Korean J Physiol Pharmacol.
2013 Aug;17(4):321-329. 10.4196/kjpp.2013.17.4.321.
Changes in c-Fos Expression in the Forced Swimming Test: Common and Distinct Modulation in Rat Brain by Desipramine and Citalopram
- Affiliations
-
- 1Department of Pharmacology, Korea University College of Medicine, Seoul 136-705, Korea. kyungho@korea.ac.kr
- KMID: 2285467
- DOI: http://doi.org/10.4196/kjpp.2013.17.4.321
Abstract
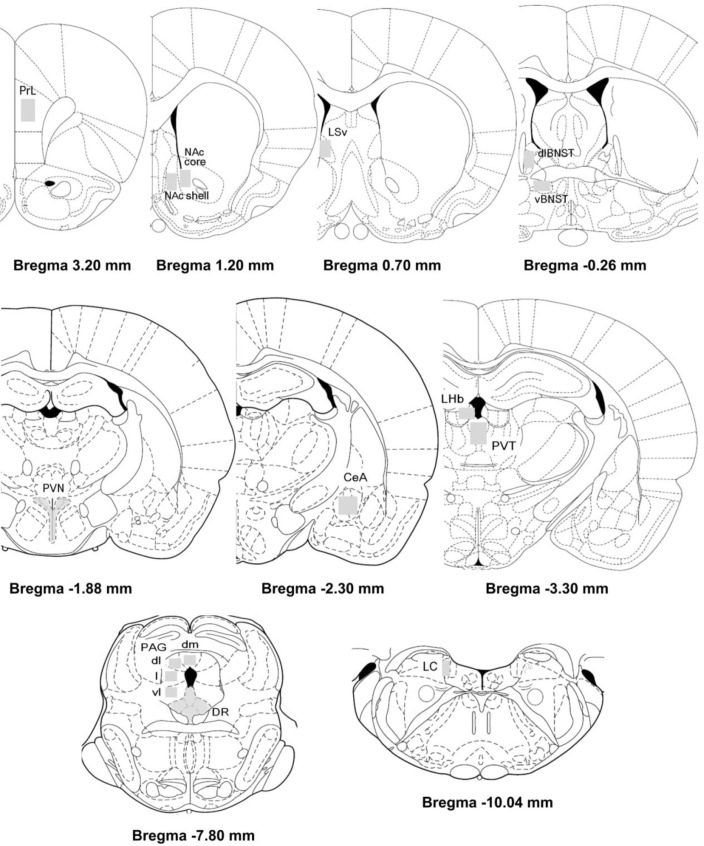
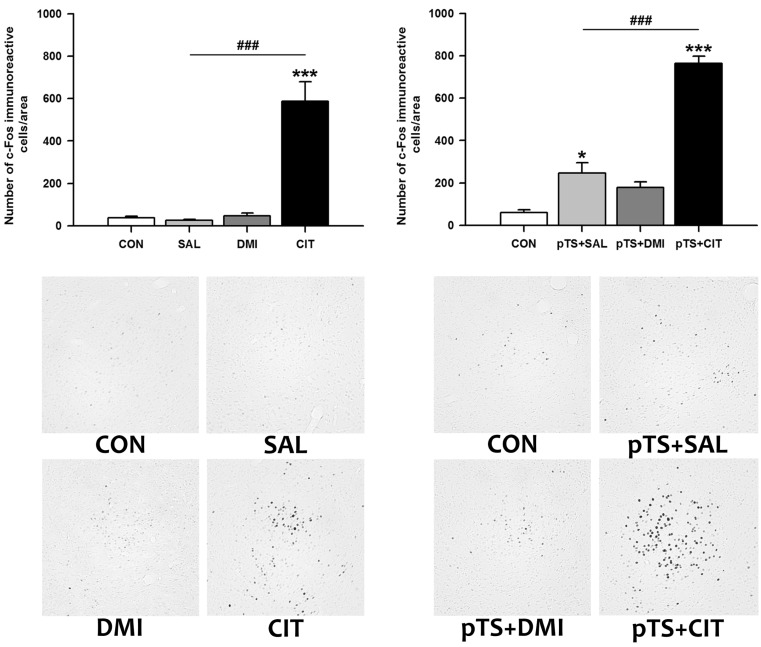
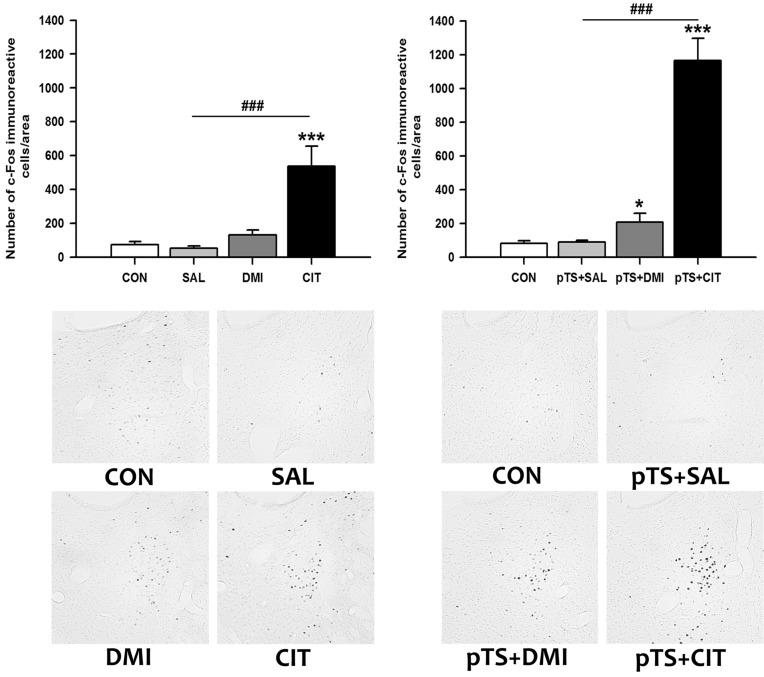
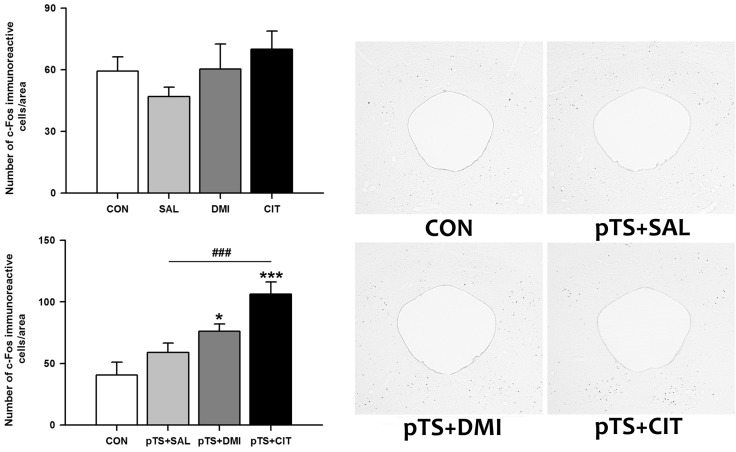
- Rodents exposed to a 15-min pretest swim in the forced swimming test (FST) exhibit prolonged immobility in a subsequent 5-min test swim, and antidepressant treatment before the test swim reduces immobility. At present, neuronal circuits recruited by antidepressant before the test swim remain unclear, and also less is known about whether antidepressants with different mechanisms of action could influence neural circuits differentially. To reveal the neural circuits associated with antidepressant effect in the FST, we injected desipramine or citalopram 0.5 h, 19 h, and 23 h after the pretest swim and observed changes in c-Fos expression in rats before the test swim, namely 24 h after the pretest swim. Desipramine treatment alone in the absence of pretest swim was without effect, whereas citalopram treatment alone significantly increased the number of c-Fos-like immunoreactive cells in the central nucleus of the amygdala and bed nucleus of the stria terminalis, where this pattern of increase appears to be maintained after the pretest swim. Both desipramine and citalopram treatment after the pretest swim significantly increased the number of c-Fos-like immunoreactive cells in the ventral lateral septum and ventrolateral periaqueductal gray before the test swim. These results suggest that citalopram may affect c-Fos expression in the central nucleus of the amygdala and bed nucleus of the stria terminalis distinctively and raise the possibility that upregulation of c-Fos in the ventral lateral septum and ventrolateral periaqueductal gray before the test swim may be one of the probable common mechanisms underlying antidepressant effect in the FST.
Keyword
MeSH Terms
Figure
Reference
-
1. Detke MJ, Lucki I. Detection of serotonergic and noradrenergic antidepressants in the rat forced swimming test: the effects of water depth. Behav Brain Res. 1996; 73:43–46. PMID: 8788475.
Article2. Richelson E. Synaptic effects of antidepressants. J Clin Psychopharmacol. 1996; 16(3 Suppl 2):1S–7S. PMID: 8784643.
Article3. Cryan JF, Markou A, Lucki I. Assessing antidepressant activity in rodents: recent developments and future needs. Trends Pharmacol Sci. 2002; 23:238–245. PMID: 12008002.
Article4. Cryan JF, Valentino RJ, Lucki I. Assessing substrates underlying the behavioral effects of antidepressants using the modified rat forced swimming test. Neurosci Biobehav Rev. 2005; 29:547–569. PMID: 15893822.
Article5. Duncan GE, Knapp DJ, Johnson KB, Breese GR. Functional classification of antidepressants based on antagonism of swim stress-induced fos-like immunoreactivity. J Pharmacol Exp Ther. 1996; 277:1076–1089. PMID: 8627519.6. Kawahara R, Soeda F, Kawaura K, Honda S, Miki R, Noguchi T, Shirasaki T, Takahama K. Effect of tipepidine with novel antidepressant-like action on c-fos-like protein expression in rat brain. Brain Res. 2013; 1513:135–142. PMID: 23548603.
Article7. Silva M, Aguiar DC, Diniz CR, Guimarães FS, Joca SR. Neuronal NOS inhibitor and conventional antidepressant drugs attenuate stress-induced fos expression in overlapping brain regions. Cell Mol Neurobiol. 2012; 32:443–453. PMID: 22120186.
Article8. Jama A, Cecchi M, Calvo N, Watson SJ, Akil H. Interindividual differences in novelty-seeking behavior in rats predict differential responses to desipramine in the forced swim test. Psychopharmacology (Berl). 2008; 198:333–340. PMID: 18438645.
Article9. Owens MJ, Morgan WN, Plott SJ, Nemeroff CB. Neurotransmitter receptor and transporter binding profile of antidepressants and their metabolites. J Pharmacol Exp Ther. 1997; 283:1305–1322. PMID: 9400006.10. Porsolt RD, Le Pichon M, Jalfre M. Depression: a new animal model sensitive to antidepressant treatments. Nature. 1977; 266:730–732. PMID: 559941.
Article11. Detke MJ, Rickels M, Lucki I. Active behaviors in the rat forced swimming test differentially produced by serotonergic and noradrenergic antidepressants. Psychopharmacology (Berl). 1995; 121:66–72. PMID: 8539342.
Article12. Porsolt RD, Anton G, Blavet N, Jalfre M. Behavioural despair in rats: a new model sensitive to antidepressant treatments. Eur J Pharmacol. 1978; 47:379–391. PMID: 204499.
Article13. Kang S, Kim HJ, Kim HJ, Shin SK, Choi SH, Lee MS, Shin KH. Effects of reboxetine and citalopram pretreatment on changes in cocaine and amphetamine regulated transcript (CART) expression in rat brain induced by the forced swimming test. Eur J Pharmacol. 2010; 647:110–116. PMID: 20826136.
Article14. Briand LA, Vassoler FM, Pierce RC, Valentino RJ, Blendy JA. Ventral tegmental afferents in stress-induced reinstatement: the role of cAMP response element-binding protein. J Neurosci. 2010; 30:16149–16159. PMID: 21123561.
Article15. Paxinos G, Watson C. The rat brain in stereotaxic coordinates (Deluxe Edition). Fourth Edition. 4th ed. San Diego: Academic Press;1998.16. Bilang-Bleuel A, Rech J, De Carli S, Holsboer F, Reul JM. Forced swimming evokes a biphasic response in CREB phosphorylation in extrahypothalamic limbic and neocortical brain structures in the rat. Eur J Neurosci. 2002; 15:1048–1060. PMID: 11918664.
Article17. Slattery DA, Morrow JA, Hudson AL, Hill DR, Nutt DJ, Henry B. Comparison of alterations in c-fos and Egr-1 (zif268) expression throughout the rat brain following acute administration of different classes of antidepressant compounds. Neuropsychopharmacology. 2005; 30:1278–1287. PMID: 15812568.
Article18. Patel S, Roelke CT, Rademacher DJ, Hillard CJ. Inhibition of restraint stress-induced neural and behavioural activation by endogenous cannabinoid signalling. Eur J Neurosci. 2005; 21:1057–1069. PMID: 15787710.
Article19. Singewald GM, Rjabokon A, Singewald N, Ebner K. The modulatory role of the lateral septum on neuroendocrine and behavioral stress responses. Neuropsychopharmacology. 2011; 36:793–804. PMID: 21160468.
Article20. Steciuk M, Kram M, Kramer GL, Petty F. Decrease in stress-induced c-Fos-like immunoreactivity in the lateral septal nucleus of learned helpless rats. Brain Res. 1999; 822:256–259. PMID: 10082906.
Article21. Tzavara ET, Davis RJ, Perry KW, Li X, Salhoff C, Bymaster FP, Witkin JM, Nomikos GG. The CB1 receptor antagonist SR141716A selectively increases monoaminergic neurotransmission in the medial prefrontal cortex: implications for therapeutic actions. Br J Pharmacol. 2003; 138:544–553. PMID: 12598408.
Article22. Beck CH. Acute treatment with antidepressant drugs selectively increases the expression of c-fos in the rat brain. J Psychiatry Neurosci. 1995; 20:25–32. PMID: 7865498.23. Sumner BE, Cruise LA, Slattery DA, Hill DR, Shahid M, Henry B. Testing the validity of c-fos expression profiling to aid the therapeutic classification of psychoactive drugs. Psychopharmacology (Berl). 2004; 171:306–321. PMID: 13680075.24. Davis M, Walker DL, Miles L, Grillon C. Phasic vs sustained fear in rats and humans: role of the extended amygdala in fear vs anxiety. Neuropsychopharmacology. 2010; 35:105–135. PMID: 19693004.
Article26. Bagdy G, Graf M, Anheuer ZE, Modos EA, Kantor S. Anxiety-like effects induced by acute fluoxetine, sertraline or m-CPP treatment are reversed by pretreatment with the 5-HT2C receptor antagonist SB-242084 but not the 5-HT1A receptor antagonist WAY-100635. Int J Neuropsychopharmacol. 2001; 4:399–408. PMID: 11806866.
Article27. Belzung C, Le Guisquet AM, Barreau S, Calatayud F. An investigation of the mechanisms responsible for acute fluoxetine-induced anxiogenic-like effects in mice. Behav Pharmacol. 2001; 12:151–162. PMID: 11485052.
Article28. Kurt M, Arik AC, Celik S. The effects of sertraline and fluoxetine on anxiety in the elevated plus-maze test in mice. J Basic Clin Physiol Pharmacol. 2000; 11:173–180. PMID: 11037770.
Article29. Singewald N, Salchner P, Sharp T. Induction of c-Fos expression in specific areas of the fear circuitry in rat forebrain by anxiogenic drugs. Biol Psychiatry. 2003; 53:275–283. PMID: 12586446.
Article30. Salchner P, Singewald N. Neuroanatomical substrates involved in the anxiogenic-like effect of acute fluoxetine treatment. Neuropharmacology. 2002; 43:1238–1248. PMID: 12527473.
Article31. Veening JG, Coolen LM, Spooren WJ, Joosten H, van Oorschot R, Mos J, Ronken E, Olivier B. Patterns of c-fos expression induced by fluvoxamine are different after acute vs chronic oral administration. Eur Neuropsychopharmacol. 1998; 8:213–226. PMID: 9716316.
Article32. Thomsen C, Helboe L. Regional pattern of binding and c-Fos induction by (R)- and (S)-citalopram in rat brain. Neuroreport. 2003; 14:2411–2414. PMID: 14663201.
Article33. Morelli M, Pinna A, Ruiu S, Del Zompo M. Induction of Fos-like-immunoreactivity in the central extended amygdala by antidepressant drugs. Synapse. 1999; 31:1–4. PMID: 10025677.
Article34. Lino-de-Oliveira C, Sales AJ, Del Bel EA, Silveira MC, Guimarães FS. Effects of acute and chronic fluoxetine treatments on restraint stress-induced Fos expression. Brain Res Bull. 2001; 55:747–754. PMID: 11595358.
Article35. Muigg P, Hoelzl U, Palfrader K, Neumann I, Wigger A, Landgraf R, Singewald N. Altered brain activation pattern associated with drug-induced attenuation of enhanced depression-like behavior in rats bred for high anxiety. Biol Psychiatry. 2007; 61:782–796. PMID: 17224133.
Article36. Walker DL, Toufexis DJ, Davis M. Role of the bed nucleus of the stria terminalis versus the amygdala in fear, stress, and anxiety. Eur J Pharmacol. 2003; 463:199–216. PMID: 12600711.
Article37. Crestani CC, Alves FH, Correa FM, Guimarães FS, Joca SR. Acute reversible inactivation of the bed nucleus of stria terminalis induces antidepressant-like effect in the rat forced swimming test. Behav Brain Funct. 2010; 6:30. PMID: 20515458.
Article38. Bandler R, Keay KA, Floyd N, Price J. Central circuits mediating patterned autonomic activity during active vs. passive emotional coping. Brain Res Bull. 2000; 53:95–104. PMID: 11033213.
Article39. Keay KA, Bandler R. Parallel circuits mediating distinct emotional coping reactions to different types of stress. Neurosci Biobehav Rev. 2001; 25:669–678. PMID: 11801292.
Article40. Lino-de-Oliveira C, De Lima TC, Carobrez AP. Dorsal periaqueductal gray matter inhibits passive coping strategy elicited by forced swimming stress in rats. Neurosci Lett. 2002; 335:87–90. PMID: 12459505.
Article41. Lino-de-Oliveira C, de Oliveira RM, Pádua Carobrez A, de Lima TC, del Bel EA, Guimarães FS. Antidepressant treatment reduces Fos-like immunoreactivity induced by swim stress in different columns of the periaqueductal gray matter. Brain Res Bull. 2006; 70:414–421. PMID: 17027777.
Article42. Bellchambers CE, Chieng B, Keay KA, Christie MJ. Swim-stress but not opioid withdrawal increases expression of c-fos immunoreactivity in rat periaqueductal gray neurons which project to the rostral ventromedial medulla. Neuroscience. 1998; 83:517–524. PMID: 9460759.
Article43. Berton O, Covington HE 3rd, Ebner K, Tsankova NM, Carle TL, Ulery P, Bhonsle A, Barrot M, Krishnan V, Singewald GM, Singewald N, Birnbaum S, Neve RL, Nestler EJ. Induction of deltaFosB in the periaqueductal gray by stress promotes active coping responses. Neuron. 2007; 55:289–300. PMID: 17640529.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of Hypoxia-ischemia on c-fos Expression in the Neonatal Rat Brain
- Effect of P. ginseng on the expression of c-Fos in the brain of Wistar rats with testosterone induced benign prostatic hyperplasia
- Studies on the Activation of Neurons involved in Osmoregulation in Developing and Aging Rat
- Expression Changes of c-Fos Protein of Rat Brain Following Pentylenetetrazol-induced Seizures
- The Effects of Superior Cervical Ganglionectomy on C-fos Expression in Rat Brain