Nutr Res Pract.
2009 Sep;3(3):242-246.
Alterations in the blood glucose, serum lipids and renal oxidative stress in diabetic rats by supplementation of onion (Allium cepa. Linn)
- Affiliations
-
- 1Solar Salt Biotechnology Research Center, Mokpo National University, Muan-gun, Jeonnam 534-729, Korea.
- 2Major in Food and Nutrition, Mokpo National University, Muan-gun, Jeonnam 534-729, Korea. kha@mokpo.ac.kr
Abstract
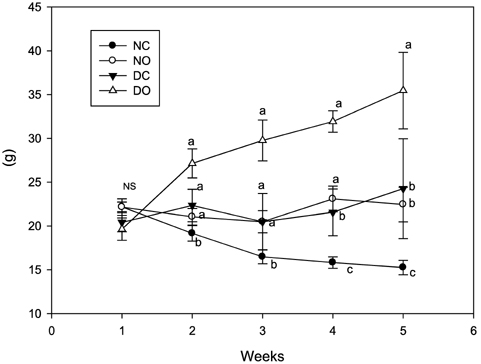
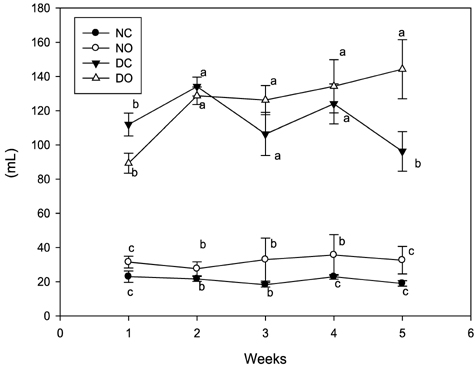
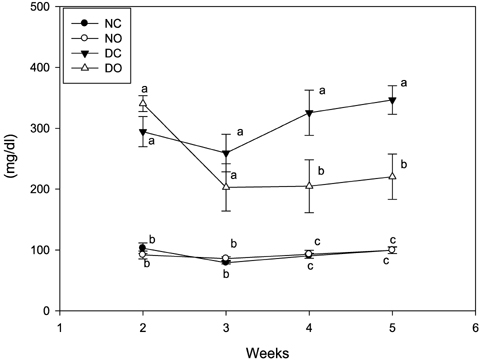
- This study examined the anti-diabetic effect of onion (Allium cepa. Linn) in the streptozotocin (STZ)-induced diabetic rats. Male Sprague-Dawley rats were divided into normal rats fed control diet or supplemented with onion powder (7% w/w) and diabetic rats fed control diet or supplemented with onion powder. Diabetes was induced by a single injection of STZ (60 mg/kg, ip) in citrate buffer. The animals were fed each of the experimental diet for 5 weeks. Blood glucose levels of rats supplemented with onion were lower than those of rats fed control diet in the diabetic rats. Onion also decreased the total serum lipid, triglyceride, and atherogenic index and increased HDL-cholesterol/total cholesterol ratio in the diabetic rats. Glutathione peroxidase, glutathione reductase and glutathione S-transferase activities were high in the diabetic rats compared to normal rats and reverted to near-control values by onion. These results indicate that onion decreased blood glucose, serum lipid levels and reduced renal oxidative stress in STZ-induced diabetic rats and this effect might exert the anti-diabetic effect of onion.
Keyword
MeSH Terms
Figure
Reference
-
1. Babu PS, Srinivasan K. Renal lesions in streptozotocin-induced diabetic rats maintained on onion and capsaicin containing diets. J Nutr Biochem. 1999. 10:477–483.
Article2. Banginski ES, Foa PP, Zak B. Methods of Enzymatic Analysis. 1983. 2. New York. USA: Academic Press;876–880.3. Bordia A, Bansal HC, Arora SK, Singh SV. Effect of essential oils of garlic and onion on alimentary hyperlipidemia. Atherosclerosis. 1975. 21:15–19.
Article4. Bordia A, Verma SK, Vyas AK. Effect of essential oils of onion and garlic on experimental atherosclerosis in rabbits. Atherosclerosis. 1977. 26:375–386.5. Carlberg I, Mannervick B. Glutathione reductase. Methods Enzymol. 1985. 113:484–499.6. Carson JF. Chemistry and biological properties of onion and garlic. Food Reviews Internationl. 1987. 3:71–103.7. Dincer Y, Alademir Z, Ilkova H, Akcay T. Susceptibility of gluathione and glutathione-related antioxidant activity to hydrogen peroxide in patients with type 2 diabetes: effect of glycemic control. Clin Biochem. 2002. 35:297–301.8. Feldt-Rasmussen B, Mathiesen ER, Deckert T. Effect of 2 years of strict metabolic control on progression of incipient nephropathy in insulin dependent diabetes. Lancet. 1986. 2:1300–1304.9. Ghosh R, Mukherjee B, Chatterjee M. A novel effect of selenium on streptozotocin-induced diabetic mice. Diabetes Res. 1994. 25:165–171.10. Habig WH, Pabst MJ, Jakoby WB. Glutathione S-transferase. J Biol Chem. 1974. 249:7130–7139.11. Hartnett ME, Stratton RD, Browne RW, Rosner BA, Lanham RJ, Armstrong D. Serum markers of oxidative stress and severity of diabetic retinopathy. Diabetes Care. 2000. 23:234–240.
Article12. Haygaard N. Cellular mechanisms of oxygen toxicity. Physiological Reviews. 1968. 48:311–373.
Article13. Howard BV, Savage PJ, Bennion LJ, Bennett PH. Lipoprotein composition in diabetes mellitus. Atherosclerosis. 1978. 30:153–162.
Article14. Irizar A, Ioannides C. Extrahepatic expression of P450 proteins in insulin-dependent diabetes mellitus. Xenobiotica. 1995. 25:941–949.
Article15. Jung YS, Kim MH, Lee SH, Baik EJ, Park SW, Moon CH. Antithrombotic effect of onion in streptozotocin-induced diabetic rat. Prostaglandins. 2002. 66:453–458.
Article16. Kakkar R, Mantha SV, Radhi J, Prasad K, Kalra J. Antioxidant defense system in diabetic kidney: a time course study. Life Sci. 1997. 60:667–679.
Article17. Kesavulu M, Rao BK, Giri R, Vijaya J, Subramanyam G, Apparao Ch. Lipid peroxidation and antioxidant enzyme status in Type 2 diabetic with coronary heart disease. Diabetes Res Clin Pract. 2001. 53:33.
Article18. Lowry OH, Rosebrough NJ, Farr AL, Randall RT. Proteinmeasurement with the folin phenol reagent. J Biol Chem. 1951. 193:265–275.19. Mak DH, Ip SP, Li PC, Poon MK, Ko KM. Alterations in tissue glutathione antioxidant system in streptozotocin-induced diabetic rats. Mol Cell Biochem. 1996. 162:153–158.
Article20. Medvedeva IV, Pufacheva TA, Dorodneva EF. Influence of glucose control on the main parameters of serum lipid profile and platelet membranes in patients with metabolic syndrome and type 2 diabetes mellitus. Atheroscler Suppl. 2002. 3:163.
Article21. Miranda M, Muriach M, Almansa I, Arnal E, Messeguer A, Diaz-Llopis M, Romero FJ, Bosch-Morell F. CR-6 protects glutathione peroxidase activity in experimental diabetes. Free Radic Biol Med. 2007. 43:1494–1498.
Article22. Moorhead JF. Lipids and progressive kidney disease. Kidney Int Suppl. 1991. 31:S35–S40.23. Mulec H, Johnson SA, Bjorck S. Relationship between serum cholesterol and diabetic nephropathy. Lancet. 1990. 335:1537–1538.24. Nikkila EA, Kekki M. Plasma triglyceride transport in diabetes mellitus. Metabolism. 1973. 22:1–22.25. Nourooz-Zadeh J, Rahini A, Tajaddini-Sarmadi J. Relationships between plasma measures of oxidative stress and metabolic controls in NIDDM. Diabetologia. 1997. 40:647–653.
Article26. Nuutila AM, Puupponen-Pimia R, Aarni M, Marja K, Caldentey O. Comparison of antioxidant activities of onion and garlic extracts by inhibition of lipid peroxidation and radical scavenging activity. Food Chem. 2003. 81:485–493.
Article27. Obrosova I, Cao X, Greene DA, Stevens MJ. Diabetes-induced changes in lens antioxidant status, glucose utilization and energy metabolism: effect of DL-alpha-lipoic acid. Diabetologia. 1998. 41:1442–1450.
Article28. Qureshi AA, Sami SA, Khan FA. Effects of stabilized rice bran, its soluble and fiber fractions on blood glucose levels and serum lipid parameters in human with diabetes mellitus type I and II. J Nutr Biochem. 2002. 13:175–187.
Article29. Reddy AS, Rao CV, Rivenson A, Kelloff G. Chemoprevention of colon carcinogenesis by organosulfur compounds. Cancer Res. 1993. 53:3493–3498.30. Tapple AL. Glutathione peroxidase and hydroperoxides. Meth Enzymol. 1978. 52:506–513.31. Wohaieb SA, Godin DV. Alterations in free radical tissue-defense mechanisms in streptozotocin-induced diabetes in rat: effects of insulin treatment. Diabetes. 1987. 36:1014–1018.
Article32. Zhang SL, Chen X, Hsieh TJ, Leclerc M, Henley N, Allidina A, Halle JP, Brunette MG, Filep JC, Tang SS, Ingelfinger JR, Chan JS. Hyperglycemia includes insulin resistance on angiotensinogen gene expression in diabetic rat kidney proximal tubular cells. J Endocrinol. 2000. 172:333–344.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Phenolic Acid Changes in Mycelia of Sclerotium rolfsii After Garlic and Onion Supplementation in a Broth Medium
- Effects of Allium Vegetable Intake on Levels of Plasma Glucose, Lipid and Minerals in Streptozotocin Induced Diabetic Rats
- Effects of onion peel water extracts on swimming endurance in rat
- Effects of beta-Carotene Supplementation on Lipid Peroxide Levels and Antioxidative Enzyme Activities in Diabetic Rats
- Effects of Short Term Antioxidant Cocktail Supplementation on the Oxidative Stress and Inflammatory Response of Renal Inflammation in Diabetic Mice