of Mycobacterium kansasii isolates from respiratory specimens. PLoS One. 2015; 10:e0139621.64. Philley JV, Griffith DE. Treatment of slowly growing mycobacteria. Clin Chest Med. 2015; 36:79–90.65. Kasperbauer SH, De Groote MA. The treatment of rapidly growing mycobacterial infections. Clin Chest Med. 2015; 36:67–78.66. Nie W, Duan H, Huang H, Lu Y, Bi D, Chu N. Species identification of Mycobacterium abscessus subsp. abscessus and Mycobacterium abscessus subsp. bolletii using rpoB and hsp65, and susceptibility testing to eight antibiotics. Int J Infect Dis. 2014; 25:170–174.67. Kim SY, Kim CK, Bae IK, Jeong SH, Yim JJ, Jung JY, et al. The drug susceptibility profile and inducible resistance to macrolides of Mycobacterium abscessus and Mycobacterium massiliense in Korea. Diagn Microbiol Infect Dis. 2015; 81:107–111.68. Lee SH, Yoo HK, Kim SH, Koh WJ, Kim CK, Park YK, et al. The drug resistance profile of Mycobacterium abscessus group strains from Korea. Ann Lab Med. 2014; 34:31–37.69. Maurer FP, Bruderer VL, Ritter C, Castelberg C, Bloemberg GV, Bottger EC. Lack of antimicrobial bactericidal activity in Mycobacterium abscessus. Antimicrob Agents Chemother. 2014; 58:3828–3836.70. Ferro BE, van Ingen J, Wattenberg M, van Soolingen D, Mouton JW. Time-kill kinetics of antibiotics active against rapidly growing mycobacteria. J Antimicrob Chemother. 2015; 70:811–817.71. Maurer FP, Bruderer VL, Castelberg C, Ritter C, Scherbakov D, Bloemberg GV, et al. Aminoglycoside-modifying enzymes determine the innate susceptibility to aminoglycoside antibiotics in rapidly growing mycobacteria. J Antimicrob Chemother. 2015; 70:1412–1419.72. Maurer FP, Castelberg C, Quiblier C, Bottger EC, Somoskovi A. Erm(41)-dependent inducible resistance to azithromycin and clarithromycin in clinical isolates of Mycobacterium abscessus. J Antimicrob Chemother. 2014; 69:1559–1563.73. Koh WJ, Jeon K, Lee NY, Kim BJ, Kook YH, Lee SH, et al. Clinical significance of differentiation of Mycobacterium massiliense from Mycobacterium abscessus. Am J Respir Crit Care Med. 2011; 183:405–410.74. Kim HS, Lee KS, Koh WJ, Jeon K, Lee EJ, Kang H, et al. Serial CT findings of Mycobacterium massiliense pulmonary disease compared with Mycobacterium abscessus disease after treatment with antibiotic therapy. Radiology. 2012; 263:260–270.75. Lyu J, Kim BJ, Kim BJ, Song JW, Choi CM, Oh YM, et al. A shorter treatment duration may be sufficient for patients with Mycobacterium massiliense lung disease than with Mycobacterium abscessus lung disease. Respir Med. 2014; 108:1706–1712.76. Jarand J, Levin A, Zhang L, Huitt G, Mitchell JD, Daley CL. Clinical and microbiologic outcomes in patients receiving treatment for Mycobacterium abscessus pulmonary disease. Clin Infect Dis. 2011; 52:565–571.77. Mirsaeidi M, Farshidpour M, Ebrahimi G, Aliberti S, Falkinham JO 3rd. Management of nontuberculous mycobacterial infection in the elderly. Eur J Intern Med. 2014; 25:356–363.78. van Ingen J, Egelund EF, Levin A, Totten SE, Boeree MJ, Mouton JW, et al. The pharmacokinetics and pharmacodynamics of pulmonary Mycobacterium avium complex disease treatment. Am J Respir Crit Care Med. 2012; 186:559–565.79. van Ingen J, Ferro BE, Hoefsloot W, Boeree MJ, van Soolingen D. Drug treatment of pulmonary nontuberculous mycobacterial disease in HIV-negative patients: the evidence. Expert Rev Anti Infect Ther. 2013; 11:1065–1077.80. Westphal JF. Macrolide-induced clinically relevant drug interactions with cytochrome P-450A (CYP) 3A4: an update focused on clarithromycin, azithromycin and dirithromycin. Br J Clin Pharmacol. 2000; 50:285–295.81. Rose SJ, Neville ME, Gupta R, Bermudez LE. Delivery of aerosolized liposomal amikacin as a novel approach for the treatment of nontuberculous mycobacteria in an experimental model of pulmonary infection. PLoS One. 2014; 9:e108703.82. Winthrop KL, Ku JH, Marras TK, Griffith DE, Daley CL, Olivier KN, et al. The tolerability of linezolid in the treatment of nontuberculous mycobacterial disease. Eur Respir J. 2015; 45:1177–1179.83. Wallace RJ Jr, Brown-Elliott BA, Crist CJ, Mann L, Wilson RW. Comparison of the in vitro activity of the glycylcycline tigecycline (formerly GAR-936) with those of tetracycline, minocycline, and doxycycline against isolates of nontuberculous mycobacteria. Antimicrob Agents Chemother. 2002; 46:3164–3167.84. Wallace RJ Jr, Dukart G, Brown-Elliott BA, Griffith DE, Scerpella EG, Marshall B. Clinical experience in 52 patients with tigecycline-containing regimens for salvage treatment of Mycobacterium abscessus and Mycobacterium chelonae infections. J Antimicrob Chemother. 2014; 69:1945–1953.85. Huang CW, Chen JH, Hu ST, Huang WC, Lee YC, Huang CC, et al. Synergistic activities of tigecycline with clarithromycin or amikacin against rapidly growing mycobacteria in Taiwan. Int J Antimicrob Agents. 2013; 41:218–223.86. Tang S, Yao L, Hao X, Liu Y, Zeng L, Liu G, et al. Clofazimine for the treatment of multidrug-resistant tuberculosis: prospective, multicenter, randomized controlled study in China. Clin Infect Dis. 2015; 60:1361–1367.87. Shen GH, Wu BD, Hu ST, Lin CF, Wu KM, Chen JH. High efficacy of clofazimine and its synergistic effect with amikacin against rapidly growing mycobacteria. Int J Antimicrob Agents. 2010; 35:400–404.88. Philley JV, Wallace RJ Jr, Benwill JL, Taskar V, Brown-Elliott BA, Thakkar F, et al. Preliminary results of bedaquiline as salvage therapy for patients with nontuberculous mycobacterial lung disease. Chest. 2015; 148:499–506.89. Bjarnsholt T, Hoiby N, Donelli G, Imbert C, Forsberg A. Understanding biofilms: are we there yet? FEMS Immunol Med Microbiol. 2012; 65:125–126.90. Qvist T, Eickhardt S, Kragh KN, Andersen CB, Iversen M, Hoiby N, et al. Chronic pulmonary disease with Mycobacterium abscessus complex is a biofilm infection. Eur Respir J. 2015; 46:1823–1826.91. Fennelly KP, Ojano-Dirain C, Yang Q, Liu L, Lu L, Progulske-Fox A, et al. Biofilm formation by Mycobacterium abscessus in a lung cavity. Am J Respir Crit Care Med. 2016; 193:692–693.92. Rose SJ, Babrak LM, Bermudez LE. Mycobacterium avium possesses extracellular DNA that contributes to biofilm formation, structural integrity, and tolerance to antibiotics. PLoS One. 2015; 10:e0128772.93. Aung TT, Yam JK, Lin S, Salleh SM, Givskov M, Liu S, et al. Biofilms of pathogenic nontuberculous mycobacteria targeted by new therapeutic approaches. Antimicrob Agents Chemother. 2015; 60:24–35.94. Griffith DE, Aksamit TR. Therapy of refractory nontuberculous mycobacterial lung disease. Curr Opin Infect Dis. 2012; 25:218–227.95. Mitchell JD. Surgical approach to pulmonary nontuberculous mycobacterial infections. Clin Chest Med. 2015; 36:117–122.96. Yu JA, Pomerantz M, Bishop A, Weyant MJ, Mitchell JD. Lady Windermere revisited: treatment with thoracoscopic lobectomy/segmentectomy for right middle lobe and lingular bronchiectasis associated with non-tuberculous mycobacterial disease. Eur J Cardiothorac Surg. 2011; 40:671–675.97. Shiraishi Y, Katsuragi N, Kita H, Hyogotani A, Saito MH, Shimoda K. Adjuvant surgical treatment of nontuberculous mycobacterial lung disease. Ann Thorac Surg. 2013; 96:287–291.98. Kang HK, Park HY, Kim D, Jeong BH, Jeon K, Cho JH, et al. Treatment outcomes of adjuvant resectional surgery for nontuberculous mycobacterial lung disease. BMC Infect Dis. 2015; 15:76.99. Shiraishi Y. Surgical treatment of nontuberculous mycobacterial lung disease. Gen Thorac Cardiovasc Surg. 2014; 62:475–480.100. Shiraishi Y. Current status of nontuberculous mycobacterial surgery in Japan: analysis of data from the annual survey by the Japanese Association for Thoracic Surgery. Gen Thorac Cardiovasc Surg. 2016; 64:14–17.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Nontuberculous Mycobacterial Lung Disease
- Diagnosis of Pulmonary Tuberculosis and Nontuberculous Mycobacterial Lung Disease in Korea
- Respiratory Review of 2009: Nontuberculous Mycobacterium
- Radiologic Diagnosis of Nontuberculous Mycobacterial Pulmonary Disease
- Diagnosis and treatment of nontuberculous mycobacterial lung disease