J Adv Prosthodont.
2013 Feb;5(1):2-8. 10.4047/jap.2013.5.1.2.
Sterilization effect of atmospheric pressure non-thermal air plasma on dental instruments
- Affiliations
-
- 1Department of Prosthodontics, School of Dentistry, Pusan National University, Yangsan, Republic of Korea. jeonyc@paran.com
- 2Department of Maxillofacial Prosthodontics, Cleveland Clinic, Head and Neck Institute, Cleveland, Ohio, USA.
- KMID: 2284765
- DOI: http://doi.org/10.4047/jap.2013.5.1.2
Abstract
- PURPOSE
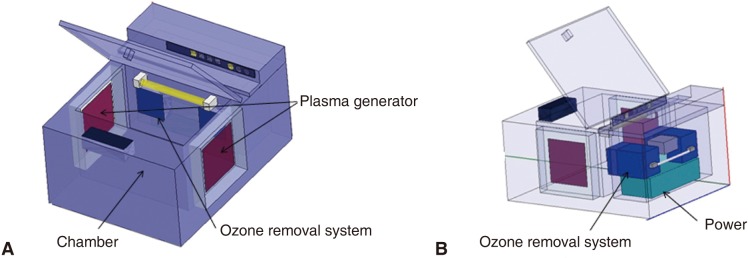
Autoclaves and UV sterilizers have been commonly used to prevent cross-infections between dental patients and dental instruments or materials contaminated by saliva and blood. To develop a dental sterilizer which can sterilize most materials, such as metals, rubbers, and plastics, the sterilization effect of an atmospheric pressure non-thermal air plasma device was evaluated.
MATERIALS AND METHODS
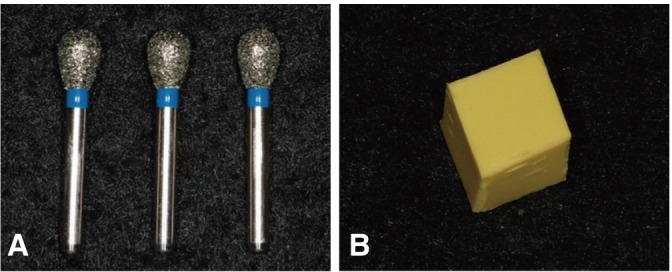
After inoculating E. coli and B. subtilis the diamond burs and polyvinyl siloxane materials were sterilized by exposing them to the plasma for different lengths of time (30, 60, 90, 120, 180 and, 240 seconds). The diamond burs and polyvinyl siloxane materials were immersed in PBS solutions, cultured on agar plates and quantified by counting the colony forming units. The data were analyzed using one-way ANOVA and significance was assessed by the LSD post hoc test (alpha=0.05).
RESULTS
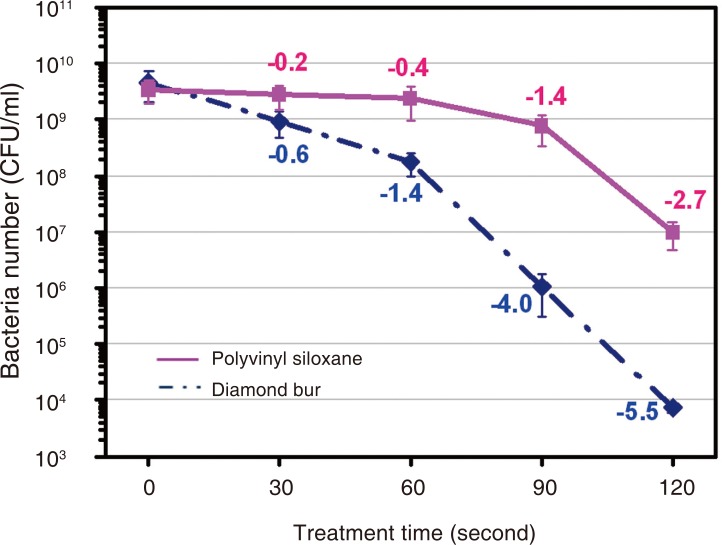
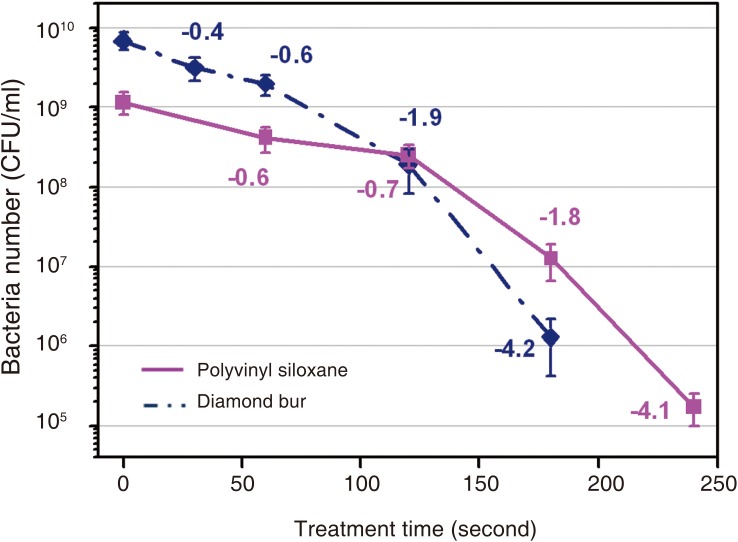
The device was effective in killing E. coli contained in the plasma device compared with the UV sterilizer. The atmospheric pressure non-thermal air plasma device contributed greatly to the sterilization of diamond burs and polyvinyl siloxane materials inoculated with E. coli and B. subtilis. Diamond burs and polyvinyl siloxane materials inoculated with E. coli was effective after 60 and 90 seconds. The diamond burs and polyvinyl siloxane materials inoculated with B. subtilis was effective after 120 and 180 seconds.
CONCLUSION
The atmospheric pressure non-thermal air plasma device was effective in killing both E. coli and B. subtilis, and was more effective in killing E. coli than the UV sterilizer.
MeSH Terms
Figure
Reference
-
1. Choi HN, Bae HS, Cho YS. Literature review of dental infection control in Korea. J Dent Hyg Sci. 2010; 10:199–209.2. Hutchings ML, Vandewalle KS, Schwartz RS, Charlton DG. Immersion disinfection of irreversible hydrocolloid impressions in pH-adjusted sodium hypochlorite. Part 2: Effect on gypsum casts. Int J Prosthodont. 1996; 9:223–229. PMID: 8957856.3. Leung RL, Schonfeld SE. Gypsum casts as a potential source of microbial cross-contamination. J Prosthet Dent. 1983; 49:210–211. PMID: 6572259.
Article4. McNeill MR, Coulter WA, Hussey DL. Disinfection of irreversible hydrocolloid impressions: a comparative study. Int J Prosthodont. 1992; 5:563–567. PMID: 1339136.5. Runnells RR. An overview of infection control in dental practice. J Prosthet Dent. 1988; 59:625–629. PMID: 3290458.
Article6. Jennings KJ, Samaranayake LP. The persistence of microorganisms on impression materials following disinfection. Int J Prosthodont. 1991; 4:382–387. PMID: 1811634.7. Morfill GE, Shimizu T, Steffes B, Schmidt HU. Nosocomial infections-a new approach towards preventive medicine using plasmas. New J Phys. 2009; 11:115019.
Article8. Samaranayake L. Essential Microbiology for dentistry. 2007. 3rd ed. Churchill Livingstone: p. 385–412.9. Ministry of Health and Welfare Notification No. 2010-61. Guideline of sterilization of appliance and article at medical institution. 2010. 8. 13. enactment.10. Moreau M, Orange N, Feuilloley MG. Non-thermal plasma technologies: new tools for bio-decontamination. Biotechnol Adv. 2008; 26:610–617. PMID: 18775485.
Article11. Lee K, Paek KH, Ju WT, Lee Y. Sterilization of bacteria, yeast, and bacterial endospores by atmospheric-pressure cold plasma using helium and oxygen. J Microbiol. 2006; 44:269–275. PMID: 16820756.12. Dobrynin D, Fridman G, Friedman G, Fridman A. Physical and biological mechanisms of direct plasma interaction with living tissue. New J Phys. 2009; 11:115020.
Article13. Fridman G, Friedman G, Gutsol A, Shekhter AB, Vasilets VN, Fridman A. Applied plasma Medicine. Plasma Process Polym. 2008; 5:503–533.
Article14. McCombs GB, Darby ML. New discoveries and directions for medical, dental and dental hygiene research: low temperature atmospheric pressure plasma. Int J Dent Hyg. 2010; 8:10–15. PMID: 20096076.
Article15. Sladek REJ, Stoffels E. Deactivation of Escherichia coli by the plasma needle. J Phys D Appl Phys. 2005; 38:1716–1721.16. Lee HW, Kim GJ, Kim JM, Park JK, Lee JK, Kim GC. Tooth bleaching with nonthermal atmospheric pressure plasma. J Endod. 2009; 35:587–591. PMID: 19345811.
Article17. Samaranayake LP, Hunjan M, Jennings KJ. Carriage of oral flora on irreversible hydrocolloid and elastomeric impression materials. J Prosthet Dent. 1991; 65:244–249. PMID: 2051359.
Article18. Smith A, Creanor S, Hurrell D, Bagg J, McCowan M. Management of infection control in dental practice. J Hosp Infect. 2009; 71:353–358. PMID: 19162375.
Article19. Seo YS, Mohamed AAH, Woo KC, Lee HW, Lee JK, Kim KT. Comparative studies of atmospheric pressure plasma characteristics between He and Ar working gases for sterilization. IEEE Trans Plasma Sci IEEE Nucl Plasma Sci Soc. 2010; 38:1954–1962.
Article20. Iza F, Kim GJ, Lee SM, Lee JK, Walsh JL, Zhang YT, Kong MG. Microplasmas: Sources, particle kinetics, and biomedical applications. Plasma Process Polym. 2008; 5:322–344.
Article21. Laroussi M. Low Temperature Plasma-Based Sterilization: Overview and state-of-the Art. Plasma Process Polym. 2005; 2:391–400.22. Fridman G, Brooks AD, Balasubramanian M, Fridman A, Gutsol A, Vasilets VN, Ayan H, Friedman G. Comparison of direct and indirect effects of non-thermal atmospheric-pressure plasma on bacteria. Plasma Process Polym. 2007; 4:370–375.
Article23. Laroussi M. Nonthermal decontamination of biological media by atmospheric-pressure plasma: review, analysis, and prospects. IEEE Trans Plasma Sci IEEE Nucl Plasma Sci Soc. 2002; 30:1409–1415.24. Suzuki T, Oizumi M, Furuya J, Okamoto Y, Rosenstiel SF. Influence of ozone on oxidation of dental alloys. Int J Prosthodont. 1999; 12:179–183. PMID: 10371921.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Non-Thermal Atmospheric-Pressure Plasma Possible Application in Wound Healing
- Evaluation of the safety of non-thermal atmospheric-pressure plasma in hairless mouse tissues
- Inactivation Efficacy of a Non-thermal Atmospheric Pressure Plasma Generator against Mycobacterium tuberculosis
- New Conversing Technology; Plasma Medicine
- Anti-inflammatory effect of non-thermal atmospheric pressure plasma for periodontitis treatment: in vitro pilot study