Korean J Physiol Pharmacol.
2009 Dec;13(6):469-473. 10.4196/kjpp.2009.13.6.469.
Intraocular Injection of Muscimol Induces Illusory Motion Reversal in Goldfish
- Affiliations
-
- 1Department of Natural Sciences, College of Medicine, The Catholic University of Korea, Seoul 137-701, Korea. cjung@catholic.ac.kr
- KMID: 2285383
- DOI: http://doi.org/10.4196/kjpp.2009.13.6.469
Abstract
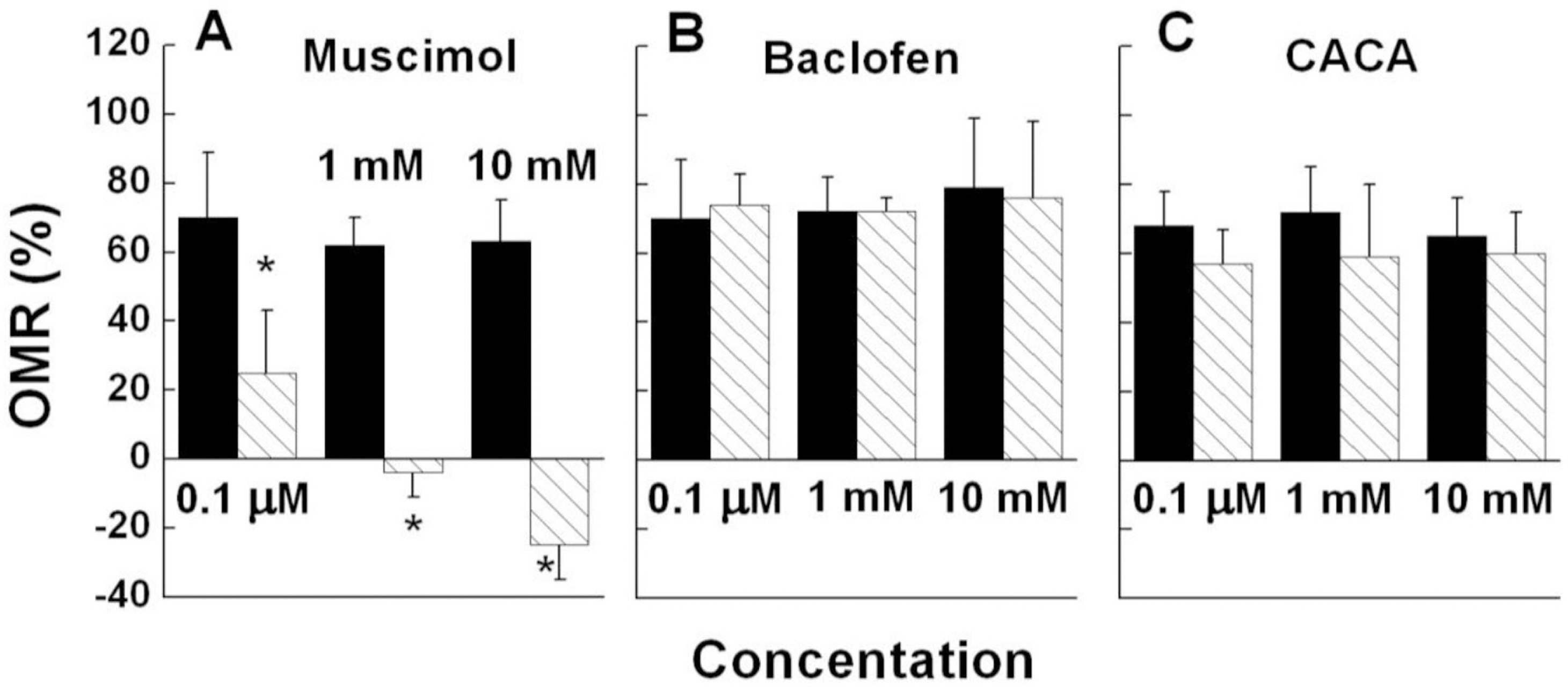
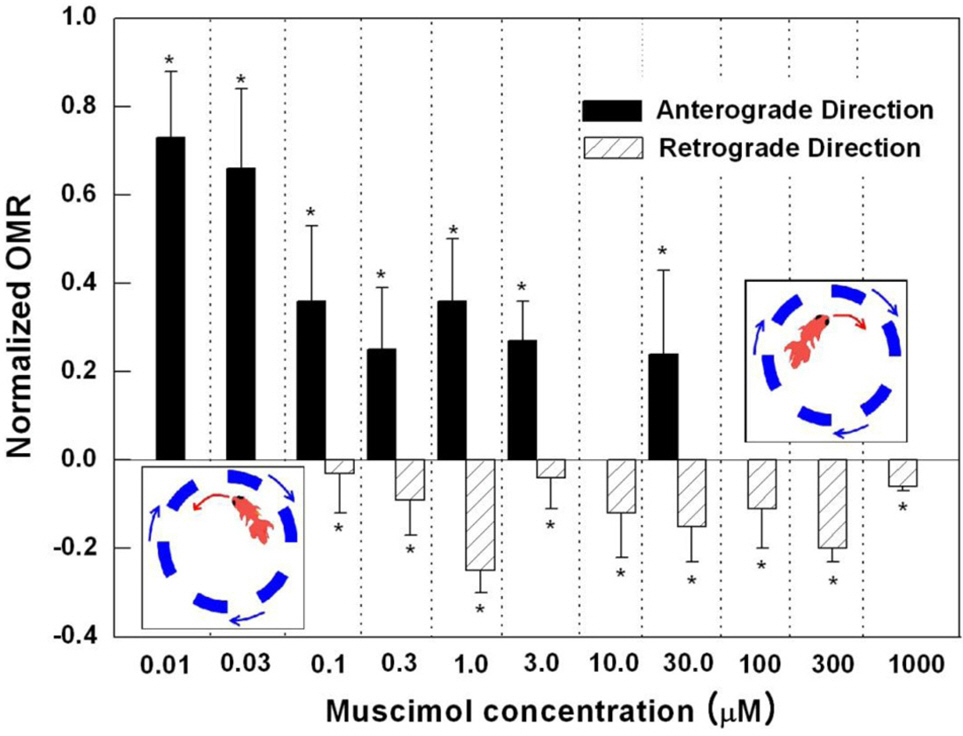
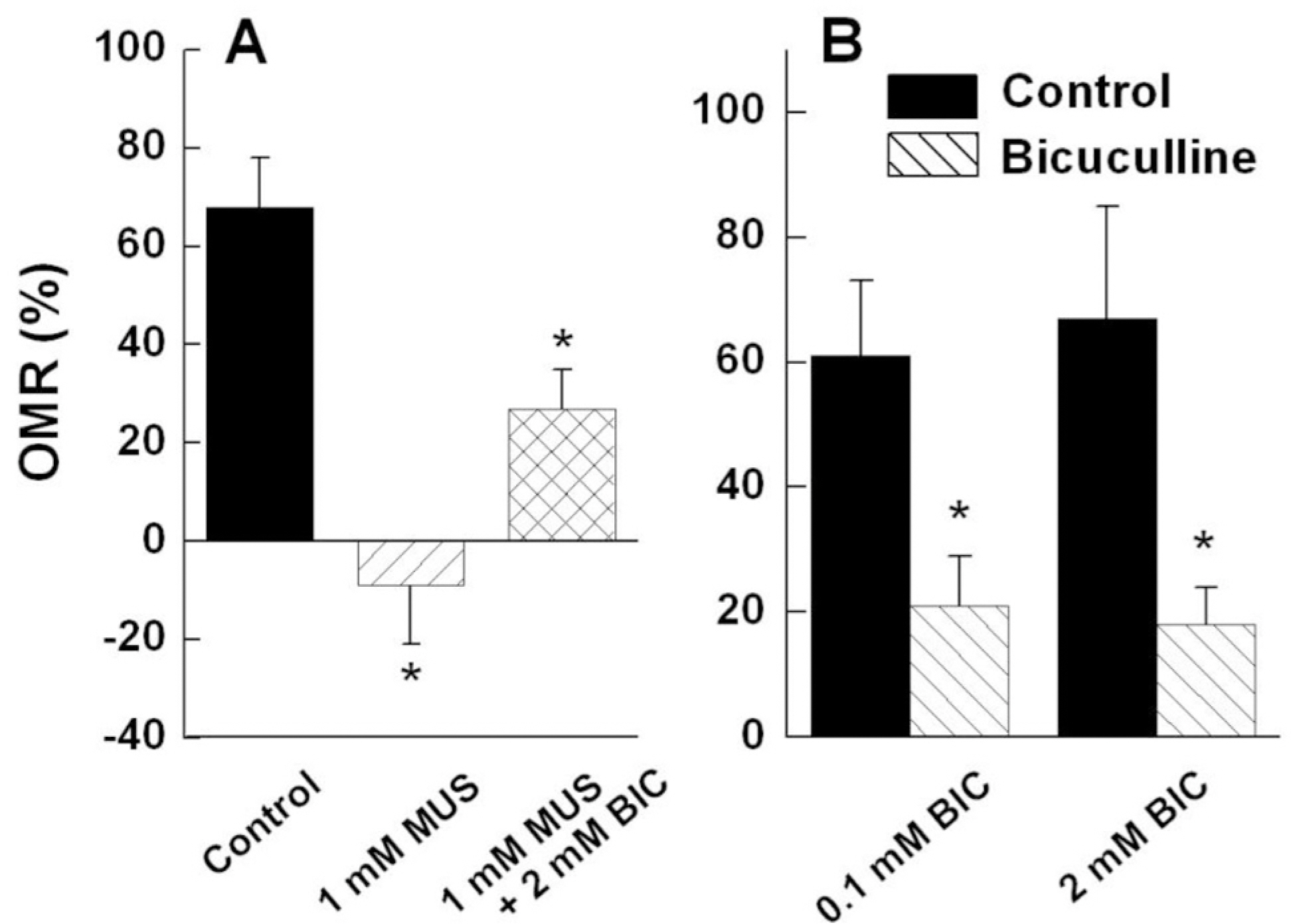
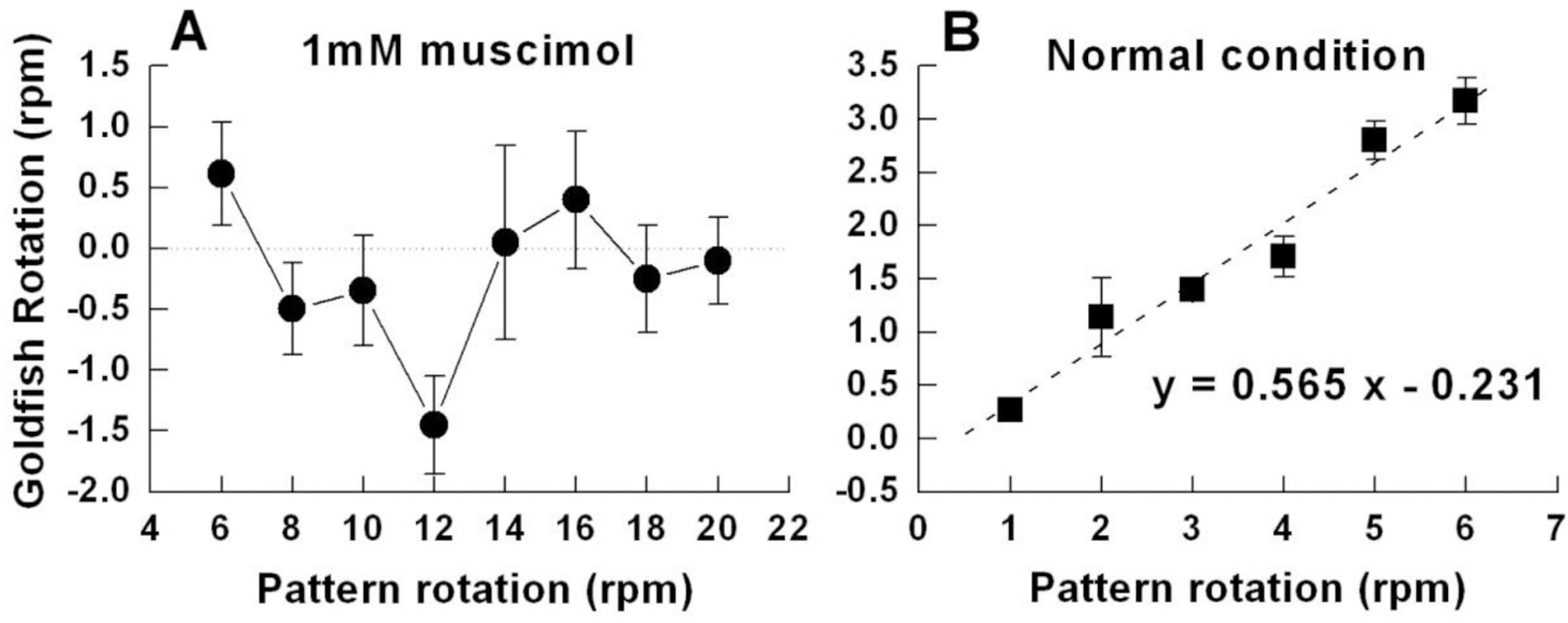
- Induced activation of the gamma-aminobutyric acidA (GABA(A)) receptor in the retina of goldfish caused the fish to rotate in the opposite direction to that of the spinning pattern during an optomotor response (OMR) measurement. Muscimol, a GABA(A) receptor agonist, modified OMR in a concentration-dependent manner. The GABA(B) receptor agonist baclofen and GABA(C) receptor agonist CACA did not affect OMR. The observed modifications in OMR included decreased anterograde rotation (0.01~0.03 micrometer), coexistence of retrograde rotation and decreased anterograde rotation (0.1~30 micrometer) and only retrograde rotation (100 micrometer~1 mM). In contrast, the GABA(A) receptor antagonist bicuculline blocked muscimolinduced retrograde rotation. Based on these results, we inferred that the coding inducing retrograde movement of the goldfish retina is essentially associated with the GABA(A) receptor-related visual pathway. Furthermore, from our novel approach using observations of goldfish behavior the induced discrete snapshot duration was approximately 573 ms when the fish were under the influence of muscimol.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
The Role of the Pattern Edge in Goldfish Visual Motion Detection
Sun-Hee Kim, Chang-Sub Jung
Korean J Physiol Pharmacol. 2010;14(6):413-417. doi: 10.4196/kjpp.2010.14.6.413.
Reference
-
References
Barlow HB., Hill RM. Selective sensitivity to direction of movement in ganglion cells of the rabbit retina. Science. 139:412–414. 1963.
ArticleBarlow HB., Levick WR. The mechanism of directionally selective units in rabbit's retina. J Physiol. 178:477–504. 1965.
ArticleCaldwell JH., Daw NW., Wyatt HJ. Effects of picrotoxin and strychnine on rabbit retinal ganglion cells: lateral interactions for cells with more complex receptive fields. J Physiol. 276:277–298. 1978.
ArticleDemb JB. Cellular mechanisms for direction selectivity in the retina. Neuron. 55:179–186. 2007.
ArticleEgelhaaf M., Borst A. Pilz B. The role of GABA in detecting visual motion. Brain Res. 509:156–160. 1990.Götz KG. Optomotorische Untersuchung des visuellen Systems einiger Augenmutanten der fruchtfliege Drosophila. Biol Cybern. 2:77–92. 1964.
ArticleHe S., Masland RH. Retinal direction selectivity after targeted laser ablation of starburst amacrine cells. Nature. 389:378–382. 1997.
ArticleIshida AT., Cohen BN. GABA-activated whole-cell currents in isolated retinal ganglion cells. J Neurophysiol. 60:381–396. 1988.
ArticleKaneko A., Tachibana M. GABA mediates the negative feedback from amacrine to bipolar cells. Neurosci Res Suppl. 6:S239–S251. 1987.
ArticleKittila CA., Massey SC. Effect of ON pathway blockade on directional selectivity in the rabbit retina. J Neurophysiol. 73:703–712. 1995.
ArticleKline K., Holcombe AO., Eagleman DM. Illusory motion reversal is caused by rivalry, not by perceptual snapshots of the visual field. Vision Res. 44:2653–2658. 2004.
ArticleKlooster J., Cardozo BN., Yazulla S., Kamermans M. Postsynaptic localization of gamma-aminobutyric acid transporters and receptors in the outer plexiform layer of the goldfish retina: An ultrastructural study. J Comp Neurol. 474:58–74. 2004.Lin ZS., Yazulla S. Heterogeneity of GABAA receptor in goldfish retina. J Comp Neurol. 345:429–439. 1994.Maaswinkel H., Li L. Spatio-temporal frequency characteristics of the optomotor response in zebrafish. Vision Res. 43:21–30. 2003.
ArticleMora-Ferrer C., Hausselt S., Hoffmann RS., Ebisch B., Schick S., Wollenberg K., Schneider C., Teege P., Jürgens K. Pharmacological properties of motion vision in goldfish measured with the optomotor response. Brain Res. 1058:17–29. 2005.
ArticleOyster CW., Takahashi E., Collewijn H. Direction-selective retinal ganglion cells and control of optokinetic nystagmus in the rabbit. Vision Res. 12:183–193. 1972.
ArticlePaik SS., Park NG., Lee SJ., Han HK., Jung CS., Bai SH., Chun MH. GABA receptors on horizontal cells in the goldfish retina. Vision Res. 43:2101–2106. 2003.
ArticlePakarian P., Yasamy MT. Wagon-wheel illusion under steady illumination: real or illusory? Perception. 32:1307–1310. 2003.
ArticlePan ZH., Slaughter MM. Control of retinal information coding by GABAB receptors. J Neurosci. 11:1810–1821. 1991.Purves D., Paydarfar JA., Andrews TJ. The wagon wheel illusion in movies and reality. Proc Natl Acad Sci USA. 93:3693–3697. 1996.
ArticleReichardt W. Evaluation of optical motion information by movement detectors. J Comp Physiol A. 161:533–547. 1987.
ArticleSchaerer S., Neumeyer C. Motion detection in goldfish investigated with the optomotor response is “color blind”. Vision Res. 36:4025–4034. 1996.
ArticleSrinivasan MV., Poteser M., Kral K. Motion detection in insect orientation and navigation. Vision Res. 39:2749–2766. 1999.
ArticleVanRullen R. The continuous wagon wheel illusion depends on, but is not identical to neuronal adaptation. Vision Res. 47:2143–2149. 2007.
ArticleVanRullen R., Reddy L., Koch C. The continuous wagon wheel illusion is associated with changes in electroencephalogram power at approximately 13 Hz. J Neurosci. 26:502–507. 2006.Watanabe S., Koizumi A., Matsunaga S., Stocker JW., Kaneko A. GABA-mediated inhibition between amacrine cells in the goldfish retina. J Neurophysiol. 84:1826–1834. 2000.Werblin FS. Response of retinal cells to moving spots: intracellular recording in Necturus maculosus. J Neurophysiol. 33:342–350. 1969.
ArticleWyatt HJ., Daw NW. Specific effects of neurotransmitter antagonists on ganglion cells in rabbit retina. Science. 191:204–205. 1976.
ArticleYazulla S., Studholme KM., Vitorica J., de Blas AL. Immunocy-tochemical localization of GABAA receptors in goldfish and chicken retinas. J Comp Neurol. 280:15–26. 1989.Zanker JM., Srinivasan MV., Egelhaaf M. Speed tuning in elementary motion detectors of the correlation type. Biol Cybern. 80:109–116. 1999.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Role of the Pattern Edge in Goldfish Visual Motion Detection
- Reversal of Optic Disc Cupping in Adults with Advanced Glaucoma
- Intraocular Pressure Changes After Gas Injection into the Rabbit Vitreous Cavity
- Effect of Ocular Hypotensives and Honan Intraocular Pressure Reducer on Ocular Tension after Retrobulbar Anesthesia
- Effects of taurine on cadmium exposure in muscle, gill, and bone tissues of Carassius auratus