J Korean Med Assoc.
2006 Jan;49(1):78-85. 10.5124/jkma.2006.49.1.78.
Understanding the Drug-Drug Interaction
- Affiliations
-
- 1Department of Pharmacology, The Catholic University of Korea, College of Medicine, Korea. yimds@catholic.ac.kr
- KMID: 2184639
- DOI: http://doi.org/10.5124/jkma.2006.49.1.78
Abstract
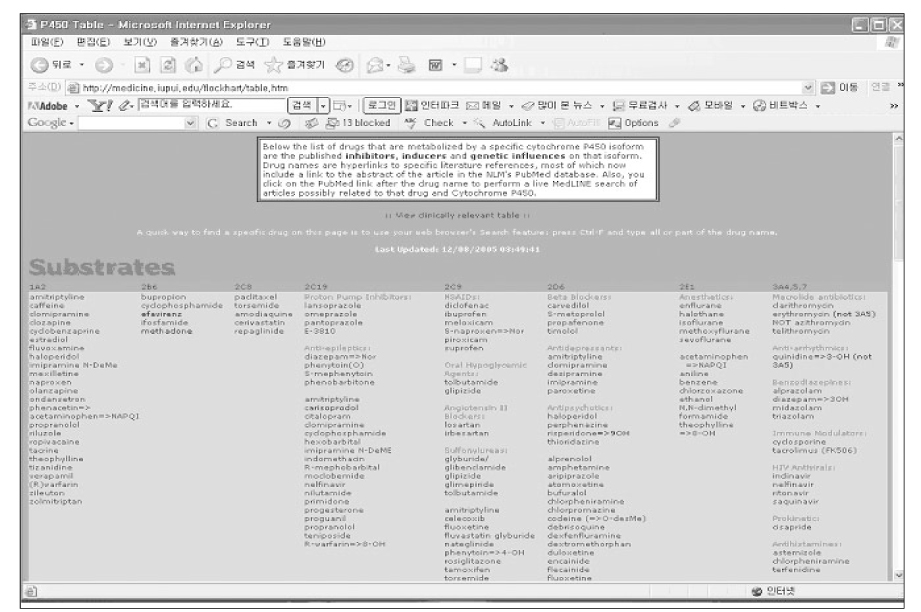
- Drug-drug interaction (DDI) is defined as a change in effect or safety of a drug by another co-administered drug. The fact that more than half of the market withdrawal cases for the past ten years was caused by potentially fatal DDI's demonstrates its clinical importance. The mechanism of DDI can be categorized into pharmacokinetic and pharmacodynamic interactions. Most of the clinically important drug interactions are caused by inhibition or induction of oxidative metabolism by cytochrome P450 (CYP) isozymes. Recent researches are also focusing on drug transporter interactions as another significant factor underlying DDI's. It is hard to prevent unexpected or rare DDI's. However, most of the cases of DDI occur from an erroneous prescription of drugs that are already known to result in deleterious interactions. To avoid such well-established DDI's, physicians are first recommended to utilize hands-on summary tables for CYP substrates before prescribing. It should also be remembered that old age, polypharmacy and damaged hepatic or renal function are risk factors of DDI as well as adverse drug reactions. Moreover, patients treated with drugs with a narrow therapeutic index (immunosuppressants, antiarrhythmics, anticoagulants, digoxin, theophylline etc) deserve a special consideration when their prescriptions are changed. In Korea, the clinical significance of DDI has been underemphasized. The fundamental prescription to this old prescription habit is to teach medical students and physicians clinical pharmacology and therapeutics, which have long been neglected in Korea.
Keyword
MeSH Terms
-
Anticoagulants
Cytochrome P-450 Enzyme System
Digoxin
Drug Interactions
Drug-Related Side Effects and Adverse Reactions
Humans
Isoenzymes
Korea
Metabolism
Pharmacology, Clinical
Polypharmacy
Prescriptions
Product Recalls and Withdrawals
Risk Factors
Students, Medical
Theophylline
Anticoagulants
Cytochrome P-450 Enzyme System
Digoxin
Isoenzymes
Theophylline
Figure
Reference
-
1. Monahan BP, Ferguson CL, Killeavy ES, Lloyd BK, Troy J, Cantilena LR Jr. Torsades de pointes occurring in association with terfenadine use. JAMA. 1990. 264:2788–2790.
Article2. Lazarou J, Pomeranz BH, Corey PN. Incidence of adverse drug reactions in hospitalized patients: a meta-analysis of prospective studies. JAMA. 1998. 279:1200–1205.
Article3. Ernst FR, Grizzle AJ. Drug-related morbidity and mortality: updating the cost-of-illness model. J Am Pharm Assoc. 2001. 41:192–199.
Article4. Jankel CA, Fitterman LK. Epidemiology of drug-drug interactions as a cause of hospital admissions. Drug Saf. 1993. 9:51–59.
Article5. Juurlink DN, Mamdani M, Kopp A, Laupacis A, Redelmeier DA. Drug-drug interactions among elderly patients hospitalized for drug toxicity. JAMA. 2003. 289:1652–1658.
Article6. Malone DC, Abarca J, Hansten PD, Grizzle AJ, Armstrong EP, Lipton RB, et al. Identification of serious drug-drug interactions: results of the partnership to prevent drug-drug interactions. J Am Pharm Assoc. 2004. 44:142–151.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Inhibition of Cytochrome P450 Enzymes by Drugs—Molecular Basis and Practical Applications
- Basic Principles of Drug Interaction
- Antidepressants and Related Drug Interactions
- Drug interaction: focusing on response surface models
- Drug-drug Interaction between Psychotropic Medications and Medications Used in COVID-19: Comparison of Online Databases