Anesth Pain Med.
2016 Jan;11(1):1-13. 10.17085/apm.2016.11.1.1.
An overview of pharmacodynamic drug interaction with isobole and response surface model in anesthesia
- Affiliations
-
- 1Department of Anesthesiology and Pain Medicine, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea. byungmoonchoi7@gmail.com
- KMID: 2169061
- DOI: http://doi.org/10.17085/apm.2016.11.1.1
Abstract
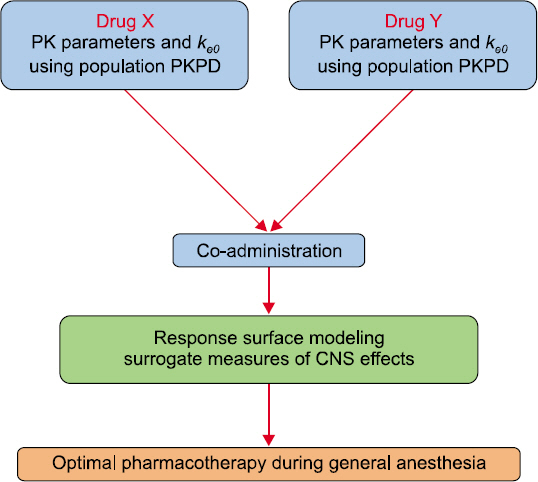
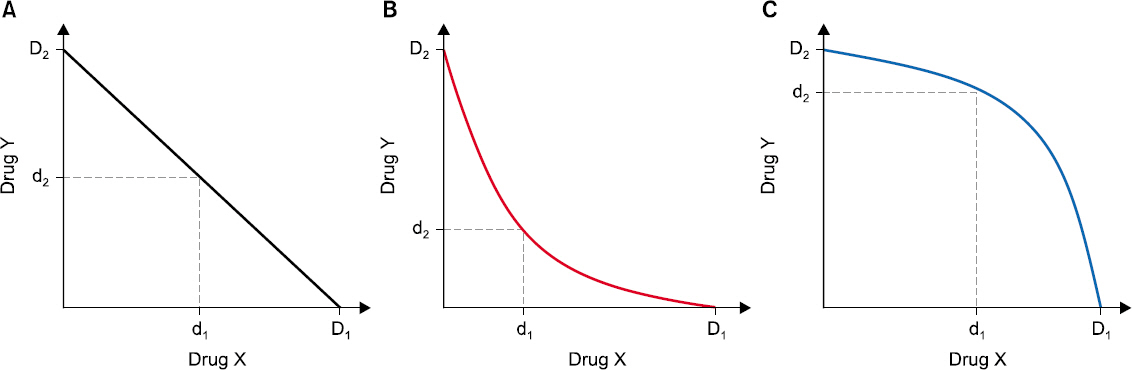
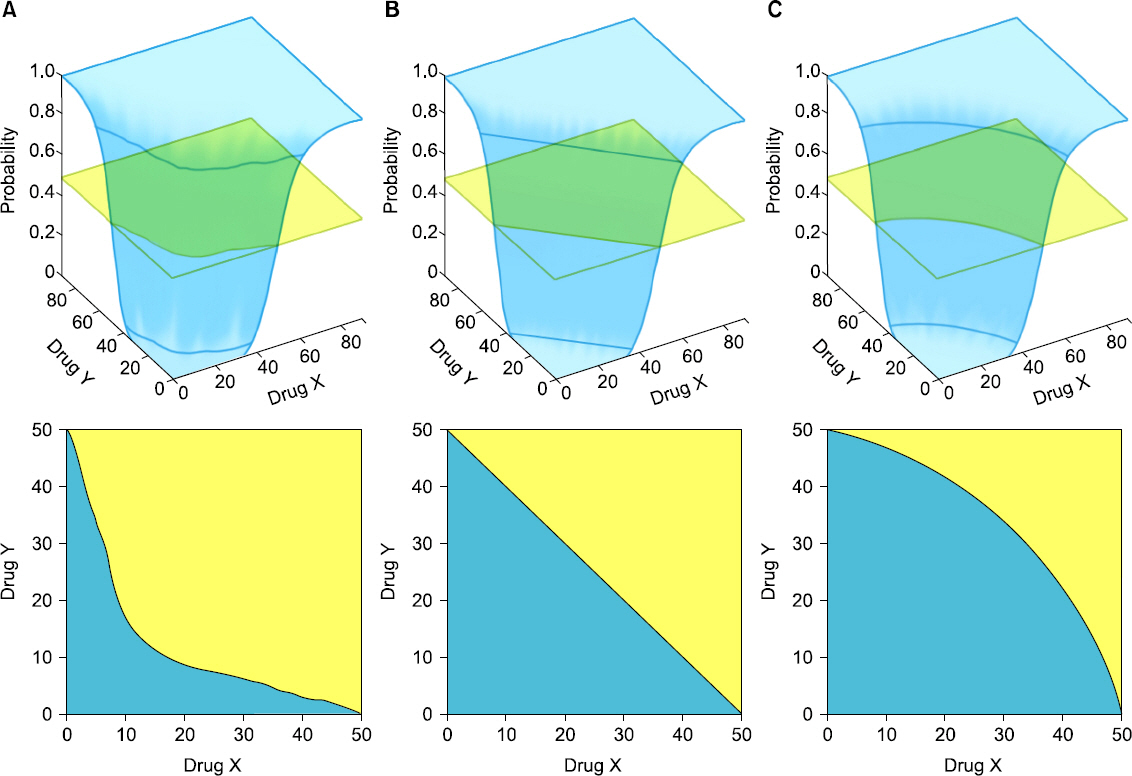
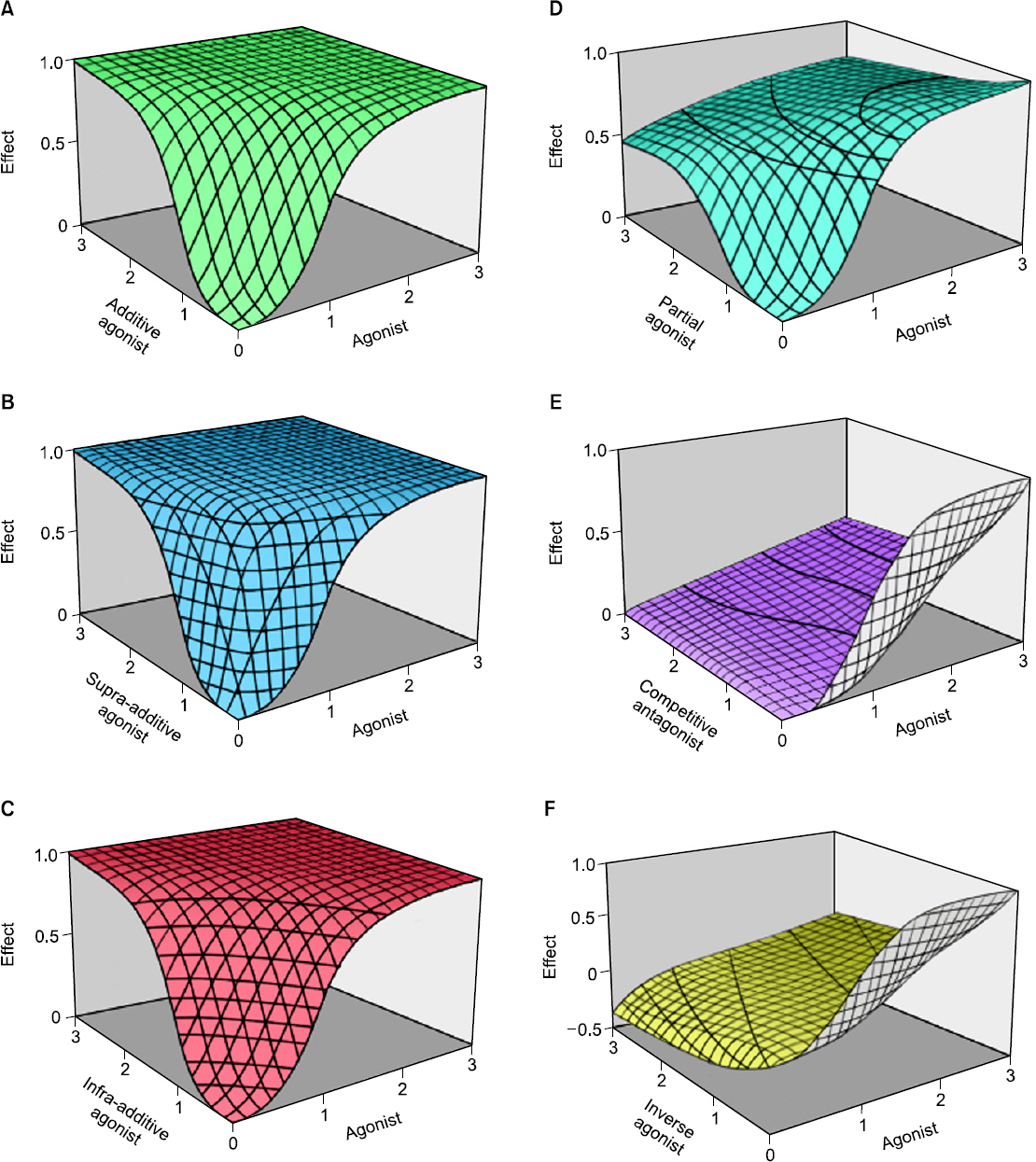
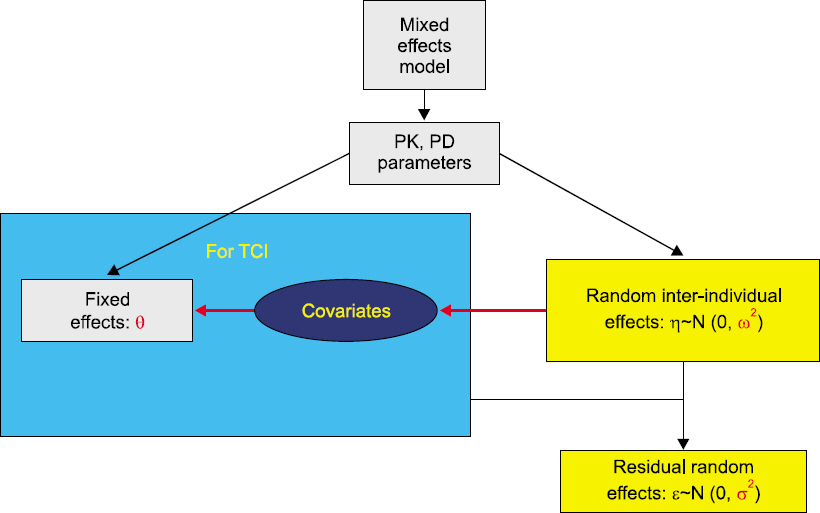
- Drug interaction is the principal concept of anesthetic practice. Typically, drug interactions are divided into three categories i.e., additive, synergistic, or infra-additive interactions. Pharmacodynamic drug interactions are typically described using mathematical models. The traditional model is an isobole, which is an iso-effect curve that shows dose or concentration combinations that result in equal effect. Response surface model is a pharmacodynamic tool that describes all isoboles and concentration effect curves for a given endpoint in one equation. In summarizing concentration-effect relationships, the response surface model allows anesthesiologists the versatility to work with precise and safe drug interactions. The aim of this review is to provide the reader with principal concepts of the isobole and response surface model and evaluate characteristics of most commonly used models, including 4 response surface models i.e., the Greco model, reduced Greco model, Minto model, and the Hierarchy model. In addition, the concept of population analysis using nonlinear mixed effects modeling is introduced.
Keyword
Figure
Reference
-
1. Hendrickx JF, Eger EI 2nd, Sonner JM, Shafer SL. Is synergy the rules? A review of anesthetic interactions producing hypnosis and immobility. Anesth Analg. 2008; 107:494–506. DOI: 10.1213/ane.0b013e31817b859e. PMID: 18633028.2. Minto CF, Schnider TW, Short TG, Gregg KM, Gentilini A, Shafer SL. Response surface model for anesthetic drug interactions. Anesthesiology. 2000; 92:1603–16. DOI: 10.1097/00000542-200006000-00017. PMID: 10839909.
Article3. Greco WR, Bravo G, Parsons JC. The search for synergy: a critical review from a response surface perspective. Pharmacol Rev. 1995; 47:331–85. PMID: 7568331.4. Chou TC. Derivation and properties of Michaelis-Menten type and Hill type equations for reference ligands. J Theor Biol. 1976; 59:253–76. DOI: 10.1016/0022-5193(76)90169-7.
Article5. Lee SI. Drug interaction: focusing on response surface models. Korean J Anesthesiol. 2010; 58:421–34. DOI: 10.4097/kjae.2010.58.5.421. PMID: 20532049. PMCID: PMC2881515.
Article6. Liou JY, Tsou MY, Ting CK. Response surface models in the field of anesthesia: A crash course. Acta Anaesthesiol Taiwan. 2015; 53:139–45. DOI: 10.1016/j.aat.2015.06.005. PMID: 26321504.
Article7. Mertens MJ, Olofsen E, Engbers FH, Burm AG, Bovill JG, Vuyk J. Propofol reduces perioperative remifentanil requirements in a synergistic manner: response surface modeling of perioperative remifentanil-propofol interactions. Anesthesiology. 2003; 99:347–59. DOI: 10.1097/00000542-200308000-00016. PMID: 12883407.8. Heyse B, Proost JH, Schumacher PM, Bouillon TW, Vereecke HE, Eleveld DJ, et al. Sevoflurane remifentanil interaction: comparison of different response surface models. Anesthesiology. 2012; 116:311–23. DOI: 10.1097/ALN.0b013e318242a2ec. PMID: 22222473.9. Kissin I. General anesthetic action: an obsolete notion? Anesth Analg. 1993; 76:215–8. DOI: 10.1213/00000539-199302000-00002. PMID: 8424493.10. Glass PS. Anesthetic drug interactions: an insight into general anesthesia--its mechanism and dosing strategies. Anesthesiology. 1998; 88:5–6. DOI: 10.1097/00000542-199801000-00004. PMID: 9447849.11. Bouillon TW, Bruhn J, Radulescu L, Andresen C, Shafer TJ, Cohane C, et al. Pharmacodynamic interaction between propofol and remifentanil regarding hypnosis, tolerance of laryngoscopy, bispectral index, and electroencephalographic approximate entropy. Anesthesiology. 2004; 100:1353–72. DOI: 10.1097/00000542-200406000-00006. PMID: 15166553.
Article12. Short TG, Ho TY, Minto CF, Schnider TW, Shafer SL. Efficient trial design for eliciting a pharmacokinetic-pharmacodynamic model-based response surface describing the interaction between two intravenous anesthetic drugs. Anesthesiology. 2002; 96:400–8. DOI: 10.1097/00000542-200202000-00027. PMID: 11818774.13. Kern SE, Xie G, White JL, Egan TD. A response surface analysis of propofol-remifentanil pharmacodynamic interaction in volunteers. Anesthesiology. 2004; 100:1373–81. DOI: 10.1097/00000542-200406000-00007. PMID: 15166554.
Article14. Manyam SC, Gupta DK, Johnson KB, White JL, Pace NL, Westenskow DR, et al. Opioid-volatile anesthetic synergy: a response surface model with remifentanil and sevoflurane as prototypes. Anesthesiology. 2006; 105:267–78. DOI: 10.1097/00000542-200608000-00009. PMID: 16871060.15. Wang HL, Yang L, Guo XY, Zhang LP, Bi SS, Lu W. Response surface analysis of sevoflurane-remifentanil interactions on consciousness during anesthesia. Chin Med J (Engl). 2012; 125:2682–7.16. Bi SS, Deng CH, Zhou TY, Guan Z, Li L, Li HQ, et al. Remifentanil-sevoflurane interaction models of circulatory response to laryngoscopy and circulatory depression. Br J Anaesth. 2013; 110:729–40. DOI: 10.1093/bja/aes504. PMID: 23388507.
Article17. Heyse B, Proost JH, Hannivoort LN, Eleveld DJ, Luginbuhl M, Struys MM, et al. A response surface model approach for continuous measures of hypnotic and analgesic effect during sevoflurane-remifentanil interaction: quantifying the pharmacodynamic shift evoked by stimulation. Anesthesiology. 2014; 120:1390–9. DOI: 10.1097/ALN.0000000000000180. PMID: 24566244.18. Ting CK, Johnson KB, Teng WN, Synoid ND, Lapierre C, Yu L, et al. Response surface model predictions of wake-up time during scoliosis surgery. Anesth Analg. 2014; 118:546–53. DOI: 10.1213/ane.0000000000000094.
Article19. Johnson KB, Syroid ND, Gupta DK, Manyam SC, Egan TD, Huntington J, et al. An evaluation of remifentanil propofol response surfaces for loss of responsiveness, loss of response to surrogates of painful stimuli and laryngoscopy in patients undergoing elective surgery. Anesth Analg. 2008; 106:471–9. DOI: 10.1213/ane.0b013e3181606c62. PMID: 18227302. PMCID: PMC3050649.
Article20. LaPierre CD, Johnson KB, Randall BR, White JL, Egan TD. An exploration of remifentanil-propofol combinations that lead to a loss of response to esophageal instrumentation, a loss of responsiveness, and/or onset of intolerable ventilatory depression. Anesth Analg. 2011; 113:490–9. DOI: 10.1213/ane.0b013e318210fc45.
Article21. LaPierre CD, Johnson KB, Randall BR, Egan TD. A simulation study of common propofol and propofol-opioid dosing regimens for upper endoscopy: implications on the time course of recovery. Anesthesiology. 2012; 117:252–62. DOI: 10.1097/ALN.0b013e31825fb1b2. PMID: 22728781.22. Luginbuhl M, Schumacher PM, Vuilleumier P, Vereecke H, Heyse B, Bouillon TW, et al. Noxious stimulation response index: a novel anesthetic state index based on hypnotic-opioid interaction. Anesthesiology. 2010; 112:872–80. DOI: 10.1097/ALN.0b013e3181d40368. PMID: 20216387.23. Nieuwenhuijs DJ, Olofsen E, Romberg RR, Sarton E, Ward D, Engbers F, et al. Response surface modeling of remifentanil- propofol interaction on cardiorespiratory control and bispectral index. Anesthesiology. 2003; 98:312–22. DOI: 10.1097/00000542-200302000-00008. PMID: 12552187.24. Glass PS, Gan TJ, Howell S, Ginsberg B. Drug interactions: volatile anesthetics and opioids. J Clin Anesth. 1997; 9(6 Suppl):S18–22. DOI: 10.1016/S0952-8180(97)00122-0.
Article25. McEwan AI, Smith C, Dyar O, Goodman D, Smith LR, Glass PS. Isoflurane minimum alveolar concentration reduction by fentanyl. Anesthesiology. 1993; 78:864–9. DOI: 10.1097/00000542-199305000-00009. PMID: 8489058.
Article26. Shafer SL, Varvel JR. Pharmacokinetics, pharmacodynamics, and rational opioid selection. Anesthesiology. 1991; 74:53–63. DOI: 10.1097/00000542-199101000-00010.
Article27. Egan TD, Minto CF, Hermann DJ, Barr J, Muir KT, Shafer SL. Remifentanil versus alfentanil: comparative pharmacokinetics and pharmacodynamics in healthy adult male volunteers. Anesthesiology. 1996; 84:821–33. DOI: 10.1097/00000542-199604000-00009. PMID: 8638836.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Drug interaction: focusing on response surface models
- Synergism between rocuronium and cisatracurium: comparison of the Minto and Greco interaction models
- Basic Principles of Drug Interaction
- A semi-compartmental model describing the pharmacokinetic-pharmacodynamic relationship
- Neuromuscular blockade and pharmacokinetic-pharmacodynamic modeling