Endocrinol Metab.
2011 Sep;26(3):210-217. 10.3803/EnM.2011.26.3.210.
Somatostatin Inhibits Gonadotropin Releasing Hormone Neuronal Activities in Juvenile Mice
- Affiliations
-
- 1Department of Oral Physiology, School of Dentistry & Institute of Oral Bioscience, Chonbuk National University, Jeonju, Korea. skhan@jbnu.ac.kr
- KMID: 2169171
- DOI: http://doi.org/10.3803/EnM.2011.26.3.210
Abstract
- BACKGROUND
The gonadotropin releasing hormone (GnRH) neurons perform a pivotal function in the central regulation of fertility. Somatostatin (SST) is an important neuromodulatory peptide in the central nervous system and alters neuronal activities via G protein- coupled SST receptors. A number of studies have shown that SST modulates the reproductive axis at the hypothalamic level. However, the precise action mechanisms of SST and related receptor subtypes have yet to be fully understood. In this study, we evaluated the direct effects of SST on GnRH neurons in juvenile mice.
METHODS
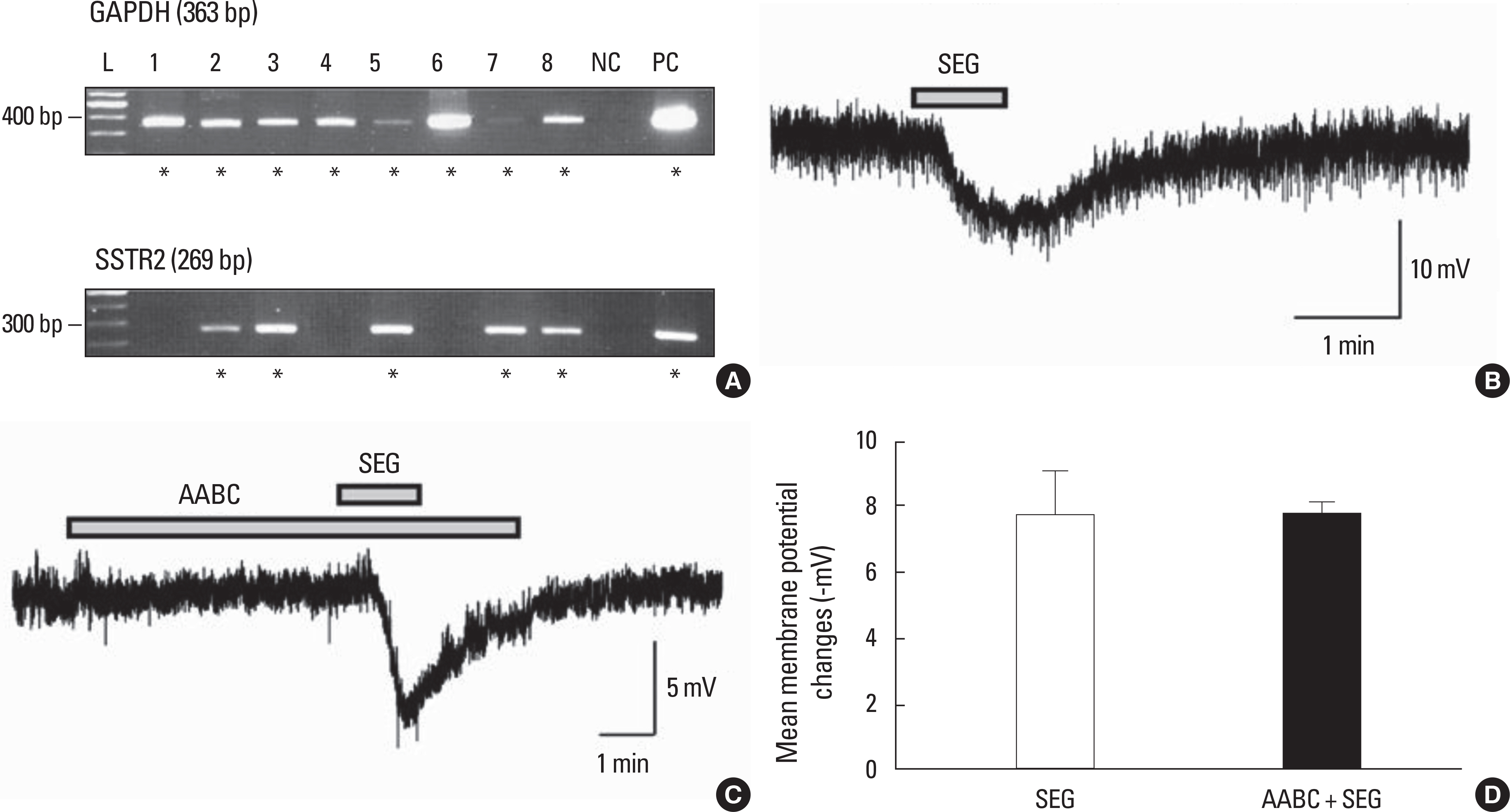
Juvenile (postnatal days, < PND 30) GnRH-GFP transgenic mice expressing green fluorescent protein were used in this study. Acute coronal brain slices containing the preoptic area were prepared and all identified GnRH neurons were recorded using the gramicidin perforated-patch clamp technique; type II SST receptor (SSTR2) mRNA expression was evaluated via single cell reverse transcription-polymerase chain reaction (RT-PCR).
RESULTS
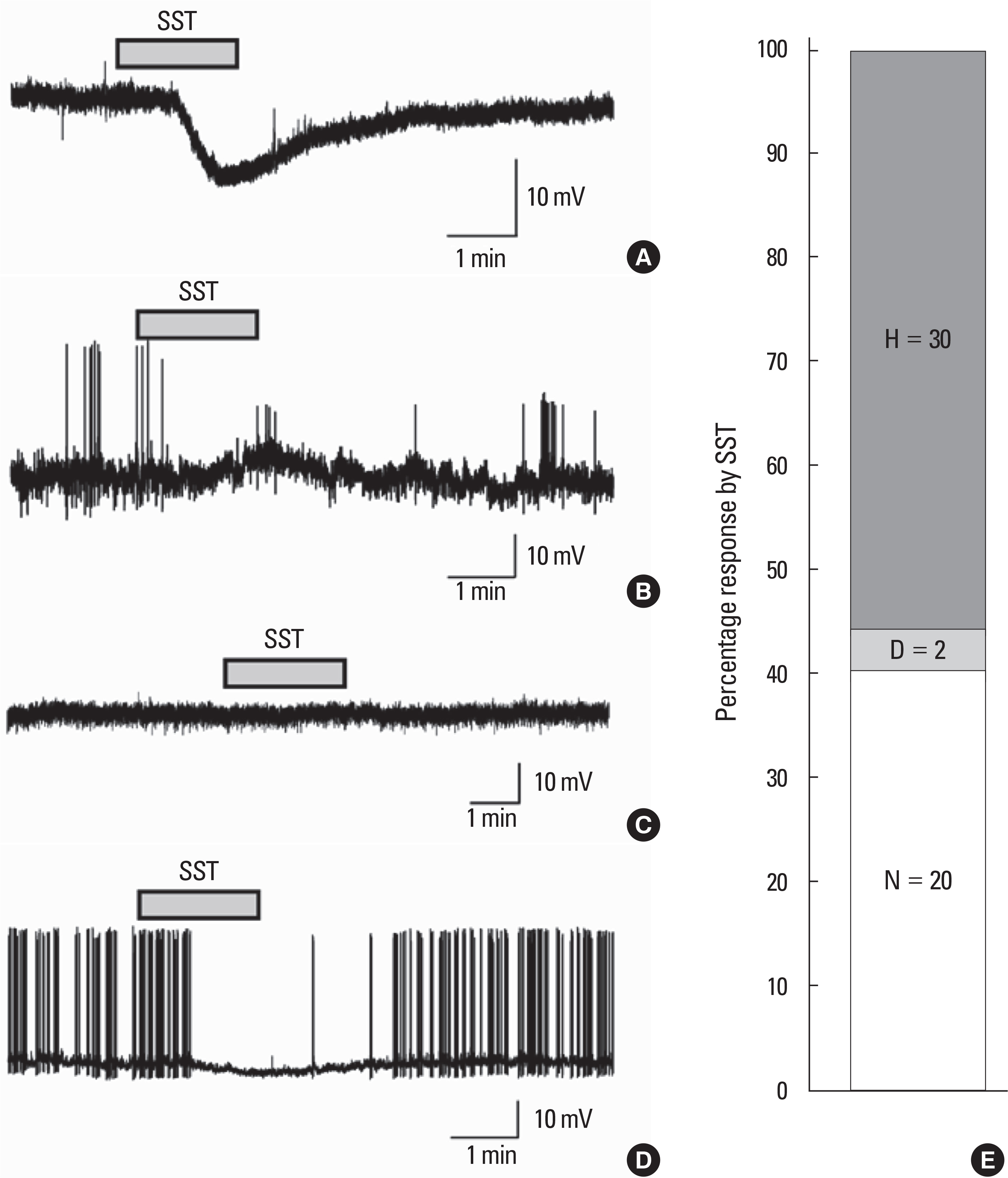
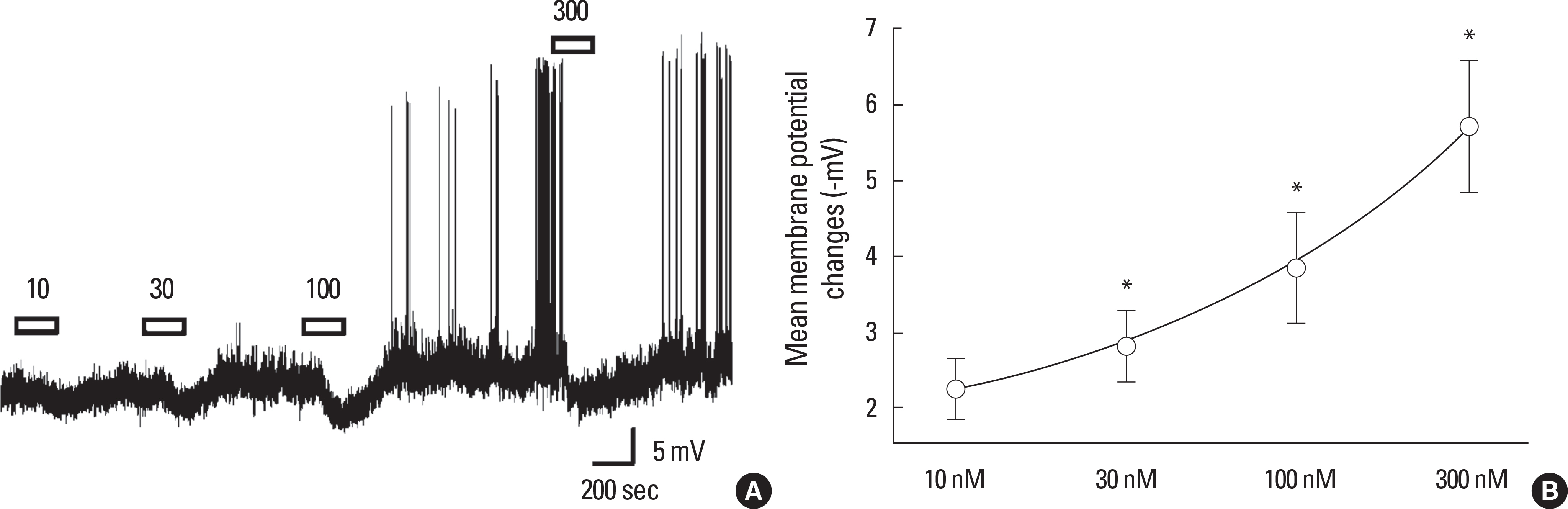
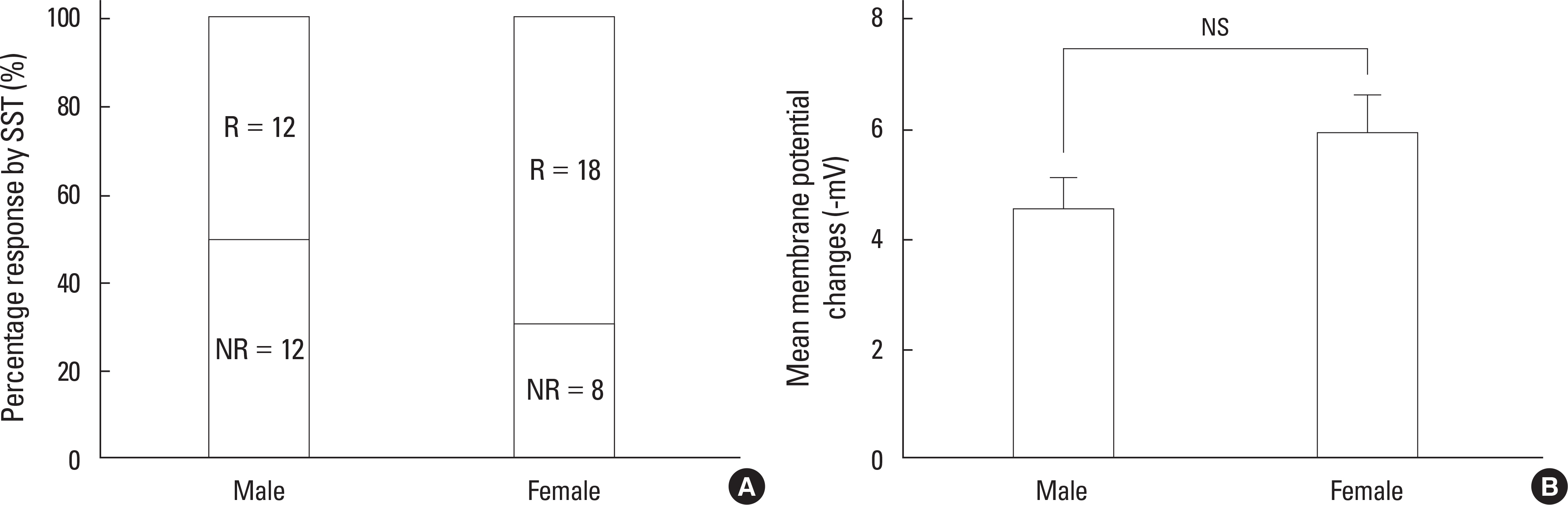
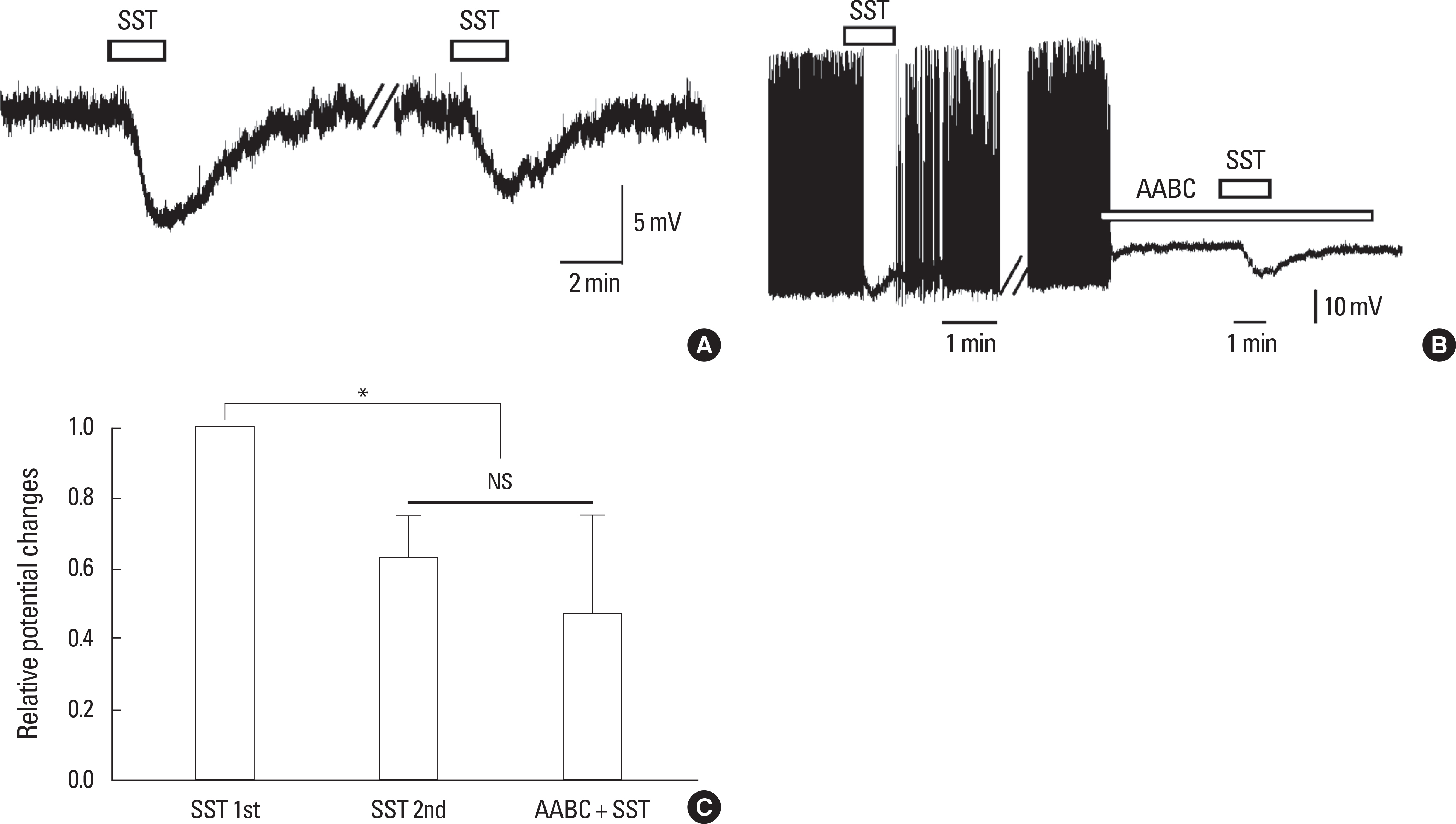
SST caused membrane hyperpolarization, depolarization, no response, or membrane hyperpolarization with a reduction of action potential. Most (57.7%, 30/52) of the GnRH neurons tested were hyperpolarized by SST and this SST-induced hyperpolarization was found to be concentration-dependent. The percentage of responses, membrane potential changes (MPC), and resting membrane potential (RMP) by SST were not significantly different in juvenile male and female GnRH neurons. The SST-induced hyperpolarization was maintained in the presence of tetrodotoxin (TTX), a sodium channel blocker, and an amino acid blocking cocktail (AABC) containing AP-5 (NMDA receptor antagonist), CNQX (non-NMDA glutamate receptor antagonist), picrotoxin (GABAA receptor antagonist), and strychnine (glycine receptor antagonist). SSTR2 mRNA was expressed on 10 (38%) among 26 GnRH neurons. Seglitide, an SSTR2 agonist, mimicked this SST-induced hyperpolarization (11/23 47.8%) and this response was maintained in the presence of TTX and AABC.
CONCLUSION
Our data show that SST can exert potent inhibitory action against GnRH neuronal excitability via SSTR2 activation in juvenile mice.
Keyword
MeSH Terms
-
6-Cyano-7-nitroquinoxaline-2,3-dione
Action Potentials
Animals
Brain
Central Nervous System
Female
Fertility
Gonadotropin-Releasing Hormone
Gonadotropins
Gramicidin
Humans
Male
Membrane Potentials
Membranes
Mice
Mice, Transgenic
Neurons
Peptides, Cyclic
Picrotoxin
Preoptic Area
Receptors, Glutamate
RNA, Messenger
Sodium Channels
Somatostatin
Strychnine
Tetrodotoxin
Axis, Cervical Vertebra
6-Cyano-7-nitroquinoxaline-2,3-dione
Gonadotropin-Releasing Hormone
Gonadotropins
Gramicidin
Peptides, Cyclic
Picrotoxin
RNA, Messenger
Receptors, Glutamate
Sodium Channels
Somatostatin
Strychnine
Tetrodotoxin
Figure
Reference
-
1. Herbison AE. GnRH neuron. Henry H, Norman A, editors. Encyclopedia of hormones. p. 171–177. San Diego: Academic Press;2003.2. Yu WH, Kimura M, McCann SM. Effect of somatostatin on the release of gonadotropins in male rats. Proc Soc Exp Biol Med. 214:83–86. 1997.
Article3. Starcevic V, Milosevic V, Brkic B, Severs WB. Somatostatin affects morphol-ogy and secretion of pituitary luteinizing hormone (LH) cells in male rats. Life Sci. 70:3019–3027. 2002.
Article4. Prelevic GM, Wurzburger MI, Balint-Peric L, Nesic JS. Inhibitory effect of sandostatin on secretion of luteinising hormone and ovarian steroids in polycystic ovary syndrome. Lancet. 336:900–903. 1990.
Article5. Pillon D, Caraty A, Fabre-Nys C, Lomet D, Cateau M, Bruneau G. Regulation by estradiol of hypothalamic somatostatin gene expression: possible involvement of somatostatin in the control of luteinizing hormone secretion in the ewe. Biol Reprod. 71:38–44. 2004.
Article6. Van Vugt HH, Swarts HJ, Van de Heijning BJ, Van der Beek EM. Cen-trally applied somatostatin inhibits the estrogen-induced luteinizing hormone surge via hypothalamic gonadotropin-releasing hormone cell activation in female rats. Biol Reprod. 71:813–819. 2004.7. Herbison AE. Multimodal influence of estrogen upon gonadotropin-releas-ing hormone neurons. Endocr Rev. 19:302–330. 1998.
Article8. Flanagan-Cato LM. Estrogen-induced remodeling of hypothalamic neural circuitry. Front Neuroendocrinol. 21:309–329. 2000.
Article9. Robinson JE, Birch RA, Grindrod JA, Taylor JA, Unsworth WP. Sexually differentiated regulation of gnRH release by gonadal steroid hormones in sheep. Reprod Suppl. 61:299–310. 2003.
Article10. Bhattarai JP, Kaszas A, Park SA, Yin H, Park SJ, Herbison AE, Han SK, Abraham IM. Somatostatin inhibition of gonadotropin-releasing hormone neurons in female and male mice. Endocrinology. 151:3258–3266. 2010.
Article11. Rozen S, Skaletsky H. Primer3 on the WWW for general users and for bi-ologist programmers. Misener S, Krawetz S, editors. Bioinformatics methods and protocols: methods in molecular biology. p. 365–386. Totowa: Humana Press;2000.
Article12. Rhee JS, Ebihara S, Akaike N. Gramicidin perforated patch-clamp tech-nique reveals glycine-gated outward chloride current in dissociated nucleus solitarii neurons of the rat. J Neurophysiol. 72:1103–1108. 1994.
Article13. Han SK, Herbison AE. Norepinephrine suppresses gonadotropin-releasing hormone neuron excitability in the adult mouse. Endocrinology. 149:1129–1135. 2008.
Article14. Mollenholt P, Rawal N, Gordh T Jr, Olsson Y. Intrathecal and epidural somatostatin for patients with cancer. Analgesic effects and postmortem neuropathologic investigations of spinal cord and nerve roots. Anesthesiol-ogy. 81:534–542. 1994.15. Jiang N, Furue H, Katafuchi T, Yoshimura M. Somatostatin directly inhibits substantia gelatinosa neurons in adult rat spinal dorsal horn in vitro. Neu-rosci Res. 47:97–107. 2003.
Article16. Takeda M, Kadoi J, Takahashi M, Nasu M, Matsumoto S. Somatostatin inhibits the excitability of rat small-diameter trigeminal ganglion neurons that innervate nasal mucosa and project to the upper cervical dorsal horn via activation of somatostatin 2a receptor. Neuroscience. 148:744–756. 2007.
Article17. Pittman QJ, Siggins GR. Somatostatin hyperpolarizes hippocampal pyra-midal cells in vitro. Brain Res. 221:402–408. 1981.
Article18. Moore SD, Madamba SG, Joels M, Siggins GR. Somatostatin augments the M-current in hippocampal neurons. Science. 239:278–280. 1988.
Article19. Yin H, Lee KE, Park SA, Bhattarai JP, Suh BJ, Jeon JG, Kim BG, Park SJ, Han SK. Inhibitory effects of somatostatin on the substantia gelatinosa neurons of trigeminal subnucleus caudalis via somatostatin type 2 receptors in juvenile mice. Brain Res. 1304:49–56. 2009.
Article20. Lefkowitz RJ. G protein-coupled receptors. III. New roles for receptor kinases and beta-arrestins in receptor signaling and desensitization. J Biol Chem. 273:18677–18680. 1998.21. Bohm SK, Grady EF, Bunnett NW. Regulatory mechanisms that modu-late signalling by G-protein-coupled receptors. Biochem J. 322(Pt 1):1–18. 1997.22. Premont RT, Inglese J, Lefkowitz RJ. Protein kinases that phosphorylate activated G protein-coupled receptors. FASEB J. 9:175–182. 1995.
Article23. Horie K, Insel PA. Retrovirally mediated transfer of a G protein-coupled receptor kinase (GRK) dominant-negative mutant enhances endogenous calcitonin receptor signaling in Chinese hamster ovary cells. GRK inhibition enhances expression of receptors and receptor mRNA. J Biol Chem. 275:29433–29440. 2000.24. Yoon SH, Jin W, Spencer RJ, Loh HH, Thayer SA. Desensitization of del-ta-opioid-induced mobilization of Ca2+ stores in NG108-15 cells. Brain Res. 802:9–18. 1998.25. Kramer HK, Simon EJ. Role of protein kinase C (PKC) in agonist-induced mu-opioid receptor down-regulation: I. PKC translocation to the membrane of SH-SY5Y neuroblastoma cells is induced by mu-opioid agonists. J Neurochem. 72:585–593. 1999.26. Rapacciuolo A, Suvarna S, Barki-Harrington L, Luttrell LM, Cong M, Lefkowitz RJ, Rockman HA. Protein kinase A and G protein-coupled receptor kinase phosphorylation mediates beta-1 adrenergic receptor endocy-tosis through different pathways. J Biol Chem. 278:35403–35411. 2003.27. Holliday ND, Tough IR, Cox HM. A functional comparison of recombi-nant and native somatostatin sst2 receptor variants in epithelia. Br J Pharmacol. 152:132–140. 2007.
Article28. Young Shim E, Jung Kim H, Kim MJ, Rhie DJ, Jo YH, Kim MS, June Hahn S, Lee MY, Yoon SH. Desensitization of somatostatin-induced inhibition of low extracellular magnesium concentration-induced calcium spikes in cultured rat hippocampal neurons. Brain Res. 1111:61–71. 2006.
Article29. Wang HL, Dichter M, Reisine T. Lack of cross-desensitization of somatosta-tin-14 and somatostatin-28 receptors coupled to potassium channels in rat neocortical neurons. Mol Pharmacol. 38:357–361. 1990.30. Hoyer D, Bell GI, Berelowitz M, Epelbaum J, Feniuk W, Humphrey PP, O'Carroll AM, Patel YC, Schonbrunn A, Taylor JE, Reisine T. Classifica-tion and nomenclature of somatostatin receptors. Trends Pharmacol Sci. 16:86–88. 1995.
Article31. Patel YC. Somatostatin and its receptor family. Front Neuroendocrinol. 20:157–198. 1999.
Article32. Schonbrunn A, Gu YZ, Dournard P, Beaudet A, Tannenbaum GS, Brown PJ. Somatostatin receptor subtypes: specific expression and signaling prop-erties. Metabolism. 45:8–11. 1996.
Article33. Schwanzel-Fukuda M, Pfaff DW. The migration of luteinizing hormone-releasing hormone (LHRH) neurons from the medial olfactory placode into the medial basal forebrain. Experientia. 46:956–962. 1990.34. Han SK, Abraham IM, Herbison AE. Effect of GABA on GnRH neurons switches from depolarization to hyperpolarization at puberty in the female mouse. Endocrinology. 143:1459–1466. 2002.
Article35. Han SK, Todman MG, Herbison AE. Endogenous GABA release inhibits the firing of adult gonadotropin-releasing hormone neurons. Endocrinology. 145:495–499. 2004.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The mechanism of Arginine-stimulated growth hormone secretion
- Response of Human Chorionic Gonadotropin to 6-D-Trp-Gonadotropin-Releasing Hormone and Gonadotropin-Releasing Hormone Stimulation in the Culture Media of Normal Human Placenta of Different Gestational Ages
- GnRH pre-mRNA Splicing in Hypogonadal Mice
- The Role of Gonadotropin: Releasing Hormone ( GnRH ) and Its Receptor in the Extrapituitary Tissues
- Immunohistochemical study on the somatostatin and corticotropin-releasing factor neurons in the hypothalamus of the dog