J Korean Ophthalmol Soc.
2009 Oct;50(10):1531-1538. 10.3341/jkos.2009.50.10.1531.
Photopic Negative Response (PhNR) in Normal Subjects
- Affiliations
-
- 1Department of Ophthalmology, College of Medicine, Soonchunhyang University, Bucheon, Korea. yhohn@schbc.ac.kr
- 2Department of Ophthalmology, College of Medicine, Soonchunhyang University, Gumi, Korea.
- 3Department of Ophthalmology, Gangnam Sacred Heart Hospital, College of Medicine, Hallym University, Seoul, Korea.
- KMID: 2212772
- DOI: http://doi.org/10.3341/jkos.2009.50.10.1531
Abstract
- PURPOSE
To evaluate the nature of photopic negative response (PhNR) in normal subjects.
METHODS
The electroretinogram (ERG) waves were recorded in 52 eyes of 26 normal subjects with UTAS E-3000(R) (LKC Technologies Inc., Gaithersberg, MD, USA). Photopic cone response was obtained for analysis from a white flash with white background illumination (group 1) and a red flash with blue background illumination (group 2), after stimulations ranging from 0.007674 cd.s/m2 (-25dB) to 7.736 cd.s/m2 (5dB) with a 5dB interval.
RESULTS
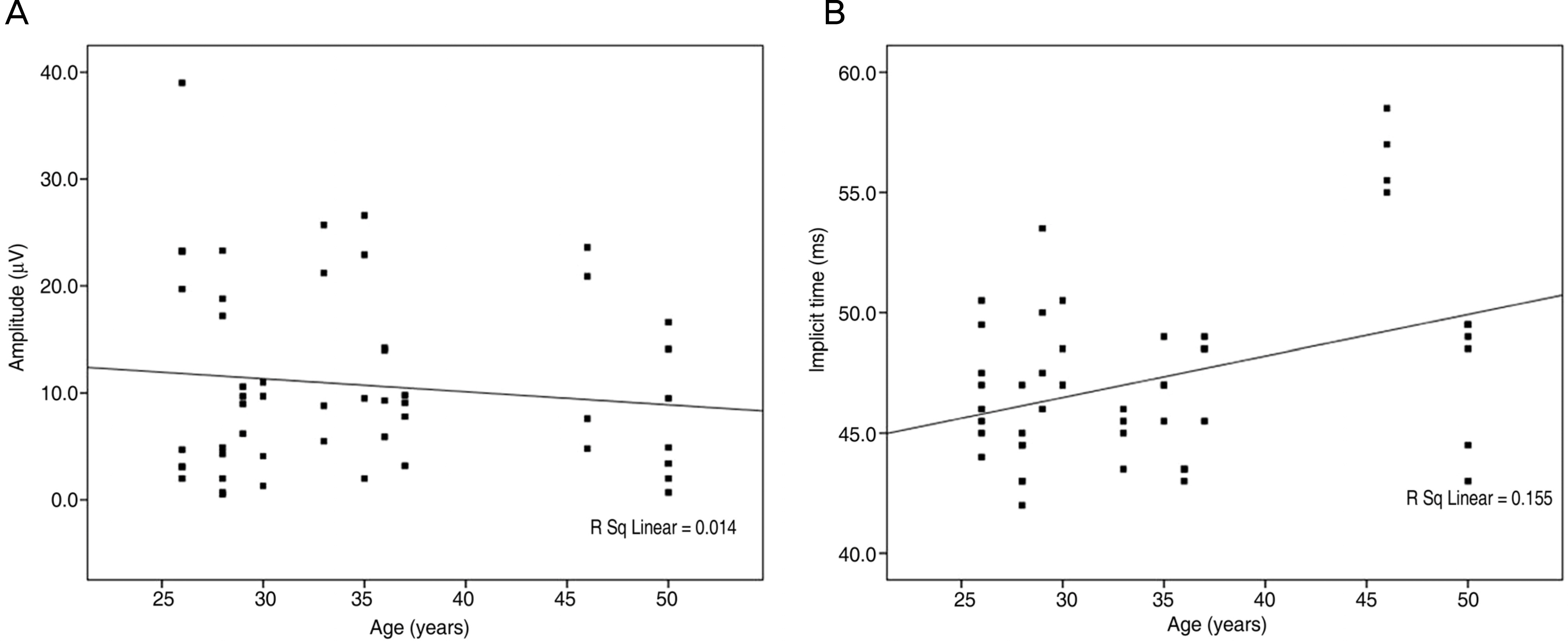
PhNRs were observed in all 52 eyes in group 1 stimulated with the white flash at 0.9933 cd.s/m2 (-4dB). PhNRs were also observed in all 52 eyes in group 2 at 2.4297 cd.s/m2 (0dB) after stimulation with the red flash. There was correlation between the amplitudes of PhNR and intensity of stimuli (p<0.001). Implicit times of PhNR were correlated with age in both groups, but amplitudes decreased with age in group 1.
CONCLUSIONS
The amplitudes of PhNR were correlated with intensity of stimuli and age had an influence on PhNR. Standardization of examination conditions is necessary when recording PhNR.
Figure
Cited by 1 articles
-
The Role of Electroretinography in Assessing the Progression of Diabetic Retinopathy
Su Eun Park, Hae Jung Sun, Hyun Joon Lee, Tae Kwann Park, Young-Hoon Ohn
J Korean Ophthalmol Soc. 2010;51(5):693-699. doi: 10.3341/jkos.2010.51.5.693.
Reference
-
References
1. Marmer MF, Arden GB, Nisson SE, Zrenner E. Standard for clinical eletroretinography. Arch Ophthalmol. 1989; 107:816–9.2. Marmer MF, Zrenner E. Standard for clinical electroretinography (1994 update). Doc Ophthalmol. 1995; 89:199–210.
Article3. Mammer MF. An updated standard for clinical electroretinography. Arch ophthalmol. 1995; 113:1375–6.
Article4. Mammer MF. The ERG is alive and well. Arch Ophthalmol. 1995; 113:1371–2.
Article5. Bush RA, Sieving PA. A proxima retinal component in the primate photopic ERG a-wave. Invest Ophthalmol Vis Sci. 1994; 35:635–45.6. Sieving PA, Murayama K, Naarendorp F. Push-pull model of the primate photopic electroretinogram: a role for hyperpolarizing neurons in shaping the b-wave. Vis Neurosci. 1994; 11:519–32.
Article7. Drasdo N, Aldedasi YH, Chiti Z, et al. The s-cone PhNR and pattern ERG in primary open angle glaucoma. Invest Ophthalmol Vis Sci. 2001; 42:1266–72.8. Viswanathan S, Frishman LJ, Robson JG, Walters JW. The photopic negative response of the flash electroretinogram in primary open angle glaucoma. Invest Ophthalmol Vis Sci. 2001; 42:514–22.9. Colotto A, Falsini B, Salgarello T, et al. Photopic negative response of the human ERG: losses associated with glaucomatous damage. Invest Ophthalmol Vis Sci. 2000; 41:2205–11.10. Machida S, Gotoh Y, Tanaka M, Tazawa Y. Predominant loss of the photopic negative response in central retinal artery occlusion. Am J Ophthalmol. 2004; 137:938–40.
Article11. Peyman GA, Sanders DR, Galdberg NR. Principles and practice of ophthalmology. Philadelphia: WB Saunders Co.;1980. p. 857.12. Riggs LA. Continuous and reproducible records of electrical activity of the human retina. Proc Soc Exp Biol Med. 1941; 48:204–7.13. Viswanathan S, Frishman LJ, Robson JG, et al. The photopic negative response of the macaque electroretingram: reduction by experimental glaucoma. Invest Ophthalmol Vis Sci. 1999; 40:1124–36.14. Drasdo N, Aldebasi YH, Mortlock KE, et al. Ocular optics, electroretinography and primary open angle glaucoma. Ophthalmic Physiol Opt. 2002; 22:455–62.
Article15. Miyata K, Nakamura M, Kondo M, et al. Reduction of oscillatory potentials and photopic negative response in patients with autosomal dominant optic atrophy with OPA1 mutations. Invest Ophthalmol Vis Sci. 2007; 48:820–4.16. Chen H, Wu D, Huang S, Yan H. The photopic negative response of the flash electroretinogram in retinal vein occlusion. Doc Ophthalmol. 2006; 113:53–9.
Article17. Cursiefen C, Korth M, Horn FK. The negative response of the flash electroretinogram in glaucoma. Doc Ophthalmol. 2001; 103:1–12.18. Aguilar M, Stiles WS. Saturation of the rod mechanism of the retina at the high levels of stimulation. Optica Acta. 1954; 1:59–65.19. Danias J, Lee KC, Zamora MF, et al. Quantitative analysis of retinal ganglion cell (RGC) loss in aging DBA/1NNia glaucomatous mice: Comparison with RGC loss in aging C57/BL6 mice. Invest Ophthlmol Vis Sci. 2003; 44:5151–62.20. Neufeld AH, Gachie EN. The inherent, age-dependent loss of retinal ganglion cells is related to the lifespan of the species. Neurobiol Aging. 2003; 24:167–72.
Article21. Harman A, Abrahams B, Moore S, Hoskin R. Neuronal density in the human retinal ganglion cell layer from 16-77 years. Anat Rec. 2000; 260:124–31.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Clinical Applications of Photopic Negative Response (PhNR) for the Treatment of Glaucoma and Diabetic Retinopathy
- ON and OFF Responses of the Electroretinogram in Patients with Glaucoma
- The Role of Electroretinography in Assessing the Progression of Diabetic Retinopathy
- Blue-on-Green Flash Induces Maximal Photopic Negative Response and Oscillatory Potential and Serves as a Diagnostic Marker for Glaucoma in Rat Retina
- Photopic ON- and OFF- Responses in Korean Normal Subjects