Korean J Physiol Pharmacol.
2013 Dec;17(6):525-530. 10.4196/kjpp.2013.17.6.525.
Functional Characterization of ABCB4 Mutations Found in Low Phospholipid-Associated Cholelithiasis (LPAC)
- Affiliations
-
- 1Department of Pharmacology, Tissue Injury Defense Research Center, School of Medicine, Ewha Womans University, Seoul 158-710, Korea. jihachoi@ewha.ac.kr
- KMID: 2285480
- DOI: http://doi.org/10.4196/kjpp.2013.17.6.525
Abstract
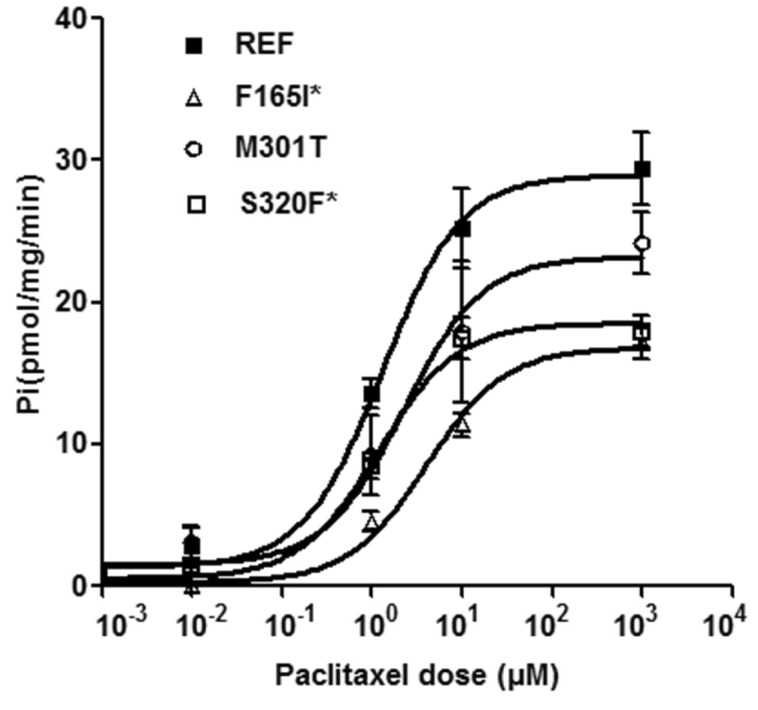
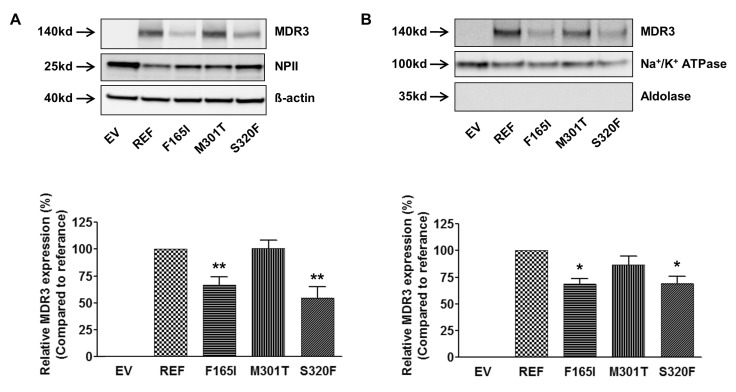
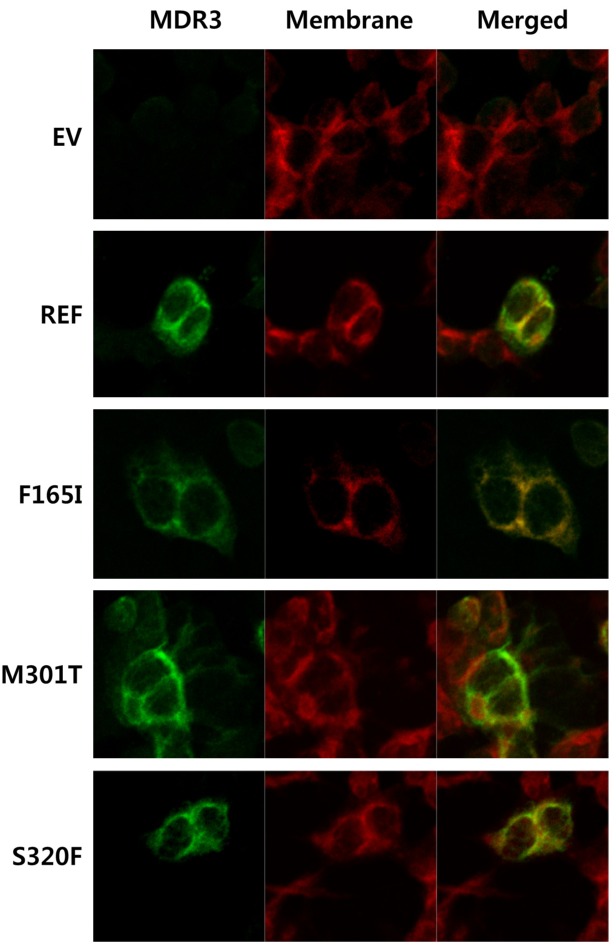
- Multidrug resistance 3 (MDR3) is expressed on the canalicular membrane of the hepatocytes and plays an important role in protecting the liver from bile acids. Altered ABCB4 gene expression can lead to a rare hepatic disease, low phospholipid-associated cholelithiasis (LPAC). In this study, we characterized 3 ABCB4 mutations in LPAC patients using various in vitro assay systems. We first measured the ability of each mutant to transport paclitaxel and then the mechanisms by which these mutations might change MDR3 transport activity were determined using immunoblotting, cell surface protein biotinylation, and immunofluorescence. Through a membrane vesicular transport assay, we observed that the uptake of paclitaxel was significantly reduced in membrane vesicles expressing 2 ABCB4 mutations, F165I and S320F. Both mutants showed significantly decreased total and cell surface MDR3 expression. These data suggest two missense mutations of ABCB4 may alter function of MDR3 and ultimately can be determined as LPAC-causing mutations.
Keyword
MeSH Terms
Figure
Reference
-
1. Oude Elferink RP, Paulusma CC. Function and pathophysiological importance of ABCB4 (MDR3 P-glycoprotein). Pflugers Arch. 2007; 453:601–610. PMID: 16622704.
Article2. Ruetz S, Gros P. Phosphatidylcholine translocase: a physiological role for the mdr2 gene. Cell. 1994; 77:1071–1081. PMID: 7912658.
Article3. Rosmorduc O, Hermelin B, Boelle PY, Parc R, Taboury J, Poupon R. ABCB4 gene mutation-associated cholelithiasis in adults. Gastroenterology. 2003; 125:452–459. PMID: 12891548.
Article4. Deleuze JF, Jacquemin E, Dubuisson C, Cresteil D, Dumont M, Erlinger S, Bernard O, Hadchouel M. Defect of multidrug-resistance 3 gene expression in a subtype of progressive familial intrahepatic cholestasis. Hepatology. 1996; 23:904–908. PMID: 8666348.
Article5. Eloranta ML, Heiskanen JT, Hiltunen MJ, Mannermaa AJ, Punnonen KR, Heinonen ST. Multidrug resistance 3 gene mutation 1712delT and estrogen receptor alpha gene polymorphisms in Finnish women with obstetric cholestasis. Eur J Obstet Gynecol Reprod Biol. 2002; 104:109–112. PMID: 12206920.6. Yoshikado T, Takada T, Yamamoto T, Yamaji H, Ito K, Santa T, Yokota H, Yatomi Y, Yoshida H, Goto J, Tsuji S, Suzuki H. Itraconazole-induced cholestasis: involvement of the inhibition of bile canalicular phospholipid translocator MDR3/ABCB4. Mol Pharmacol. 2011; 79:241–250. PMID: 21056966.
Article7. Lang C, Meier Y, Stieger B, Beuers U, Lang T, Kerb R, Kullak-Ublick GA, Meier PJ, Pauli-Magnus C. Mutations and polymorphisms in the bile salt export pump and the multidrug resistance protein 3 associated with drug-induced liver injury. Pharmacogenet Genomics. 2007; 17:47–60. PMID: 17264802.
Article8. Rosmorduc O, Poupon R. Low phospholipid associated cholelithiasis: association with mutation in the MDR3/ABCB4 gene. Orphanet J Rare Dis. 2007; 2:29. PMID: 17562004.
Article9. Poupon R, Rosmorduc O, Boëlle PY, Chrétien Y, Corpechot C, Chazouillères O, Housset C, Barbu V. Genotype-phenotype relationships in the low-phospholipid-associated cholelithiasis syndrome: a study of 156 consecutive patients. Hepatology. 2013; 58:1105–1110. PMID: 23533021.
Article10. Delaunay JL, Durand-Schneider AM, Delautier D, Rada A, Gautherot J, Jacquemin E, Aït-Slimane T, Maurice M. A missense mutation in ABCB4 gene involved in progressive familial intrahepatic cholestasis type 3 leads to a folding defect that can be rescued by low temperature. Hepatology. 2009; 49:1218–1227. PMID: 19185004.
Article11. Gautherot J, Durand-Schneider AM, Delautier D, Delaunay JL, Rada A, Gabillet J, Housset C, Maurice M, Aït-Slimane T. Effects of cellular, chemical, and pharmacological chaperones on the rescue of a trafficking-defective mutant of the ATP-binding cassette transporter proteins ABCB1/ABCB4. J Biol Chem. 2012; 287:5070–5078. PMID: 22184139.
Article12. Jang GH, Kim TH, Choe Y, Ham A, Choi JH. Functional characterization of genetic variations in the MDR3 promoter. Biochem Biophys Res Commun. 2013; 430:1312–1318. PMID: 23261441.
Article13. Kim WJ, Lee JH, Yi J, Cho YJ, Heo K, Lee SH, Kim SW, Kim MK, Kim KH, In Lee B, Lee MG. A nonsynonymous variation in MRP2/ABCC2 is associated with neurological adverse drug reactions of carbamazepine in patients with epilepsy. Pharmacogenet Genomics. 2010; 20:249–256. PMID: 20216337.
Article14. Kwak JO, Lee SH, Lee GS, Kim MS, Ahn YG, Lee JH, Kim SW, Kim KH, Lee MG. Selective inhibition of MDR1 (ABCB1) by HM30181 increases oral bioavailability and therapeutic efficacy of paclitaxel. Eur J Pharmacol. 2010; 627:92–98. PMID: 19903471.
Article15. Degiorgio D, Colombo C, Seia M, Porcaro L, Costantino L, Zazzeron L, Bordo D, Coviello DA. Molecular characterization and structural implications of 25 new ABCB4 mutations in progressive familial intrahepatic cholestasis type 3 (PFIC3). Eur J Hum Genet. 2007; 15:1230–1238. PMID: 17726488.
Article16. Smith AJ, van Helvoort A, van Meer G, Szabo K, Welker E, Szakacs G, Varadi A, Sarkadi B, Borst P. MDR3 P-glycoprotein, a phosphatidylcholine translocase, transports several cytotoxic drugs and directly interacts with drugs as judged by interference with nucleotide trapping. J Biol Chem. 2000; 275:23530–23539. PMID: 10918072.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Phenotypic and Molecular Characteristics of Children with Progressive Familial Intrahepatic Cholestasis in South China
- A case of Wilson disease associated with hemolytic anemia and cholelithiasis
- Clinical Review of Cholelithiasis after Gastric Resection in Patients with Stomach Cancer
- Long-term Results of Roux-en-Y Procedure in Choledochal Cyst and in Cholelithiasis
- Cholelithiasis