Korean J Urol.
2006 Feb;47(2):180-188. 10.4111/kju.2006.47.2.180.
The Mechanism of Damage to the Contralateral Testis Following Testicular Torsion and Detorsion in Rats and the Effect of Allopurinol Administration
- Affiliations
-
- 1Department of Urology, Seoul Municipal Boramae Hospital, Korea.
- 2Department of Urology, Seoul National University College of Medicine, Seoul, Korea. hchoi@snu.ac.kr
- KMID: 2294199
- DOI: http://doi.org/10.4111/kju.2006.47.2.180
Abstract
-
PURPOSE: This study was performed to determine the mechanism of contralateral testis damage after repair of testicular torsion. In addition, we investigated the effect of allopurinol pretreatment on the contralateral testis damage.
MATERIALS AND METHODS
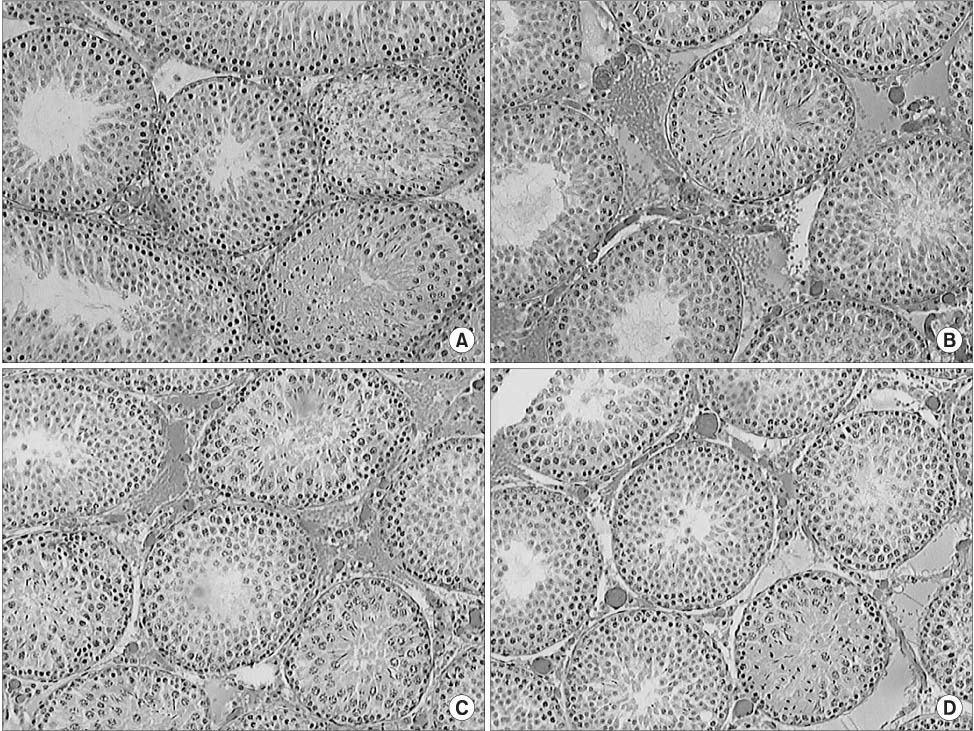
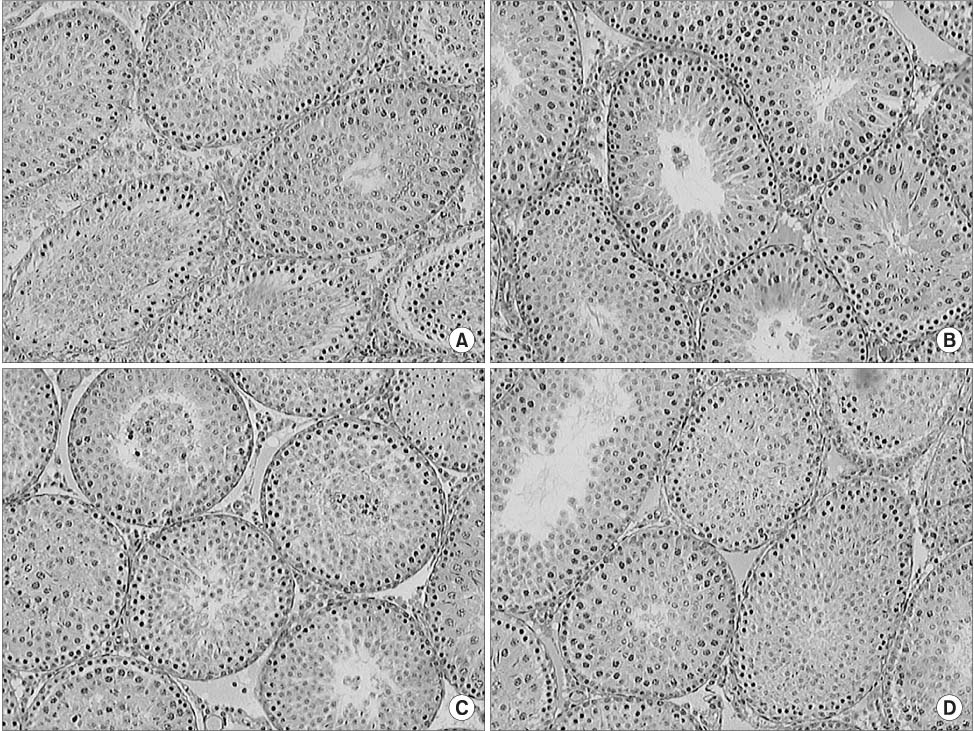
The levels of malondialdehyde (MDA) in the testes, kidneys and serum samples were determined in rats following torsion, detorsion or detorsion after allopurinol pretreatment, as were the levels of superoxide dismutase (SOD) in the testes samples. H&E staining was performed on the testes specimens and we calculated the mean numbers of spermatids per tubule in each group.
RESULTS
Torsion caused a significant increase in the MDA levels in the contralateral testes, and detorsion caused a further significant increase in MDA in the contralateral testes and in the serum. Moreover, pretreatment with allopurinol prevented this further increase. The levels of SOD in the contralateral testes were not significantly different between the sham and torsion groups, whereas detorsion caused a significant reduction in SOD. In addition, pretreatment with allopurinol also prevented this decrease. The contralateral testes were histologically normal in all groups and the mean numbers of spermatids per tubule in the ipsilateral and contralateral testes were similar in all groups.
CONCLUSIONS
Injury of the contralateral testis after the repair of testicular torsion seems to be caused by lipid peroxidation that is due to oxygen free radicals induced by the reperfusion of ischemic torsed testis. Toxic substances such as oxygen free radicals seem to be transferred to the contralateral testes through the blood stream, and allopurinol appears to protect the contralateral testes from the injury caused by detorsion.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Electroejaculation in the Male Rat
Sung Hyun Paick, Byung Ki Lee, Sung Wook Yoon, Minki Baek, Hyeong Gon Kim, Eun Young Song, Young Soo Lyoo, Yong Soo Lho
Korean J Urol. 2008;49(3):266-270. doi: 10.4111/kju.2008.49.3.266.
Reference
-
1. Cuckow PM, Frank JD. Torsion of the testis. BJU Int. 2000. 86:349–353.2. Visser AJ, Heyns CF. Testicular function after torsion of the spermatic cord. BJU Int. 2003. 92:200–203.3. Anderson JB, Williamson RC. The fate of the human testes following unilateral torsion of the spermatic cord. Br J Urol. 1986. 58:698–704.4. Anderson MJ, Dunn JK, Lipschults LI, Coburn M. Semen quality and endocrine parameters after acute testicular torsion. J Urol. 1992. 147:1545–1550.5. Goldwasser B, Weissenberg B, Lunenfeld B, Nativ O, Many M. Semen quality and hormonal status of patients following testicular torsion. Andrologia. 1984. 16:239–243.6. Thomas WE, Anderson JB, Williamson RC. Autoimmunization in testicular torsion. Br J Surg. 1985. 72:672.7. Karaguzel G, Gedikoglu G, Tanyel FC, Buyukpamukcu N, Hicsonmez A. Subsequent biological effects of chemical sympathectomy in rats undergoing unilateral testicular torsion. Eur Urol. 1995. 28:147–151.8. Choi H, Choo MS, Kim KM, Kim WH, Lee YS, Chung MH. The alterations of cellular metabolism in the contralateral testis following spermatic cord torsion in rats. J Urol. 1993. 150:577–580.9. Kizilcan F, Bernay I, Tanyel FC, Buyukpamukcu N, Bekdik C, Hicsonmez A. Ipsilateral and contralateral testicular blood flows during unilateral testicular torsion by 133Xe clearance technique. Int Urol Nephrol. 1992. 24:515–520.10. Sarica K, Kupeli B, Budak M, Kosar A, Kavukcu M, Durak I, et al. Influence of experimental spermatic cord torsion on the contralateral testis in rats. Evaluation of tissue free oxygen radical scavenger enzyme levels. Urol Int. 1997. 58:208–212.11. Oh SJ, Park CS, Lee KH, Kim DJ, Lim DJ, Jie JR, et al. Alteration of nitric oxide synthase subtype expression in contralateral testis of the rat in response to unilateral testicular torsion followed by detorsion. Korean J Urol. 2000. 41:650–658.12. Park EC, Kwak C, Son H, Oh SJ, Lee SE, Choi H. The role of clusterin in apoptosis induced by testicular torsion. Korean J Urol. 2004. 45:380–389.13. Turner TT, Tung KS, Tomomasa H, Wilson LW. Acute testicular ischemia results in germ cell-specific apoptosis in the rat. Biol Reprod. 1997. 57:1267–1274.14. Klein AS, Joh JW, Rangan U, Wang D, Bulkley GB. Allopurinol: discrimination of antioxidant from enzyme inhibitory activities. Free Radic Biol Med. 1996. 21:713–717.15. Akgur FM, Kilinc K, Aktug T, Olguner M. The effect of allopurinol pretreatment before detorting testicular torsion. J Urol. 1994. 151:1715–1717.16. Chakraborty J, Sinha Hikim AP, Jhunjhunwala JS. Stagnation of blood in the microvasculature of the affected and contralateral testes of men with short-term torsion of the spermatic cord. J Androl. 1985. 6:291–299.17. Werns SW, Lucchesi BR. Free radicals and ischemic tissue injury. Trends Pharmacol Sci. 1990. 11:161–166.18. Bulkley GB. Free radical-mediated reperfusion injury: a selective review. Br J Cancer. 1987. 8:Suppl. 66–73.19. Lacy F, Gough DA, Schmid-Schonbein GW. Role of xanthine oxidase in hydrogen peroxide production. Free Radic Biol Med. 1998. 25:720–727.20. Schoenberg MH, Beger HG. Reperfusion injury after intestinal ischemia. Crit Care Med. 1993. 21:1376–1386.21. Slater TF. Free-radical mechanisms in tissue injury. Biochem J. 1984. 222:1–15.22. Kehinde EO, Mojiminiyi OA, Mahmoud AH, Al-Awadi KA, Al-Hunayan A, Omu AE. The significance of measuring the time course of serum malondialdehyde concentration in patients with torsion of the testis. J Urol. 2003. 169:2177–2180.23. Savas C, Dindar H, Bilgehan A, Ataoglu O, Yucesan S. Pentoxifylline attenuates reperfusion injury in testicular torsion. Scand J Urol Nephrol. 2002. 36:65–70.24. Savas C, Dindar H, Aras T, Yucesan S. Pentoxifylline improves blood flow to both testes in testicular torsion. Int Urol Nephrol. 2002. 33:81–85.25. Greenstein A, Smith Harrison LI, Wakely PE, Kololgi S, Salzberg AD, Koontz WW Jr. The effect of polyethylene glycol-superoxide dismutase administration on histologic damage following spermatic cord torsion. J Urol. 1992. 148:639–641.26. Zini A, Abitbol J, Girardi SK, Schulsinger D, Goldstein M, Schlegel PN. Germ cell apoptosis and endothelial nitric oxide synthase (eNOS) expression following ischemia-reperfusion injury to testis. Arch Androl. 1998. 41:57–65.27. Abasiyanik A, Dagdonderen L. Beneficial effects of melatonin compared with allopurinol in experimental testicular torsion. J Pediatr Surg. 2004. 39:1238–1241.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The effect of testicular torsion on the contralateral testis in rats
- The Effect of Testicular Torsion on the Contralateral Testis in Rats; Histologic Changes and its Reversibility
- Comparisons of the effects of testicular torsion through a scrotal wall incision and an abdominal wall incision on the contralateral testis in prepubertal rats
- Protective Effect of Capsaicin on Contralateral Testis of Rats during Unilateral Testicular Torsion
- The Effect of Testicular Torsion on the Histologic Findings and Apoptosis of the Contralateral Testis at Various Ages in Rats