Allergy Asthma Respir Dis.
2015 Jul;3(4):288-293. 10.4168/aard.2015.3.4.288.
Effectiveness of premedication and rapid desensitization in hypersensitivity to L-asparaginase
- Affiliations
-
- 1Department of Pediatrics, The Catholic University of Korea College of Medicine, Seoul, Korea. tibia78@nate.com
- KMID: 2218659
- DOI: http://doi.org/10.4168/aard.2015.3.4.288
Abstract
- PURPOSE
L-asparaginase is a crucial chemotherapeutic agent for the treatment of acute lymphoblastic leukemia. However, hypersensitivity to L-asparaginase is common which limits its clinical use.
METHODS
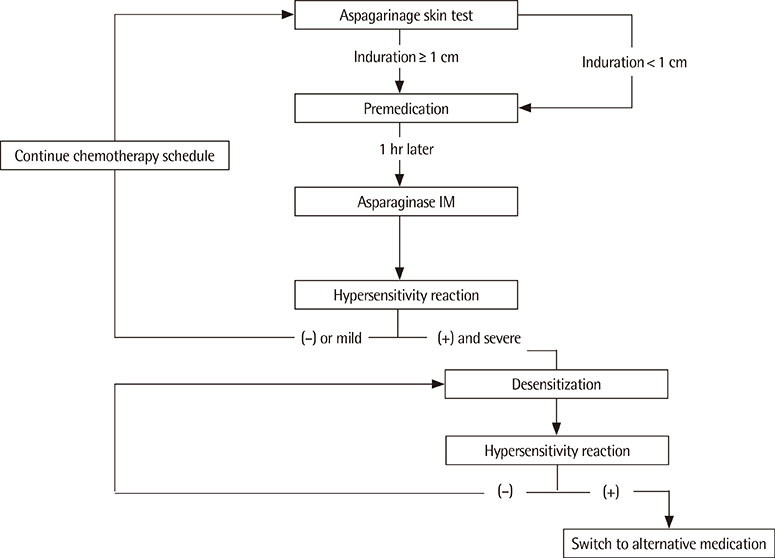
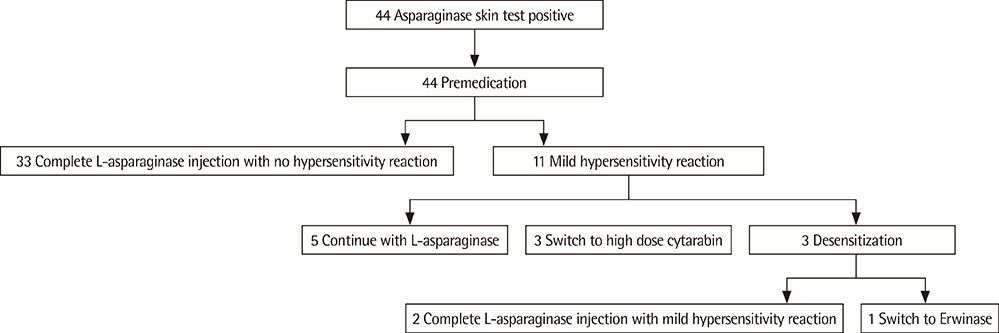
We performed 44 cases of premedication and 3 cases of desensitization in 16 patients with hypersensitivity to L-asparaginase.
RESULTS
With premedication, 33 cases completed L-asparaginase injection with no hypersensitivity reactions. Eleven cases showed mild hypersensitivity reactions, such as urticaria. Desensitization was performed in 3 cases: in 2 cases, desensitization was successful, and in 1 case the medication was switched to Erwinia asparaginase.
CONCLUSION
Premedication and desensitization appear to be useful in helping patients receive desired doses of L-asparaginase in pediatric patients with acute lymphoblastic leukemia.
Keyword
MeSH Terms
Figure
Reference
-
1. Graham ML. Pegaspargase: a review of clinical studies. Adv Drug Deliv Rev. 2003; 55:1293–1302.
Article2. Fu CH, Sakamoto KM. PEG-asparaginase. Expert Opin Pharmacother. 2007; 8:1977–1984.
Article3. Woo MH, Hak LJ, Storm MC, Evans WE, Sandlund JT, Rivera GK, et al. Anti-asparaginase antibodies following E. coli asparaginase therapy in pediatric acute lymphoblastic leukemia. Leukemia. 1998; 12:1527–1533.
Article4. Narta UK, Kanwar SS, Azmi W. Pharmacological and clinical evaluation of L-asparaginase in the treatment of leukemia. Crit Rev Oncol Hematol. 2007; 61:208–221.
Article5. Pratt CB, Simone JV, Zee P, Aur RJ, Johnson WW. Comparison of daily versus weekly L-asparaginase for the treatment of childhood acute leukemia. J Pediatr. 1970; 77:474–483.
Article6. Cheung NK, Chau IY, Coccia PF. Antibody response to Escherichia coli L-asparaginase. Prognostic significance and clinical utility of antibody measurement. Am J Pediatr Hematol Oncol. 1986; 8:99–104.7. Pratt CB, Choi SI, Holton CP. Low-dosage asparaginase treatment of childhood acute lymphocytic leukemia. Am J Dis Child. 1971; 121:406–409.
Article8. Haskell CM, Canellos GP. l-asparaginase resistance in human leukemia: asparagine synthetase. Biochem Pharmacol. 1969; 18:2578–2580.9. Soyer OU, Aytac S, Tuncer A, Cetin M, Yetgin S, Sekerel BE. Alternative algorithm for L-asparaginase allergy in children with acute lymphoblastic leukemia. J Allergy Clin Immunol. 2009; 123:895–899.
Article10. Hak LJ, Relling MV, Cheng C, Pei D, Wang B, Sandlund JT, et al. Asparaginase pharmacodynamics differ by formulation among children with newly diagnosed acute lymphoblastic leukemia. Leukemia. 2004; 18:1072–1077.
Article11. Wang B, Relling MV, Storm MC, Woo MH, Ribeiro R, Pui CH, et al. Evaluation of immunologic crossreaction of antiasparaginase antibodies in acute lymphoblastic leukemia (ALL) and lymphoma patients. Leukemia. 2003; 17:1583–1588.
Article12. Castells MC, Tennant NM, Sloane DE, Hsu FI, Barrett NA, Hong DI, et al. Hypersensitivity reactions to chemotherapy: outcomes and safety of rapid desensitization in 413 cases. J Allergy Clin Immunol. 2008; 122:574–580.
Article13. Bonno M, Kawasaki H, Hori H, Umemoto M, Komada Y, Sakurai M. Rapid desensitization for L-asparaginase hypersensitivity. J Allergy Clin Immunol. 1998; 101(4 Pt 1):571–572.
Article14. Sampson HA, Munoz-Furlong A, Campbell RL, Adkinson NF Jr, Bock SA, Branum A, et al. Second symposium on the definition and management of anaphylaxis: summary report: Second National Institute of Allergy and Infectious Disease/Food Allergy and Anaphylaxis Network symposium. J Allergy Clin Immunol. 2006; 117:391–397.
Article15. Common terminology criteriafor adverse events (CTCAE). Version 4.0 [Internet]. Bethesda (MD): U.S. Department of Health and Human Services, National Institutes of Health, National Cancer Institute;c2010. cited 2014 Aug 3. Available from: http://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03_2010-06-14_QuickReference_5x7.pdf.16. Johansson SG, Hourihane JO, Bousquet J, Bruijnzeel-Koomen C, Dreborg S, Haahtela T, et al. A revised nomenclature for allergy. An EAACI position statement from the EAACI nomenclature task force. Allergy. 2001; 56:813–824.
Article17. Aronson JK, Ferner RE. Clarification of terminology in drug safety. Drug Saf. 2005; 28:851–870.
Article18. Limsuwan T, Castells MC. Outcomes and safety of rapid desensitization for chemotherapy hypersensitivity. Expert Opin Drug Saf. 2010; 9:39–53.
Article19. Tallal L, Tan C, Oettgen H, Wollner N, McCarthy M, Helson L, et al. E. coli L-asparaginase in the treatment of leukemia and solid tumors in 131 children. Cancer. 1970; 25:306–320.
Article20. Clarkson B, Krakoff I, Burchenal J, Karnofsky D, Golbey R, Dowling M, et al. Clinical results of treatment with E. coli L-asparaginase in adults with leukemia, lymphoma, and solid tumors. Cancer. 1970; 25:279–305.
Article21. Moghrabi A, Levy DE, Asselin B, Barr R, Clavell L, Hurwitz C, et al. Results of the Dana-Farber Cancer Institute ALL Consortium Protocol 95-01 for children with acute lymphoblastic leukemia. Blood. 2007; 109:896–904.
Article22. Silverman LB, Stevenson K, Neuberg D, O'Brien J, Supko J, Sallan SE. Intravenous PEG asparaginase during remission induction for childhood ALL [abstract]. Blood. 2006; 108:1854.
Article23. Raetz EA, Salzer WL. Tolerability and efficacy of L-asparaginase therapy in pediatric patients with acute lymphoblastic leukemia. J Pediatr Hematol Oncol. 2010; 32:554–563.
Article24. Billett AL, Carls A, Gelber RD, Sallan SE. Allergic reactions to Erwinia asparaginase in children with acute lymphoblastic leukemia who had previous allergic reactions to Escherichia coli asparaginase. Cancer. 1992; 70:201–206.
Article25. Salzer WL, Asselin B, Supko JG, Devidas M, Kaiser NA, Plourde P, et al. Erwinia asparaginase achieves therapeutic activity after pegaspargase allergy: a report from the Children's Oncology Group. Blood. 2013; 122:507–514.
Article26. Muller HJ, Boos J. Use of L-asparaginase in childhood ALL. Crit Rev Oncol Hematol. 1998; 28:97–113.27. Woo MH, Hak LJ, Storm MC, Sandlund JT, Ribeiro RC, Rivera GK, et al. Hypersensitivity or development of antibodies to asparaginase does not impact treatment outcome of childhood acute lymphoblastic leukemia. J Clin Oncol. 2000; 18:1525–1532.
Article28. Larson RA, Fretzin MH, Dodge RK, Schiffer CA. Hypersensitivity reactions to L-asparaginase do not impact on the remission duration of adults with acute lymphoblastic leukemia. Leukemia. 1998; 12:660–665.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Successful desensitization of a patient with albumin hypersensitivity
- Hypersensitivity Reactions to Antineoplastic Agents: Characteristics and Premedication Strategies
- Successful desensitization for vancomycin hypersensitivity
- Rapid desensitization to isoniazid and rifampin in an adolescent with active pulmonary tuberculosis
- A case of rapid desensitization for rituximab-induced delayed hypersensitivity reaction